Advanced Manufacturing of High-Performance Antioxidants for Global Polymer Industries
Advanced Manufacturing of High-Performance Antioxidants for Global Polymer Industries
The development of robust stabilization systems for polymers remains a critical challenge in the fine chemical sector, driving continuous innovation in synthetic methodologies. Patent CN101225053A introduces a transformative preparation method for the high-performance antioxidant N,N'-bis(2-hydroxyethyl)oxamide-di[3-(3,5-di-tert-butyl-4-hydroxyphenyl)propionate], a compound essential for preventing thermal and oxidative degradation in engineering plastics. This technology addresses long-standing inefficiencies in production by integrating a direct esterification pathway with a sophisticated raw material recovery system. By shifting away from hazardous acyl chloride intermediates and optimizing the utilization of expensive phenolic acids, this process offers a compelling value proposition for manufacturers seeking both economic efficiency and environmental compliance. The strategic implementation of this patent allows for the production of ultra-high purity additives that meet the rigorous demands of the global polymer market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of this specific class of hindered phenol antioxidants relied heavily on two primary routes, both of which presented significant operational and environmental drawbacks for large-scale manufacturing. The first conventional method involved the activation of 3-(3,5-di-tert-butyl-4-hydroxyphenyl)propionic acid using thionyl chloride to form an acyl chloride intermediate prior to esterification. While chemically effective, this approach generates substantial quantities of corrosive and toxic byproducts, specifically sulfur dioxide and hydrogen chloride gas, necessitating complex and costly scrubbing systems to meet environmental regulations. The second common approach utilized a transesterification reaction between methyl or ethyl esters of the phenolic acid and the diamide core. Although safer than the acyl chloride route, this transesterification method typically suffers from incomplete conversion, resulting in crystallization yields that rarely exceed 85-90%. Furthermore, the mother liquor from these processes contains significant amounts of unreacted starting materials and mono-ester intermediates, representing a direct loss of high-value raw materials and increasing the overall cost of goods sold.
The Novel Approach
The methodology disclosed in patent CN101225053A fundamentally restructures the synthesis workflow to overcome these historical bottlenecks through a streamlined direct esterification coupled with a closed-loop recovery system. Instead of pre-activating the acid or using pre-formed esters, the process employs a direct condensation between the free carboxylic acid and N,N'-bis(2-hydroxyethyl)oxalamide in the presence of a specialized monobutyltin stannonic acid catalyst. This reaction is conducted in aromatic solvents like xylene or toluene under azeotropic conditions to continuously remove water, driving the equilibrium toward the desired di-ester product. Crucially, the innovation extends beyond the primary reaction; the process includes a dedicated downstream treatment of the crystallization mother liquor. By subjecting the filtrate to alkaline hydrolysis followed by acidification, the unreacted phenolic acid is quantitatively recovered and recycled back into the production line. This dual focus on efficient catalysis and material recovery ensures that the theoretical yield limits of traditional methods are surpassed, delivering a commercially superior manufacturing protocol.
Mechanistic Insights into Tin-Catalyzed Direct Esterification
The core chemical transformation in this patented process relies on the Lewis acidic properties of the monobutyltin stannonic acid catalyst, which activates the carbonyl group of the 3-(3,5-di-tert-butyl-4-hydroxyphenyl)propionic acid towards nucleophilic attack by the hydroxyl groups of the oxalamide derivative. Operating at temperatures between 120°C and 180°C, the catalyst facilitates the formation of the tetrahedral intermediate necessary for ester bond formation while simultaneously promoting the elimination of water. The use of xylene or toluene serves a dual purpose: acting as a high-boiling solvent to maintain the reaction temperature and forming an azeotrope with the generated water, which is removed via a Dean-Stark trap or similar apparatus. This continuous removal of water is thermodynamically essential to push the reversible esterification reaction to completion without requiring a massive excess of reagents. The specificity of the tin catalyst also minimizes side reactions, such as the degradation of the sensitive phenolic hydroxyl groups or the formation of oligomeric byproducts, thereby ensuring the structural integrity of the final antioxidant molecule.
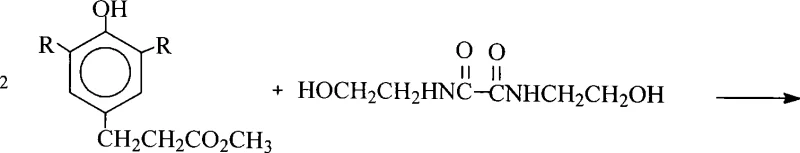
Beyond the primary synthesis, the mechanism of impurity control and yield enhancement is anchored in the post-crystallization treatment of the mother liquor. In standard processes, the mother liquor is often discarded or subjected to energy-intensive distillation to recover solvents, leaving valuable organic residues behind. In this novel approach, the residue is treated with aqueous sodium hydroxide at elevated temperatures (100-110°C), which hydrolyzes any mono-esters or unreacted di-esters back into the sodium salt of the phenolic acid. Subsequent acidification with sulfuric acid precipitates the free acid, which can be filtered, dried, and reused. This chemical recycling loop effectively converts what would be waste into feedstock, drastically altering the mass balance of the process. By recovering the 3-(3,5-di-tert-butyl-4-hydroxyphenyl)propionic acid, the process not only improves the isolated yield of the final antioxidant to over 96% based on the acid but also significantly reduces the impurity profile, as the recycled acid can be purified before re-entry into the reactor.
How to Synthesize N,N'-bis(2-hydroxyethyl)oxamide-di[3-(3,5-di-tert-butyl-4-hydroxyphenyl)propionate] Efficiently
The execution of this synthesis requires precise control over stoichiometry and thermal parameters to maximize the efficiency of the tin-catalyzed esterification. The process begins with the charging of the reactor with the aromatic solvent, followed by the addition of the diamide and the phenolic acid in a molar ratio slightly favoring the acid to drive conversion. The catalyst is introduced, and the mixture is heated to reflux to initiate water removal. Once the reaction is complete, indicated by the cessation of water collection, the mixture undergoes a decolorization step using activated carbon or attapulgite clay to remove trace colored impurities and catalyst residues. The hot filtrate is then concentrated under reduced pressure to remove the solvent, yielding a melt that is subsequently crystallized by the addition of lower alcohols such as isopropanol or ethanol. The detailed standardized operating procedures for temperature ramping, vacuum levels, and crystallization kinetics are critical for reproducibility and are outlined in the technical guide below.
- Conduct azeotropic esterification of 3-(3,5-di-tert-butyl-4-hydroxyphenyl)propionic acid and N,N'-bis(2-hydroxyethyl)oxalamide using monobutyltin stannonic acid catalyst in xylene or toluene at 120-180°C.
- Decolorize the reaction mixture with activated carbon or attapulgite clay, filter, and remove the solvent under reduced pressure to obtain the melt.
- Crystallize the product using alcohol solvents, then recover unreacted acid from the mother liquor via alkaline hydrolysis and acidification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology translates directly into enhanced operational resilience and cost competitiveness. The most significant economic driver is the drastic reduction in raw material consumption achieved through the internal recycling loop. By recovering and reusing the expensive 3-(3,5-di-tert-butyl-4-hydroxyphenyl)propionic acid from the mother liquor, the effective cost per kilogram of the final antioxidant is substantially lowered compared to processes that discard these residues. This efficiency gain provides a buffer against volatility in the pricing of phenolic acid precursors, allowing for more stable long-term contracting and budget forecasting. Furthermore, the elimination of toxic gas byproducts like sulfur dioxide removes the need for expensive scrubbing infrastructure and hazardous waste disposal services, contributing to a leaner cost structure.
- Cost Reduction in Manufacturing: The implementation of the recovery cycle fundamentally changes the cost dynamics of antioxidant production. In traditional transesterification routes, the loss of unreacted acid in the mother liquor represents a direct financial drain, often requiring the purchase of 10-15% excess raw material to compensate for yield losses. This new method captures that value, effectively turning waste streams into revenue-generating assets. Additionally, the use of a reusable organotin catalyst in relatively low loadings reduces the dependency on stoichiometric activating agents like thionyl chloride, which are not only costly but also require specialized handling and storage protocols. The cumulative effect is a manufacturing process with a significantly lower variable cost profile, enabling competitive pricing strategies in the global polymer additive market without compromising margin.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of multi-step syntheses and the regulatory hurdles associated with hazardous intermediates. By simplifying the synthetic route to a direct esterification, the number of unit operations is reduced, decreasing the probability of equipment failure or batch rejection. The reliance on commodity solvents like xylene and toluene, rather than specialized chlorinating agents, ensures that the supply chain is less vulnerable to disruptions in niche chemical markets. Moreover, the ability to recover starting materials internally acts as a buffer; if there is a temporary shortage of fresh phenolic acid, the inventory of recovered acid can sustain production for a period, thereby enhancing the overall reliability of supply for downstream polymer manufacturers who depend on just-in-time delivery schedules.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies environmental risks, but this technology is inherently designed for green manufacturing. The absence of corrosive gas emissions simplifies the permitting process for new production facilities and reduces the ongoing regulatory burden associated with air quality monitoring. The solvent recovery steps are integrated seamlessly into the workflow, minimizing volatile organic compound (VOC) emissions. From a scalability perspective, the reaction conditions (120-180°C) are well within the operating range of standard glass-lined or stainless steel reactors found in most fine chemical plants, meaning that technology transfer from pilot scale to commercial tonnage can be achieved rapidly without the need for exotic metallurgy or high-pressure equipment. This ease of scale-up ensures that supply can be ramped up quickly to meet surging demand in the automotive and packaging sectors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this antioxidant synthesis technology. These insights are derived directly from the experimental data and claims presented in patent CN101225053A, providing a clear understanding of the process capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this route for their own production lines or for procurement specialists assessing the quality consistency of suppliers utilizing this method.
Q: How does this patent improve yield compared to traditional transesterification methods?
A: Traditional methods often suffer from yields around 85-90% due to unreacted materials remaining in the mother liquor. This patented process introduces a specific recovery loop where the mother liquor is hydrolyzed to reclaim the expensive 3-(3,5-di-tert-butyl-4-hydroxyphenyl)propionic acid, pushing the overall yield based on this acid to over 96%.
Q: What catalyst is used in this novel esterification process?
A: The process utilizes monobutyltin stannonic acid as a highly efficient catalyst. This organotin catalyst facilitates the direct esterification between the carboxylic acid and the hydroxyethyl groups under azeotropic conditions, eliminating the need for toxic acyl chlorides.
Q: Does this method offer environmental advantages over the thionyl chloride route?
A: Yes, significantly. Older methods involving thionyl chloride generate hazardous byproducts like sulfur dioxide and hydrogen chloride gas. This direct esterification method avoids these toxic emissions entirely, aligning better with modern green chemistry standards and reducing waste treatment costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N,N'-bis(2-hydroxyethyl)oxamide-di[3-(3,5-di-tert-butyl-4-hydroxyphenyl)propionate] Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires more than just chemical knowledge; it demands engineering excellence and rigorous quality assurance. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the efficiencies promised by patent CN101225053A are fully realized in our manufacturing suites. Our facilities are equipped with state-of-the-art reactors capable of handling the precise thermal profiles required for tin-catalyzed esterifications, and our stringent purity specifications guarantee that every batch of antioxidant meets the exacting standards of the global polymer industry. With our rigorous QC labs, we monitor every stage of the synthesis, from raw material intake to final crystallization, to ensure consistent performance in your end applications.
We invite you to collaborate with us to optimize your supply chain for high-performance polymer additives. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our implementation of this recovery-focused synthesis can reduce your total cost of ownership. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments, allowing you to validate the superior quality and economic advantages of our manufacturing process before making any commitment. Let us be your strategic partner in delivering stability and value to your polymer products.
