Advanced Synthesis of Oxamide-Based Antioxidants: A Technical Breakthrough for Polymer Stability
Advanced Synthesis of Oxamide-Based Antioxidants: A Technical Breakthrough for Polymer Stability
The global demand for high-performance polymer stabilizers continues to surge, driven by the need for durable materials in automotive, packaging, and construction sectors. A pivotal advancement in this field is documented in patent CN101225053A, which details a superior preparation method for N,N'-bis(2-hydroxyethyl)oxamide-di[3-(3,5-di-tert-butyl-4-hydroxyphenyl)propionate]. This specific antioxidant structure represents a critical class of hindered phenol stabilizers known for their exceptional thermal stability and resistance to oxidation. Unlike legacy synthetic routes that rely on hazardous chlorinating agents or inefficient transesterification processes, this patented methodology introduces a streamlined, catalytic esterification pathway coupled with an innovative raw material recovery loop. For R&D directors and procurement strategists, understanding this shift is vital, as it promises not only higher purity profiles exceeding 98% but also a fundamentally more sustainable and cost-effective supply chain for essential polymer additives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex oxamide-based antioxidants has been plagued by significant environmental and efficiency challenges. The earliest industrial methods, such as those referenced in US Patent 4145556, often employed thionyl chloride to activate the carboxylic acid prior to amidation or esterification. This approach generates substantial quantities of corrosive hydrogen chloride gas and toxic sulfur dioxide as byproducts, necessitating expensive and energy-intensive scrubbing infrastructure to meet modern environmental regulations. Furthermore, alternative transesterification routes using methyl or ethyl esters of the phenolic acid frequently suffer from equilibrium limitations. These older processes often require a large excess of starting materials to drive the reaction forward, resulting in low atom economy and difficult purification steps to remove unreacted esters and mono-ester impurities. Consequently, the final product often requires extensive recrystallization, leading to yield losses and increased production costs that are ultimately passed down the supply chain.
The Novel Approach
The methodology outlined in CN101225053A represents a paradigm shift by utilizing direct catalytic esterification between the free acid and the hydroxy-functionalized oxamide. By employing monobutyltin stannonic acid as a highly effective catalyst in solvents like xylene or toluene, the reaction proceeds via azeotropic dehydration at temperatures between 120°C and 180°C. This eliminates the need for pre-activation with toxic reagents and avoids the equilibrium issues of transesterification. Crucially, the process incorporates a closed-loop recovery system where the mother liquor from the final crystallization is not discarded as waste. Instead, it undergoes alkaline hydrolysis followed by acidification to reclaim the valuable 3-(3,5-di-tert-butyl-4-hydroxyphenyl)propionic acid. This recovered acid is then recycled back into the reactor, ensuring that raw material utilization approaches theoretical maximums.
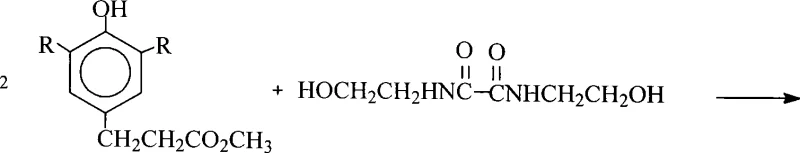
Mechanistic Insights into Monobutyltin-Catalyzed Esterification
The core of this technological breakthrough lies in the specific catalytic mechanism facilitated by the organotin compound. Monobutyltin stannonic acid acts as a Lewis acid catalyst, coordinating with the carbonyl oxygen of the carboxylic acid to increase its electrophilicity. This activation lowers the energy barrier for the nucleophilic attack by the hydroxyl groups of the N,N'-bis(2-hydroxyethyl)oxalamide. Unlike homogeneous acid catalysts which can be difficult to separate and may promote side reactions like ether formation or phenolic degradation, the tin catalyst in this system demonstrates remarkable selectivity. It promotes the formation of the di-ester while minimizing the formation of mono-ester intermediates or oligomeric byproducts. The reaction conditions, specifically the azeotropic removal of water using a Dean-Stark trap or similar apparatus in refluxing xylene, continuously shift the equilibrium towards the product side according to Le Chatelier's principle, driving the conversion to near completion without the need for excessive reagent loading.
Furthermore, the impurity control mechanism is intrinsically linked to the workup procedure described in the patent. Following the reaction, the addition of activated carbon or attapulgite clay at elevated temperatures (120-130°C) serves a dual purpose: it adsorbs colored impurities and residual catalyst species, and it helps coagulate fine particulates. The subsequent filtration at 80-100°C ensures that the filtrate is clear before solvent removal. The crystallization step, performed by adding alcohols like isopropanol or ethanol to the molten residue, is carefully controlled at 20-40°C to maximize the precipitation of the pure di-ester while keeping soluble impurities in the solution. This precise thermal management during crystallization is what allows the process to achieve purity levels of 98.5% to 98.9% consistently, as evidenced by the HPLC data in the patent examples, making it suitable for sensitive applications in food-contact plastics and medical devices.
How to Synthesize N,N'-bis(2-hydroxyethyl)oxamide-di[3-(3,5-di-tert-butyl-4-hydroxyphenyl)propionate] Efficiently
To replicate the high yields and purity described in the patent, strict adherence to the molar ratios and thermal profiles is essential. The process begins with the precise charging of 3-(3,5-di-tert-butyl-4-hydroxyphenyl)propionic acid and N,N'-bis(2-hydroxyethyl)oxalamide in a molar ratio of approximately 2.00-2.30 to 1.00. The detailed standardized synthesis steps, including specific heating rates, vacuum levels for solvent recovery, and the exact protocol for the acid recovery loop, are critical for maintaining batch-to-batch consistency. Operators must ensure that the dehydration phase is complete before proceeding to the decolorization step to prevent hydrolysis of the product during workup. The following guide outlines the operational framework derived from the patent embodiments.
- Combine 3-(3,5-di-tert-butyl-4-hydroxyphenyl)propionic acid and N,N'-bis(2-hydroxyethyl)oxalamide in xylene or toluene with a monobutyltin catalyst.
- Heat the mixture to 120-180°C for azeotropic dehydration to drive the esterification reaction to completion.
- Purify the crude melt via decolorization with activated carbon, followed by crystallization and a unique acid recovery loop from the mother liquor.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis route offers tangible strategic benefits beyond mere technical specifications. The elimination of thionyl chloride and the associated gas scrubbing requirements significantly simplifies the manufacturing infrastructure, reducing both capital expenditure (CAPEX) for new plants and operational expenditure (OPEX) for maintenance and safety compliance. Moreover, the implementation of the acid recovery loop transforms what was previously a waste stream into a valuable asset. By recovering and reusing the expensive phenolic acid starting material, the effective consumption of raw materials per kilogram of finished antioxidant is drastically reduced. This internal recycling mechanism acts as a hedge against volatility in the pricing of petrochemical-derived phenolic acids, providing greater cost stability and predictability for long-term supply contracts.
- Cost Reduction in Manufacturing: The economic model of this process is heavily favored by the high atom economy and the recycling capability. By avoiding the use of stoichiometric activating agents like thionyl chloride, the process eliminates the cost of purchasing these reagents and the cost of disposing of the resulting salt wastes. Additionally, the recovery of unreacted acid from the mother liquor means that the effective yield based on consumed raw materials is exceptionally high, often exceeding 96%. This efficiency translates directly into a lower cost of goods sold (COGS), allowing suppliers to offer more competitive pricing without sacrificing margin. The use of common, recyclable solvents like xylene and toluene further enhances the economic viability, as solvent recovery rates are high and replacement costs are low compared to specialized polar aprotic solvents.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by regulatory crackdowns on polluting processes. Since this method avoids the generation of sulfur dioxide and hydrogen chloride, it is far less susceptible to production shutdowns mandated by environmental agencies. The robustness of the tin-catalyzed reaction also means that batch failures due to sensitivity to moisture or temperature fluctuations are minimized. The process tolerances are wide enough to accommodate standard industrial equipment, reducing the risk of bottlenecks. Furthermore, the starting materials—3-(3,5-di-tert-butyl-4-hydroxyphenyl)propionic acid and the oxalamide derivative—are commodity chemicals with established supply chains, ensuring that raw material availability remains stable even during market disruptions.
- Scalability and Environmental Compliance: Scaling this process from pilot plant to multi-tonne production is straightforward because it relies on unit operations familiar to any fine chemical manufacturer: reflux, distillation, filtration, and crystallization. There are no exotic high-pressure or cryogenic steps that would require specialized reactors. From an environmental standpoint, the process aligns perfectly with Green Chemistry principles. The reduction of hazardous waste, the elimination of toxic gas emissions, and the recycling of solvents and starting materials significantly lower the facility's environmental footprint. This compliance not only mitigates regulatory risk but also enhances the brand value of the end-product, appealing to downstream customers who are increasingly demanding sustainably sourced polymer additives for their own ESG reporting.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of this advanced antioxidant. These answers are synthesized from the detailed experimental data and claims found within the patent documentation, providing clarity on the process capabilities and product quality. Understanding these nuances is essential for technical buyers evaluating this material for high-specification applications.
Q: How does the new process improve upon traditional thionyl chloride methods?
A: Traditional methods utilizing thionyl chloride generate hazardous byproducts like sulfur dioxide and hydrogen chloride gas, requiring complex scrubbing systems. The novel catalytic esterification route eliminates these toxic emissions entirely, offering a greener and safer manufacturing profile.
Q: What is the significance of the acid recovery step in this synthesis?
A: The process includes a critical hydrolysis and acidification step for the crystallization mother liquor. This allows for the quantitative recovery of unreacted 3-(3,5-di-tert-butyl-4-hydroxyphenyl)propionic acid, drastically reducing raw material waste and lowering the overall cost of goods sold.
Q: Can this synthesis be scaled for industrial polymer additive production?
A: Yes, the use of common solvents like xylene or toluene and standard unit operations such as azeotropic distillation and filtration makes this route highly scalable. The robustness of the monobutyltin catalyst ensures consistent yields suitable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable N,N'-bis(2-hydroxyethyl)oxamide-di[3-(3,5-di-tert-butyl-4-hydroxyphenyl)propionate] Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to superior manufacturing processes is key to maintaining a competitive edge in the global polymer additives market. While the patent CN101225053A highlights the potential of this specific esterification route, our team possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring such innovations to life. We are equipped with rigorous QC labs and stringent purity specifications that ensure every batch of antioxidant meets the highest international standards, whether for polyolefin stabilization or engineering plastics. Our commitment to technical excellence means we can adapt this catalytic chemistry to meet your specific volume and purity requirements efficiently.
We invite you to collaborate with us to optimize your supply chain for high-performance stabilizers. By leveraging our expertise in process optimization, we can provide a Customized Cost-Saving Analysis tailored to your specific consumption patterns. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us demonstrate how our advanced manufacturing capabilities can deliver the reliability and quality your organization demands for its next generation of polymer products.
