Advanced Solid-Phase Synthesis of Somaglutide for Commercial Scale-Up
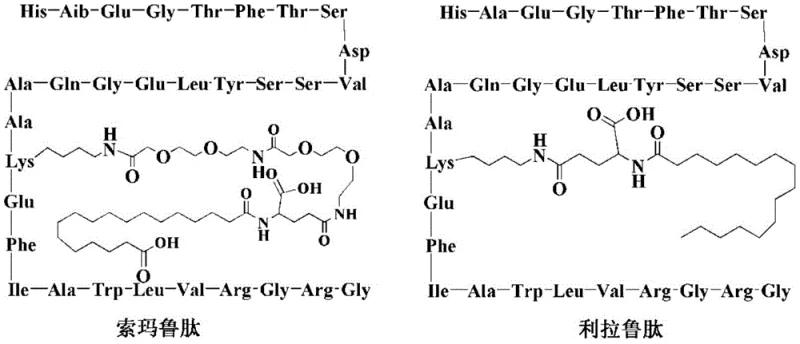
The pharmaceutical landscape for Type 2 diabetes treatment has been revolutionized by GLP-1 analogues, with Somaglutide standing out as a premier therapeutic agent due to its extended half-life and efficacy. The patent CN110294800B discloses a novel preparation method for Somaglutide that addresses critical bottlenecks in polypeptide synthesis, specifically targeting the reduction of difficult-to-remove impurities like [D-His]1-Somaglutide. This technical breakthrough offers a robust pathway for the commercial scale-up of complex pharmaceutical intermediates, ensuring that manufacturers can meet the surging global demand with high-purity products. By optimizing the introduction of the fatty acid side chain and the PEG linker through pre-synthesized building blocks, this method significantly mitigates the steric hindrance issues that plague conventional stepwise synthesis.
For R&D directors and process chemists, the ability to control the stereochemistry at the Histidine-1 position while maintaining high coupling yields is paramount. The disclosed method achieves this through a strategic combination of orthogonal protecting groups and fragment condensation. Unlike earlier methods that attempted to build the entire side chain on the resin atom-by-atom, this approach introduces the complexity in solution phase where reaction kinetics are more favorable. This shift not only improves the overall yield but also simplifies the purification burden downstream, making it an attractive candidate for cost reduction in API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for Somaglutide often rely on the gradual coupling of amino acids directly on the solid support, followed by the on-resin modification of the Lysine side chain at position 26. Historical patents, such as WO2006097537, describe using acid-sensitive protecting groups like Mmt or Mtt for the Lysine side chain. However, removing these groups typically requires dichloromethane solutions containing trifluoroacetic acid, which poses a significant risk of prematurely cleaving the peptide from the resin or removing other acid-sensitive side-chain protections. Furthermore, attempting to couple long PEG chains and fatty acids sequentially on the resin creates immense steric hindrance. As the molecular weight of the resin-bound peptide increases, the diffusion of reagents into the resin matrix becomes limited, leading to incomplete reactions, deletion sequences, and a dramatic drop in crude purity. These inefficiencies translate directly into higher production costs and longer lead times for high-purity pharmaceutical intermediates.
The Novel Approach
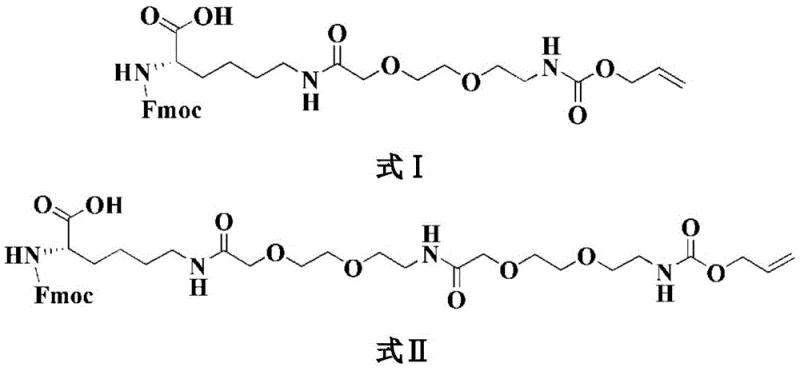
The method described in CN110294800B fundamentally reengineers the synthesis strategy by introducing the complex side chain components as pre-formed building blocks. Instead of struggling with on-resin PEGylation, the inventors utilize Fmoc-Lys(P)-OH derivatives where P represents a pre-attached PEG chain terminated with an Alloc protecting group (either -AEEA-Alloc or -AEEA-AEEA-Alloc). As illustrated in the structural formulas below, these building blocks allow the PEG moiety to be incorporated in a single, high-efficiency coupling step. This strategy bypasses the multiple activation and coupling cycles required in older methods, thereby reducing the exposure of the growing peptide chain to potentially racemizing conditions. The use of these specialized intermediates ensures that the hydrophilic PEG chain is correctly positioned to enhance solubility without compromising the integrity of the peptide backbone.
Mechanistic Insights into Orthogonal Protection and Fragment Coupling
The core mechanistic advantage of this synthesis lies in the orthogonal protection strategy employed for the Lysine-26 side chain. The Alloc (allyloxycarbonyl) group is stable under the standard piperidine/DMF conditions used for Fmoc removal, yet it can be selectively removed using palladium catalysts (such as Pd(PPh3)4) in the presence of a scavenger like phenylsilane. This orthogonality is crucial because it allows the main peptide chain to be fully assembled up to the N-terminus before the fatty acid side chain is attached. By delaying the introduction of the bulky octadecanedioic acid moiety until the end, the method avoids the severe steric crowding that would otherwise inhibit the coupling of amino acids 1 through 20. This sequential logic ensures that each peptide bond is formed under optimal kinetic conditions, maximizing the fidelity of the sequence.
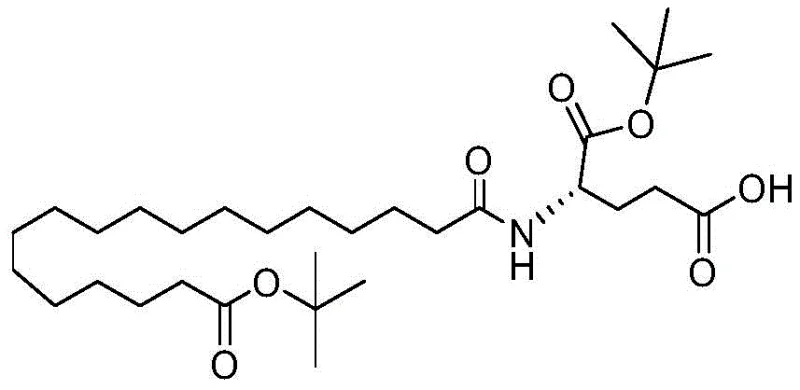
Furthermore, the patent details the preparation and use of a specific dipeptide fragment: octadecanedioic acid mono-tert-butyl ester-Glu-OtBu. Coupling this dipeptide fragment to the deprotected Lysine side chain is significantly more efficient than coupling the free fatty acid followed by Glutamic acid. The pre-activation of the fatty acid as an NHS ester or via carbodiimide chemistry in solution phase ensures a high degree of conversion before it ever touches the resin. This fragment condensation approach effectively turns a difficult, multi-step on-resin transformation into a single, robust coupling event. The result is a drastic reduction in side reactions and a cleaner crude product profile, which is essential for meeting the stringent impurity specifications required for reliable API intermediate supplier status in the global market.
How to Synthesize Somaglutide Efficiently
The synthesis protocol outlined in the patent provides a clear, step-by-step roadmap for producing Somaglutide with superior quality attributes. The process begins with the loading of Fmoc-Gly onto a suitable resin, followed by the iterative addition of protected amino acids using standard activators like DIC and HOBt. The critical deviation from standard protocols occurs at position 26, where the pre-synthesized Fmoc-Lys(AEEA-Alloc)-OH is coupled. Following the completion of the main chain assembly up to Histidine-1, the Alloc group is selectively removed, and the fatty acid-Glu dipeptide fragment is attached. This modular approach decouples the complexity of the side chain from the linearity of the main chain synthesis. For detailed operational parameters, stoichiometry, and specific reaction times, please refer to the standardized synthesis guide below.
- Load Fmoc-Gly onto Wang or 2-Chlorotrityl resin and perform sequential Fmoc-SPPS coupling to build the [21-31] fragment.
- Couple the pre-synthesized Fmoc-Lys(AEEA-Alloc)-OH or Fmoc-Lys(AEEA-AEEA-Alloc)-OH building block to the growing peptide chain.
- Remove the Alloc protecting group orthogonally and couple the octadecanedioic acid-Glu dipeptide fragment to complete the side chain modification.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain and procurement perspective, the adoption of this novel synthesis method offers substantial strategic benefits beyond mere technical elegance. By shifting complex chemical transformations from the solid phase to the solution phase, the process becomes inherently more robust and easier to control at scale. The reliance on pre-synthesized building blocks means that quality control can be performed on these key intermediates before they enter the main production line, reducing the risk of batch failures during the expensive final stages of peptide assembly. This risk mitigation is a critical factor for reducing lead time for high-purity pharmaceutical intermediates, as it minimizes the need for re-processing or discarding batches due to low coupling efficiency.
- Cost Reduction in Manufacturing: The elimination of multiple on-resin coupling steps for the side chain significantly reduces the consumption of expensive reagents, solvents, and activated amino acids. In traditional methods, the low efficiency of on-resin PEGylation often requires large excesses of reagents to drive the reaction to completion, generating substantial chemical waste. By using the dipeptide fragment and pre-modified lysine, the stoichiometry is optimized, and the number of washing and deprotection cycles is reduced. This streamlining of the process flow translates directly into lower variable costs per gram of product, allowing for more competitive pricing structures without sacrificing margin.
- Enhanced Supply Chain Reliability: The use of commercially available or easily preparable starting materials, such as Fmoc-Gly-Wang resin and standard Fmoc-amino acids, ensures a stable supply base. The method does not rely on exotic catalysts or hard-to-source reagents that could create supply bottlenecks. Furthermore, the improved crude purity achieved through this method reduces the burden on the purification team. When the crude product contains fewer deletion sequences and side-products, the preparative HPLC purification runs faster and yields more final product per cycle. This efficiency gain enhances the overall throughput of the manufacturing facility, ensuring consistent delivery schedules for downstream clients.
- Scalability and Environmental Compliance: Scaling peptide synthesis is notoriously difficult due to heat transfer and mixing limitations in large reactors. However, this method's emphasis on fragment condensation simplifies the scale-up physics. The solution-phase synthesis of the building blocks can be easily scaled in standard chemical reactors, while the solid-phase steps remain within manageable limits. Additionally, the reduction in solvent usage and chemical waste associated with fewer coupling cycles aligns with modern green chemistry principles. This environmental efficiency not only lowers waste disposal costs but also ensures compliance with increasingly strict environmental regulations governing pharmaceutical manufacturing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Somaglutide synthesis method. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a transparent view of the technology's capabilities. Understanding these nuances is vital for stakeholders evaluating the feasibility of adopting this route for their own production needs.
Q: How does this method reduce [D-His]1 impurities in Somaglutide?
A: The method utilizes specific protecting group strategies (such as Boc or Trt on Histidine) and optimized coupling conditions that minimize racemization risks during the final amino acid addition, significantly lowering the formation of the [D-His]1 epimer compared to traditional stepwise methods.
Q: What are the advantages of using Fmoc-Lys(AEEA-Alloc)-OH building blocks?
A: Using pre-synthesized PEG-modified lysine derivatives eliminates the need for difficult on-resin PEGylation steps. This approach reduces steric hindrance during the coupling of subsequent amino acids and simplifies the overall process flow, leading to higher crude purity.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the protocol is designed for scalability by using robust condensing agents like DIC/HOBt and standard Fmoc-SPPS techniques. The use of dipeptide fragments for the fatty acid side chain further enhances coupling efficiency, making it viable for multi-kilogram manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Somaglutide Supplier
The technical innovations detailed in patent CN110294800B represent a significant leap forward in the manufacturability of GLP-1 analogues. At NINGBO INNO PHARMCHEM, we recognize the value of such process optimizations and have integrated similar advanced strategies into our CDMO operations. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial manufacturing is seamless. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Somaglutide meets the highest global standards.
We invite pharmaceutical partners to collaborate with us to leverage these cost-effective synthesis routes for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and reliable manufacturing partner available in the market.
