Advanced Synthetic Route for Desoximetasone: Enhancing Purity and Scalability for Global API Markets
Introduction to Patent CN101397322B
The pharmaceutical industry continuously seeks robust synthetic pathways for potent glucocorticoids like desoximetasone, a critical active pharmaceutical ingredient used extensively in dermatological treatments for eczema and psoriasis. Patent CN101397322B discloses a groundbreaking preparation method that fundamentally restructures the synthetic logic for this steroid compound. Unlike historical approaches that relied on scarce and complex starting materials, this innovation leverages 1,4,9,16-tetraene-pregnadiene-3,20-dione as a strategic initiator. This specific precursor allows for a highly concise operational path involving key transformations at the 16,17, 9,11, and 21 positions. The technical breakthrough lies not merely in the chemical steps but in the strategic sequencing that maximizes yield while minimizing the reliance on expensive auxiliary reagents. For global procurement and R&D teams, this patent represents a significant shift towards more sustainable and economically viable manufacturing of high-purity corticosteroid intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of desoximetasone was dominated by the route developed by Russell Co., Ltd., as detailed in U.S. Patent 3099654A1. This conventional methodology utilized 16α-Methyl-11α,21-dihydroxy-1,4-pregnadiene-3,20-dione-21-acetate as the primary starting raw material. A critical bottleneck of this legacy process is the extreme difficulty in sourcing this specific starting material, which itself requires a cumbersome nine-step synthesis from 3α-hydroxy-16α-Methyl-pregnane-11,20-dione. Consequently, the overall synthetic route becomes excessively long and operationally complex. The cumulative effect of these multiple steps results in a disappointingly low overall yield, reported to be approximately 28% from start to finish. Such inefficiency imposes severe constraints on industrialization potential and makes cost control nearly impossible for large-scale API production.
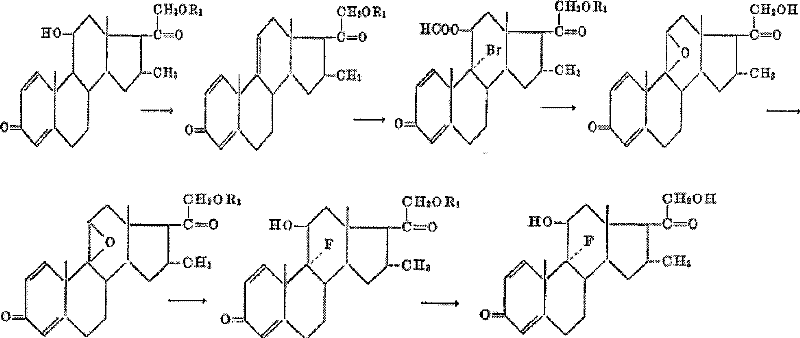
The Novel Approach
In stark contrast, the novel approach outlined in the patent data utilizes a company-existing intermediate, 1,4,9,16-tetraene-pregnadiene-3,20-dione (referenced in CN1896090), which is far more accessible and commercially viable. This new strategy streamlines the synthesis into a direct sequence involving 16,17-Grignard methylation, 9,11-epoxidation and ring-opening, and 21-esterification followed by hydrolysis. By bypassing the need for the difficult-to-obtain 16α-Methyl-11α,21-dihydroxy precursor, the new route eliminates several upstream purification burdens. The simplicity of the circuit means that raw materials are easier to obtain, and the process avoids the use of costly auxiliary materials that plagued previous methods. This structural optimization of the synthetic pathway ensures that both yield and cost metrics are obviously superior to the historical synthetic methods, facilitating a more robust supply chain for downstream drug manufacturers.
Mechanistic Insights into Steroid Functionalization and Grignard Methylation
The core chemical innovation involves a precise sequence of functional group manipulations on the steroid nucleus to achieve the required 9α-fluoro-11β-hydroxy-16α-methyl configuration. The process initiates with a 9,11-epoxidation reaction where the tetraene-dione is treated with a halide reagent, such as N-bromosuccinimide (NBS), in a polar organic solvent like acetone or tetrahydrofuran. This is followed by an alkaline treatment to form the stable 9,11-epoxy intermediate. Crucially, the subsequent 16,17-Grignard reaction introduces the 16α-methyl group using methyl magnesium bromide in the presence of a copper catalyst, such as cuprous chloride. Performing this methylation on the epoxy intermediate is mechanistically superior because the epoxy ring protects the 9,11-position from unwanted side reactions during the organometallic step. Following methylation, the epoxy ring is opened using hydrogen fluoride, which simultaneously installs the 9α-fluoro and 11β-hydroxy groups with high stereoselectivity.
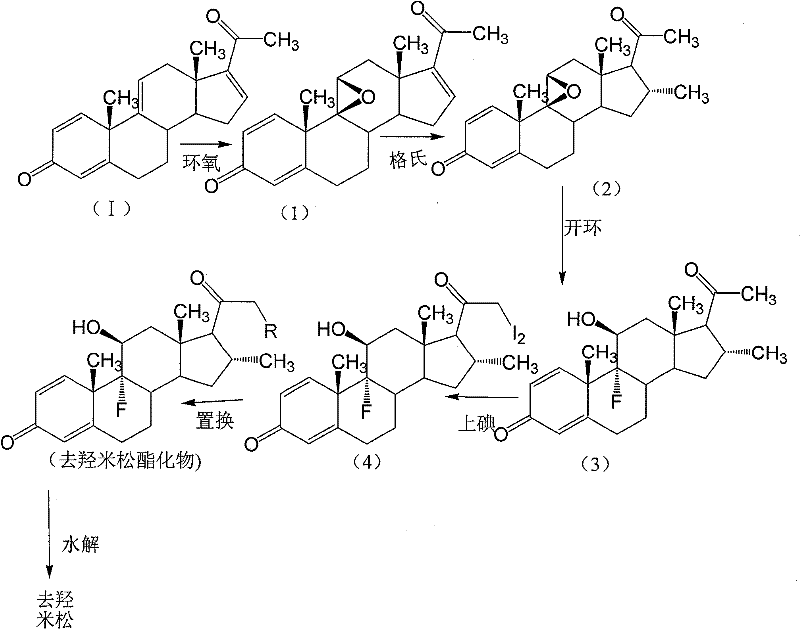
Impurity control is rigorously managed through the specific ordering of the 21-position modifications. The patent highlights that direct Grignard reaction on certain precursors is problematic due to the presence of double bonds at the 9,11-position, which can lead to competing addition reactions. Therefore, the 9,11-transformation must precede or be carefully orchestrated with the 16-methylation. Furthermore, the 21-iodination step is designed to produce primarily di-iodo substitution products, which are then converted to esters. The mechanism ensures that even if minor mono-iodo by-products are formed, they do not negatively impact the subsequent substitution reaction. This tolerance for minor impurities during the iodination phase simplifies the purification requirements before the final hydrolysis step, ensuring that the final desoximetasone product meets stringent purity specifications without requiring excessive chromatographic separation.
How to Synthesize Desoximetasone Efficiently
The synthesis of desoximetasone via this patented route offers a practical framework for laboratory and pilot-scale operations. The process is divided into six distinct unit operations: epoxidation, Grignard reaction, ring-opening, iodination, substitution, and hydrolysis. Each step utilizes common industrial solvents such as methanol, dichloromethane, and DMF, making the transition from bench to plant straightforward. The reaction conditions are moderate, with temperatures generally ranging from -20°C to 80°C depending on the specific transformation, allowing for standard reactor equipment to be used without specialized cryogenic or high-pressure infrastructure. For detailed standard operating procedures regarding reagent stoichiometry and workup protocols, please refer to the technical guide below.
- Perform 9,11-epoxidation on the starting tetraene-dione using N-bromosuccinimide (NBS) and base to form the epoxy intermediate.
- Execute a 16,17-Grignard reaction with methyl magnesium bromide to introduce the 16-alpha-methyl group.
- Conduct ring-opening with hydrogen fluoride to install the 9-alpha-fluoro and 11-beta-hydroxy groups.
- Perform 21-iodination followed by substitution with alkyl carboxylate to form the ester intermediate.
- Finalize the synthesis through alkaline hydrolysis to yield pure desoximetasone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route translates into tangible operational efficiencies and risk mitigation. The primary advantage stems from the utilization of a shared intermediate that is already produced in-house for other corticosteroid lines, such as dexamethasone and betamethasone. This synergy allows for the doubling of production capacity across multiple product lines without necessitating entirely new supply chains for raw materials. The elimination of expensive and hard-to-source starting materials drastically simplifies the procurement landscape, reducing the risk of supply disruptions caused by single-source dependencies on niche chemical vendors. Additionally, the simplified purification processes reduce the consumption of silica gel and chromatography solvents, leading to substantial cost savings in waste management and material usage.
- Cost Reduction in Manufacturing: The streamlined synthetic sequence inherently lowers the cost of goods sold by reducing the number of unit operations and minimizing material loss associated with multi-step purifications. By avoiding the use of expensive auxiliary materials and leveraging common solvents like acetone and THF, the process achieves a leaner cost structure. The ability to perform reactions at moderate temperatures further reduces energy consumption compared to processes requiring extreme thermal conditions. These factors combine to offer a significantly reduced production cost profile, making the final API more competitive in price-sensitive markets.
- Enhanced Supply Chain Reliability: Utilizing a readily available intermediate ensures a stable and continuous supply of the final product. Since the starting material is a common precursor for other major steroids, suppliers can maintain larger inventory buffers and switch production lines flexibly based on market demand. This flexibility mitigates the risk of stockouts and ensures that long-term supply agreements can be honored even during periods of fluctuating demand. The robustness of the chemical steps also means that batch failure rates are minimized, further securing the reliability of the supply chain for downstream pharmaceutical formulators.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, using reagents and solvents that are standard in the fine chemical industry. The avoidance of exotic catalysts and the use of aqueous workups for neutralization simplify the waste treatment process, aiding in environmental compliance. The high operability of each step ensures that the process can be scaled from kilogram to multi-ton quantities with predictable outcomes. This scalability supports the growing global demand for topical corticosteroids while adhering to increasingly strict environmental regulations regarding chemical manufacturing emissions and effluent discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this desoximetasone synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation. They provide clarity on the feasibility of scaling this route and the specific quality advantages it offers over legacy methods. Understanding these details is crucial for technical teams evaluating the integration of this process into their existing manufacturing portfolios.
Q: What are the primary advantages of the new desoximetasone synthesis route compared to historical methods?
A: The new route utilizes readily available intermediates (1,4,9,16-tetraene-pregnadiene-3,20-dione) rather than scarce starting materials, significantly shortening the synthetic sequence and improving overall yield compared to the traditional 6-step Russell method which suffered from low yields around 28%.
Q: How does this process impact production costs and supply chain stability?
A: By eliminating expensive auxiliary materials and utilizing common industrial solvents like THF and acetone, the process drastically simplifies purification. Furthermore, the shared intermediate allows for flexible production scheduling alongside dexamethasone and betamethasone, enhancing supply continuity.
Q: Is the Grignard reaction step compatible with the 9,11-epoxy functionality?
A: Yes, the patent specifies that performing the Grignard methylation prior to the 9,11-ring opening is critical. Introducing the hydroxyl group via ring opening after methylation prevents interference with the Grignard reagent, thereby minimizing by-product formation and ensuring high stereochemical purity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Desoximetasone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in maintaining a competitive edge in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patents like CN101397322B are fully realized in actual manufacturing. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of desoximetasone meets the highest international standards. Our commitment to technological excellence allows us to deliver high-purity intermediates and APIs that support the development of safe and effective dermatological treatments.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this novel method. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your production requirements, ensuring a seamless transition to a more efficient and reliable source of desoximetasone.
