Advanced Biocatalytic Route for High-Purity Benidipine Precursors and Commercial Scalability
The pharmaceutical industry is constantly seeking more efficient and sustainable pathways for the synthesis of critical cardiovascular drug intermediates. Patent CN111778297B introduces a groundbreaking improvement in the synthesis of 1-benzyl-3-piperidinol intermediates, specifically targeting the production of (R)-1-hydroxy-4-bromo-1-butyronitrile, a pivotal precursor for Benidipine. This patent discloses a novel biocatalytic approach that utilizes 4-bromobutanal as a starting material, undergoing a cyanogroup transfer reaction with a racemic hydrocyanic acid donor under the catalysis of carrier-free immobilized R-amygdalonitrilase. Unlike traditional chemical synthesis routes that often suffer from harsh reaction conditions and poor stereoselectivity, this enzymatic method achieves exceptional stereocontrol and yield. The technology represents a significant leap forward in green chemistry, offering a viable alternative to heavy metal catalysis and high-pressure hydrogenation, thereby addressing both environmental concerns and the growing demand for high-purity chiral intermediates in the global supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-benzyl-3-piperidinol has relied heavily on methods that pose significant challenges for modern pharmaceutical manufacturing. One common route involves the hydrogenation of 3-hydroxypyridine under high-pressure conditions using noble metal catalysts such as rhodium, palladium, or platinum. This approach not only demands expensive and specialized high-pressure equipment but also carries substantial operational risks due to the handling of hydrogen gas at elevated pressures. Furthermore, these catalytic hydrogenation processes often lack sufficient stereoselectivity, frequently resulting in racemic mixtures that require costly and inefficient resolution steps to isolate the desired (R)-enantiomer. Another conventional pathway involves the reduction of quaternary ammonium salts of 3-hydroxypyridine, which, while operating at moderate pressures, still fails to provide high stereoselectivity, yielding a mixture of (R) and (S) isomers that complicates downstream purification. Additionally, routes utilizing gamma-azido methyl-gamma-butyrolactone involve hazardous reagents like lithium aluminum hydride and high-pressure ring expansion, further exacerbating safety concerns and limiting the feasibility of continuous production on a commercial scale.
The Novel Approach
In stark contrast to these legacy methods, the improved synthesis method disclosed in the patent leverages the power of biocatalysis to overcome these inherent limitations. By employing 4-bromobutanal as a raw material and utilizing a carrier-free immobilized R-amygdalonitrilase catalyst, the process achieves a highly stereoselective cyanogroup transfer reaction. This enzymatic route operates under remarkably mild conditions, typically at temperatures between 5°C and 15°C, eliminating the need for high-pressure reactors and reducing energy consumption. The use of a specific hydrocyanic acid donor, represented by Formula I, ensures a safe and controlled source of the cyano group, avoiding the direct handling of toxic hydrogen cyanide gas. Experimental data from the patent demonstrates the superiority of this approach, with Example 1 achieving a yield of 87% and an enantiomeric excess (ee) of 94%, significantly outperforming comparative examples that utilized free enzymes or different solvents. This dramatic improvement in both yield and optical purity underscores the potential of this novel approach to streamline the manufacturing of complex chiral intermediates.
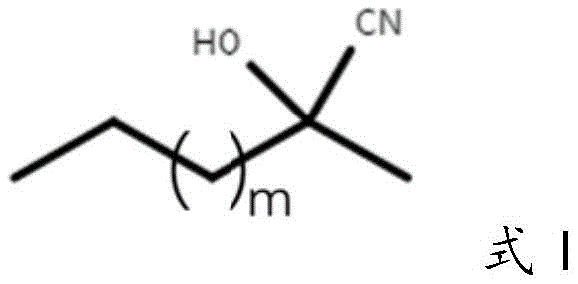
Mechanistic Insights into Carrier-free Immobilized Enzymatic Cyanation
The core innovation of this synthesis lies in the preparation and application of the carrier-free immobilized R-amygdalonitrilase. Unlike traditional immobilization techniques that attach enzymes to solid supports like silica or resin, which can introduce diffusion barriers and reduce catalytic activity, the carrier-free method involves precipitating the enzyme crude product in a tert-butanol aqueous solution and cross-linking it with glutaraldehyde. This creates a highly dense aggregate of enzyme molecules, often referred to as Cross-Linked Enzyme Aggregates (CLEAs), which exhibit superior stability and activity. The absence of an inert carrier means that the volumetric productivity of the biocatalyst is maximized, allowing for higher substrate loading and faster reaction rates. The R-amygdalonitrilase specifically recognizes the prochiral ketone substrate, 4-bromobutanal, and facilitates the addition of the cyanide ion from the donor molecule with high facial selectivity, ensuring the formation of the (R)-configured nitrile intermediate. This precise molecular recognition is crucial for achieving the high ee values observed in the patent examples, as the enzyme's active site effectively discriminates between the two possible enantiomeric transition states.
Furthermore, the reaction mechanism benefits from the biphasic solvent system employed, typically consisting of diisopropyl ether and a citric acid buffer. This system helps to solubilize the organic substrate while maintaining the aqueous environment necessary for enzyme stability. The pH of the buffer, optimized around 4.5, plays a critical role in maintaining the ionization state of the enzyme's active site residues, ensuring optimal catalytic turnover. The use of a hydrocyanic acid donor, such as 2-hydroxy-2-methylhexanitrile (where m=2 in Formula I), allows for the reversible generation of HCN in situ, which is then immediately consumed by the enzyme-catalyzed reaction, driving the equilibrium towards the product. This in situ generation minimizes the accumulation of free cyanide, enhancing safety and preventing enzyme inhibition. The combination of carrier-free immobilization, optimized solvent systems, and safe cyanide donors creates a robust catalytic cycle that is highly resistant to deactivation, allowing for prolonged reaction times and consistent product quality, which is essential for industrial applications.
How to Synthesize (R)-1-hydroxy-4-bromo-1-butyronitrile Efficiently
The synthesis of this high-value chiral intermediate involves a streamlined sequence of biocatalyst preparation followed by the cyanotransfer reaction. The process begins with the preparation of the carrier-free immobilized enzyme, where the crude R-amygdalonitrilase solution is precipitated and cross-linked to form stable biocatalyst particles. These particles are then introduced into a reaction vessel containing the substrate 4-bromobutanal and the hydrocyanic acid donor in a buffered organic-aqueous medium. The reaction proceeds under gentle stirring at controlled low temperatures to ensure maximum stereoselectivity. Following the completion of the reaction, the biocatalyst is easily removed by filtration, and the product is isolated through standard extraction and purification techniques. The detailed standardized synthesis steps, including specific reagent quantities, timing, and purification parameters, are outlined in the guide below.
- Preparation of the biocatalyst by precipitating R-amygdalonitrilase crude solution in tert-butanol and cross-linking with glutaraldehyde to form carrier-free immobilized enzymes.
- Execution of the cyanogroup transfer reaction by mixing 4-bromobutanal with a hydrocyanic acid donor (Formula I) in a biphasic solvent system containing the immobilized enzyme.
- Purification of the resulting chiral nitrile intermediate through filtration, washing, and flash column chromatography to achieve high optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this enzymatic synthesis route offers compelling strategic advantages that extend beyond mere technical performance. The shift from high-pressure hydrogenation to mild enzymatic catalysis fundamentally alters the cost structure and risk profile of manufacturing cardiovascular intermediates. By eliminating the need for expensive noble metal catalysts like palladium or rhodium, and removing the requirement for specialized high-pressure reactors, the capital expenditure (CAPEX) and operational expenditure (OPEX) associated with production facilities can be significantly reduced. Furthermore, the enhanced safety profile of using a solid hydrocyanic acid donor instead of gaseous HCN or high-pressure hydrogen reduces insurance costs and regulatory compliance burdens, contributing to overall cost reduction in pharmaceutical intermediate manufacturing. The robustness of the carrier-free enzyme also implies a longer catalyst lifespan and reduced frequency of catalyst replacement, further driving down material costs over time.
- Cost Reduction in Manufacturing: The elimination of precious metal catalysts represents a direct and substantial saving in raw material costs, as noble metals are subject to volatile market prices and supply constraints. Additionally, the mild reaction conditions (5-15°C) drastically reduce energy consumption compared to high-temperature or high-pressure processes, leading to lower utility costs. The high yield of 87% reported in the patent examples minimizes raw material waste and maximizes the output per batch, effectively lowering the cost of goods sold (COGS). The simplified downstream processing, facilitated by the easy filtration of the carrier-free enzyme, reduces solvent usage and labor hours required for purification, contributing to a leaner and more cost-efficient manufacturing operation.
- Enhanced Supply Chain Reliability: Reliance on noble metals often exposes supply chains to geopolitical risks and mining bottlenecks; switching to a biocatalytic process mitigates this vulnerability by using renewable and readily available enzyme sources. The use of stable, carrier-free immobilized enzymes ensures consistent batch-to-batch performance, reducing the risk of production delays caused by catalyst variability or failure. Moreover, the mild operating conditions allow for the use of standard glass-lined or stainless steel reactors that are widely available in the fine chemical industry, ensuring that production capacity can be easily secured or expanded without long lead times for specialized equipment. This flexibility enhances the resilience of the supply chain against disruptions and allows for more responsive inventory management.
- Scalability and Environmental Compliance: The carrier-free immobilization technique is inherently scalable, as the preparation of Cross-Linked Enzyme Aggregates (CLEAs) can be performed in large batches without the limitations of pore diffusion found in supported catalysts. This facilitates the commercial scale-up of complex pharmaceutical intermediates from pilot plant to multi-ton production with minimal re-optimization. From an environmental perspective, the process generates less hazardous waste, as it avoids heavy metal contamination and reduces the use of toxic solvents. The aqueous-organic biphasic system allows for easier solvent recovery and recycling, aligning with increasingly stringent global environmental regulations and sustainability goals. This 'green' profile not only ensures compliance but also enhances the brand value of the final pharmaceutical product by appealing to eco-conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this enzymatic synthesis technology. These answers are derived directly from the experimental data and technical disclosures within patent CN111778297B, providing a reliable basis for decision-making. Understanding these details is crucial for R&D teams evaluating process feasibility and procurement teams assessing supplier capabilities. The information covers catalyst preparation, reaction safety, and scalability potential.
Q: What are the primary advantages of using carrier-free immobilized enzymes over traditional noble metal catalysts?
A: Carrier-free immobilized enzymes eliminate the diffusion limitations associated with solid supports, offering higher catalytic efficiency and stereoselectivity (up to 94% ee) compared to traditional hydrogenation methods which often require high pressure and yield racemic mixtures.
Q: How does this enzymatic process improve safety in the manufacturing of cardiovascular drug intermediates?
A: The process utilizes a safe hydrocyanic acid donor (Formula I) instead of toxic gaseous hydrogen cyanide, and operates at mild temperatures (5-15°C), significantly reducing operational risks and equipment requirements compared to high-pressure hydrogenation.
Q: Is this synthesis method suitable for large-scale commercial production?
A: Yes, the method demonstrates high yield (87%) and robustness under mild conditions, making it highly scalable. The carrier-free nature of the enzyme simplifies downstream processing and reduces waste, facilitating efficient commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-benzyl-3-piperidinol Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the biocatalytic synthesis route described in CN111778297B for producing high-purity 1-benzyl-3-piperidinol intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial operations. Our state-of-the-art facilities are equipped to handle enzymatic reactions with precision, maintaining stringent purity specifications and rigorous QC labs to guarantee that every batch meets the highest international standards. We understand that the consistency of chiral intermediates is paramount for the efficacy and safety of the final cardiovascular drug, and our commitment to quality assurance is unwavering.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced technology for your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a stable, cost-effective, and sustainable supply of critical intermediates, driving your drug development projects forward with confidence and efficiency.
