Advanced Manufacturing of 1-Benzyl-3-Piperidinol: A Technical Breakthrough for Benidipine Production
Advanced Manufacturing of 1-Benzyl-3-Piperidinol: A Technical Breakthrough for Benidipine Production
The pharmaceutical industry constantly seeks more efficient pathways for synthesizing critical cardiovascular drug intermediates, and Patent CN113264868B presents a significant advancement in this domain. This patent discloses an improved synthesis method for 1-benzyl-3-piperidinol, a pivotal chiral building block required for the production of Benidipine, a second-generation dihydropyridine calcium antagonist. The core innovation lies in a novel enzyme immobilization technique that utilizes polyethyleneimine and oxidized dextran to stabilize R-amygdalonitrile. This approach addresses longstanding issues regarding catalyst stability and activity loss, delivering a process that boasts both high stereoselectivity and substantially improved yields compared to prior art. For R&D directors and procurement specialists, this technology represents a viable route to secure a reliable supply of high-purity pharmaceutical intermediates while optimizing manufacturing economics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1-benzyl-3-piperidinol has faced significant hurdles regarding efficiency and stereochemical control. Traditional routes often rely on the use of 4-bromobutanal as a starting material, subjected to cyano group transfer reactions using hydrocyanic acid under the influence of R-amygdaloxynitrile enzymes. While earlier iterations of this biocatalytic route demonstrated acceptable stereoselectivity with ee values around 90%, they suffered from critically low yields, often hovering near 40%. Furthermore, previous attempts to immobilize the enzyme, such as those disclosed in Chinese patent application CN111778297A, still encountered substantial loss of active sites during the catalyst preparation phase. This degradation of catalytic activity not only inflates the cost of goods due to excessive enzyme loading requirements but also introduces variability in the reaction outcome, complicating quality control and batch-to-batch consistency in large-scale operations.
The Novel Approach
The methodology outlined in Patent CN113264868B fundamentally reengineers the catalyst preparation to overcome these deficiencies. By employing a co-precipitation strategy involving the R-amygdalonitrile crude product and polyethyleneimine, followed by a fixation reaction with oxidized dextran, the inventors have created a highly stable biocatalyst. This specific combination of polymers creates a protective microenvironment around the enzyme, preserving its active conformation and preventing leaching. Experimental data from the patent indicates that this novel catalyst enables the cyano transfer reaction to proceed with remarkable efficiency, achieving yields of up to 91% for the intermediate (R)-1-hydroxy-4-bromo-1-butyronitrile with an enantiomeric excess (ee) of 97%. This dramatic improvement in both yield and purity establishes a robust foundation for the subsequent reduction and cyclization steps, ensuring a streamlined path to the final piperidine derivative.
Mechanistic Insights into Enzyme Immobilization and Cyano Transfer
The success of this synthesis hinges on the precise molecular interactions during the catalyst formation. The process begins with the coprecipitation of the enzyme crude product with polyethyleneimine (PEI) at controlled low temperatures between 0°C and 10°C. PEI, a cationic polymer with a specific molecular weight range of 5000 to 25000 Daltons, interacts electrostatically with the protein surface. 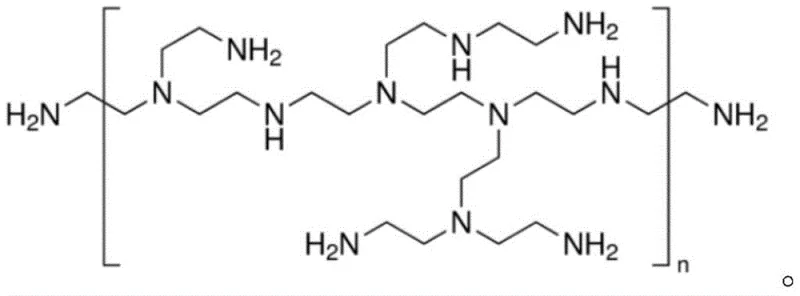 Following this, oxidized dextran is introduced to act as a cross-linking agent. The aldehyde groups generated from the oxidation of dextran by sodium periodate react with amino groups on both the enzyme and the PEI, forming a stable Schiff base network. This covalent and physical entrapment effectively locks the enzyme in place, minimizing conformational changes that lead to deactivation. The resulting catalyst exhibits enhanced resistance to the organic-aqueous biphasic solvent system used in the subsequent reaction, maintaining high activity over extended periods.
Following this, oxidized dextran is introduced to act as a cross-linking agent. The aldehyde groups generated from the oxidation of dextran by sodium periodate react with amino groups on both the enzyme and the PEI, forming a stable Schiff base network. This covalent and physical entrapment effectively locks the enzyme in place, minimizing conformational changes that lead to deactivation. The resulting catalyst exhibits enhanced resistance to the organic-aqueous biphasic solvent system used in the subsequent reaction, maintaining high activity over extended periods.
In the cyano transfer step, the stabilized catalyst facilitates the nucleophilic addition of cyanide to 4-bromobutanal. The reaction utilizes a racemic hydrocyanic acid donor, specifically compounds of Formula I where m is an integer from 0 to 5. 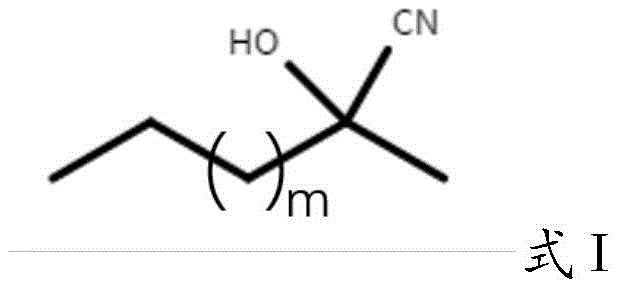 The choice of solvent system is critical; a mixture of diisopropyl ether and citric acid buffer (pH 4-5) creates an optimal interface for the biocatalytic transformation. The mild acidic conditions are essential for maintaining the protonation state of the active site residues while preventing the premature decomposition of the cyanohydrin product. The high stereoselectivity observed (ee > 95%) suggests that the immobilization matrix does not hinder the enzyme's chiral pocket, allowing it to discriminate effectively between enantiomers of the donor and direct the cyanide attack exclusively to the pro-chiral center of the aldehyde substrate.
The choice of solvent system is critical; a mixture of diisopropyl ether and citric acid buffer (pH 4-5) creates an optimal interface for the biocatalytic transformation. The mild acidic conditions are essential for maintaining the protonation state of the active site residues while preventing the premature decomposition of the cyanohydrin product. The high stereoselectivity observed (ee > 95%) suggests that the immobilization matrix does not hinder the enzyme's chiral pocket, allowing it to discriminate effectively between enantiomers of the donor and direct the cyanide attack exclusively to the pro-chiral center of the aldehyde substrate.
How to Synthesize 1-Benzyl-3-Piperidinol Efficiently
Implementing this synthesis requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and maximum yield. The process is divided into three distinct phases: catalyst preparation, biocatalytic cyano transfer, and chemical conversion to the final piperidine ring. Operators must pay close attention to the temperature controls during the precipitation and fixation steps, as deviations can alter the porosity and activity of the immobilized enzyme. The subsequent chemical steps involving borane reduction and cyclization with benzyl halides are standard organic transformations but benefit significantly from the high purity of the incoming nitrile intermediate. For a detailed breakdown of the standardized operating procedures and specific reagent quantities, please refer to the technical guide below.
- Prepare the cyano transfer catalyst by coprecipitating R-amygdalonitrile crude product with polyethyleneimine, followed by fixation with oxidized dextran.
- Perform the cyano transfer reaction using 4-bromobutanal and a hydrocyanic acid donor under mild acidic conditions to obtain the chiral nitrile intermediate.
- Execute reduction with borane-THF followed by cyclization with benzyl halide and purification to yield the final 1-benzyl-3-piperidinol product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this improved synthesis method offers compelling value propositions for procurement managers and supply chain heads focused on cost reduction in pharmaceutical intermediates manufacturing. The primary driver of economic value is the substantial increase in reaction yield. Moving from historical yields of approximately 40% to over 90% for the key intermediate effectively more than doubles the output per unit of raw material input. This efficiency gain directly translates to a lower cost of goods sold (COGS) by reducing the volume of starting materials like 4-bromobutanal and hydrocyanic acid donors required per kilogram of final product. Additionally, the enhanced stability of the immobilized catalyst implies a longer operational lifespan or the potential for catalyst recycling, further driving down the variable costs associated with biocatalyst consumption.
- Cost Reduction in Manufacturing: The elimination of inefficient catalyst systems leads to significant savings in raw material utilization. By maximizing the conversion of 4-bromobutanal into the desired chiral nitrile, the process minimizes the generation of waste byproducts that require expensive disposal or complex separation protocols. The use of relatively inexpensive and commercially available polymers like polyethyleneimine and dextran for immobilization ensures that the catalyst preparation itself remains cost-effective, avoiding the need for proprietary or exotic support matrices that could inflate upfront capital expenditure.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by processes that are sensitive to minor fluctuations in conditions. The robust nature of the PEI-dextran immobilized enzyme provides a buffer against such variabilities, ensuring consistent batch quality. This reliability reduces the risk of production delays caused by out-of-specification batches, thereby securing a steady flow of 1-benzyl-3-piperidinol for downstream API synthesis. Furthermore, the reliance on common solvents like diisopropyl ether and ethyl acetate simplifies logistics, as these materials are readily available from multiple global suppliers, mitigating the risk of single-source bottlenecks.
- Scalability and Environmental Compliance: The process operates under mild conditions, typically between 0°C and 15°C, which reduces the energy load required for heating or extreme cooling compared to high-temperature chemical catalysis. This energy efficiency aligns with modern green chemistry principles and helps facilities meet increasingly stringent environmental regulations. The aqueous-organic biphasic system facilitates easier product separation and reduces the volume of hazardous organic waste. Consequently, scaling this process from pilot plant to commercial production (100 kgs to 100 MT) involves manageable engineering challenges, making it an attractive option for rapid capacity expansion to meet market demand.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into existing production lines. The answers are derived directly from the experimental data and specifications provided in the patent documentation, ensuring accuracy and relevance for decision-makers.
Q: What are the key advantages of the new immobilization method for R-amygdalonitrile?
A: The new method utilizes polyethyleneimine and oxidized dextran to create a robust enzyme matrix. This significantly reduces the loss of active sites compared to traditional unsupported fixed enzymes, resulting in higher stereoselectivity (ee > 95%) and improved reaction yields.
Q: How does this synthesis route impact the cost of Benidipine production?
A: By achieving higher yields in the critical cyano transfer step and minimizing enzyme deactivation, the process reduces raw material waste and downstream purification costs. The mild reaction conditions (0-10°C) also lower energy consumption compared to harsher chemical alternatives.
Q: Is this process scalable for commercial API manufacturing?
A: Yes, the protocol uses standard industrial solvents like diisopropyl ether and avoids exotic reagents. The immobilization technique enhances catalyst stability, allowing for potential reuse or continuous flow applications, which are essential for large-scale commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1-Benzyl-3-Piperidinol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of cardiovascular therapeutics like Benidipine. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical clients. We are committed to delivering 1-benzyl-3-piperidinol with stringent purity specifications, utilizing advanced analytical techniques in our rigorous QC labs to verify enantiomeric excess and chemical purity. Our infrastructure is designed to support the complex requirements of chiral synthesis, providing a secure and compliant source for your supply chain.
We invite you to collaborate with us to leverage these technological advancements for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this optimized route can improve your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us assist you in securing a competitive advantage in the marketplace through superior chemical manufacturing.
