Advanced Synthesis of Liraglutide Using Depsipeptide Units for Commercial Scale-up
Advanced Synthesis of Liraglutide Using Depsipeptide Units for Commercial Scale-up
The pharmaceutical industry continuously seeks robust methodologies for the production of complex polypeptide therapeutics, particularly for high-demand molecules like Liraglutide, a long-acting GLP-1 analog used in diabetes management. Patent CN108676086B discloses a groundbreaking synthesis method that addresses the persistent challenges of solid-phase peptide synthesis (SPPS) for long sequences. By strategically introducing Depsipeptide Units into the peptide backbone during the assembly phase, this technology effectively mitigates the aggregation phenomena that traditionally plague the synthesis of hydrophobic sequences. This innovation not only enhances the chemical integrity of the growing chain but also streamlines the downstream processing requirements, offering a compelling value proposition for manufacturers aiming to optimize their supply chains for high-purity pharmaceutical intermediates.
For R&D directors and process chemists, the significance of this patent lies in its ability to bypass the steric hindrance and solubility issues associated with conventional linear elongation. The method utilizes a sophisticated combination of standard Fmoc-protected amino acids and specialized protected amino acid fragments containing ester linkages. These fragments are incorporated at critical junctions within the Liraglutide sequence, such as the Gly4-Thr5 or Val10-Ser11 sites, where intermolecular hydrogen bonding typically causes the peptide resin to collapse and resist further coupling. The subsequent conversion of these ester bonds back to amide bonds via a controlled transamidation reaction ensures the final product possesses the exact native structure required for biological activity, thereby establishing a reliable liraglutide intermediate supplier pathway that balances chemical elegance with industrial practicality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional strategies for synthesizing long peptides like Liraglutide often rely on either stepwise solid-phase synthesis or fragment condensation, both of which present significant drawbacks in a commercial setting. In stepwise SPPS, as the peptide chain extends, the accumulation of hydrophobic protecting groups and the peptide backbone itself intensifies intermolecular association effects, leading to the formation of rigid beta-sheet structures on the resin. This aggregation physically shields active amino groups, resulting in incomplete couplings, the generation of deletion sequences, and a dramatic drop in crude purity that necessitates expensive and yield-loss-inducing purification steps. Furthermore, fragment condensation, while attempting to solve length issues, introduces substantial steric hindrance during the ligation of large segments, often requiring excessive reagents and prolonged reaction times that drive up manufacturing costs and complicate waste management protocols.
The Novel Approach
The novel approach detailed in the patent circumvents these physical barriers by integrating Depsipeptide Units directly into the synthesis workflow. Instead of forcing difficult amide bond formations at aggregation-prone sites, the method employs pre-formed fragments where an ester bond temporarily replaces the amide linkage. This structural modification disrupts the hydrogen bonding network responsible for resin aggregation, maintaining the peptide chain in a more soluble and accessible conformation throughout the elongation process. 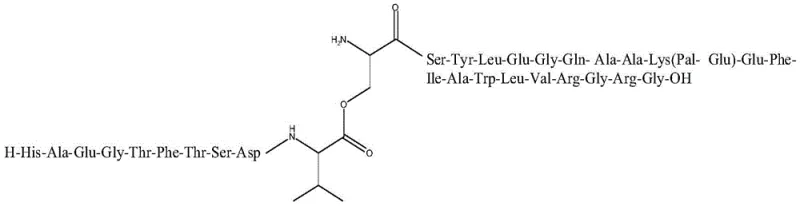 As illustrated in the structural representation, the insertion of these units allows for high-efficiency coupling even in difficult sequences. Following the assembly and cleavage of the full-protection peptide resin, a mild alkaline treatment triggers an O-to-N acyl shift, seamlessly converting the ester bonds back into the native amide bonds, thus delivering a crude product with exceptional purity and yield suitable for immediate downstream processing.
As illustrated in the structural representation, the insertion of these units allows for high-efficiency coupling even in difficult sequences. Following the assembly and cleavage of the full-protection peptide resin, a mild alkaline treatment triggers an O-to-N acyl shift, seamlessly converting the ester bonds back into the native amide bonds, thus delivering a crude product with exceptional purity and yield suitable for immediate downstream processing.
Mechanistic Insights into Depsipeptide-Mediated Aggregation Control
The core chemical mechanism driving the success of this synthesis is the strategic use of the depsipeptide motif to modulate the physicochemical properties of the intermediate resin-bound species. In standard peptide synthesis, the planar nature of the amide bond facilitates strong inter-chain hydrogen bonding, which is the root cause of the 'difficult sequences' often encountered in GLP-1 analogs. By substituting the amide nitrogen with an oxygen atom at specific positions, the Depsipeptide Unit removes the hydrogen bond donor at that locus, effectively breaking the continuity of the beta-sheet structure. 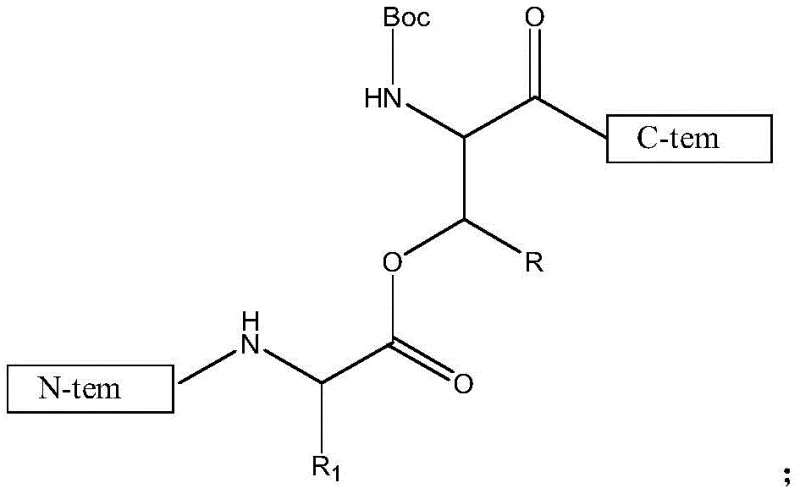 This structural disruption ensures that the growing peptide chain remains solvated and flexible, allowing coupling reagents like HBTU or PyBop to access the terminal amine groups with minimal steric resistance. The result is a drastic reduction in the formation of truncated impurities and racemization byproducts, which are common failure modes in the production of complex polypeptide drugs.
This structural disruption ensures that the growing peptide chain remains solvated and flexible, allowing coupling reagents like HBTU or PyBop to access the terminal amine groups with minimal steric resistance. The result is a drastic reduction in the formation of truncated impurities and racemization byproducts, which are common failure modes in the production of complex polypeptide drugs.
Furthermore, the final transamidation step is a kinetically controlled intramolecular rearrangement that offers high specificity. Under alkaline conditions (pH 7.5-9.5), the free amine group adjacent to the ester linkage acts as a nucleophile, attacking the carbonyl carbon of the ester to form a tetrahedral intermediate. This intermediate subsequently collapses to release the alcohol moiety and reform the thermodynamically more stable amide bond. This mechanism is highly advantageous because it occurs spontaneously in solution after cleavage, meaning the difficult chemistry is performed on a smaller, more manageable fragment level or avoided entirely during the critical resin-bound phase. For quality control teams, this translates to a cleaner impurity profile, as the reaction is driven by the inherent proximity of the functional groups, minimizing the risk of intermolecular side reactions that could lead to dimerization or oligomerization.
How to Synthesize Liraglutide Efficiently
The implementation of this synthesis route requires precise control over solid-phase reaction conditions and the preparation of specific building blocks. The process begins with the loading of Fmoc-Gly onto Wang resin, followed by the iterative addition of amino acids. Crucially, at predetermined positions such as Val10-Ser11 or Ser11-Ser12, the standard amino acid is replaced with the specialized Depsipeptide Unit fragment. The detailed standardized synthesis steps, including specific reagent ratios, activation times, and washing protocols necessary to achieve the reported 86%+ yields, are outlined in the technical guide below.
- Prepare Fmoc-Gly-Wang resin and perform solid-phase coupling using protected amino acids and pre-synthesized Depsipeptide Units at specific aggregation-prone sites.
- Cleave the fully protected peptide resin using a TFA-based cocktail (TFA: thioanisole:anisole:EDT:H2O) to obtain the crude peptide precursor containing ester linkages.
- Dissolve the crude precursor in acetonitrile/water, adjust pH to 7.5-9.5 with an alkaline reagent, and allow ester bond-transamidation to occur for 5-7 hours to yield crude Liraglutide.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this depsipeptide-based methodology represents a significant opportunity for cost reduction in peptide manufacturing. The primary economic driver is the substantial improvement in crude peptide purity and overall yield. By mitigating the formation of deletion sequences and difficult-to-remove impurities during the synthesis phase, the burden on downstream purification is drastically reduced. This means less consumption of expensive preparative HPLC columns and solvents, and a higher recovery rate of the final active pharmaceutical ingredient, directly translating to a lower cost of goods sold (COGS) per gram of Liraglutide produced.
- Cost Reduction in Manufacturing: The elimination of complex fragment isolation and purification steps, which are typical in traditional fragment condensation strategies, simplifies the production workflow significantly. Because the Depsipeptide Units allow for efficient linear assembly on the resin, the process avoids the low-yield ligation steps that often bottleneck large-scale peptide production. This streamlined approach reduces the consumption of high-cost coupling reagents and protected amino acids, as fewer excess equivalents are needed to drive difficult reactions to completion, thereby optimizing raw material utilization and minimizing waste disposal costs associated with failed batches.
- Enhanced Supply Chain Reliability: The robustness of this synthesis method enhances supply chain continuity by reducing the risk of batch failures due to 'difficult sequences.' In conventional processes, unexpected aggregation can halt production or require lengthy re-optimization, leading to unpredictable lead times. The depsipeptide strategy provides a more predictable and reproducible reaction profile, ensuring that production schedules can be met consistently. This reliability is critical for securing long-term contracts with pharmaceutical partners who require guaranteed volumes of high-purity GLP-1 analogs to meet global market demand without interruption.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, the method offers distinct advantages by reducing the volume of organic solvents and hazardous reagents required per kilogram of product. The higher efficiency means fewer synthesis cycles are needed to produce the same amount of API, lowering the overall solvent footprint. Additionally, the use of standard Fmoc chemistry combined with a simple aqueous workup for the transamidation step aligns well with green chemistry principles, facilitating easier regulatory approval and compliance with increasingly stringent environmental regulations governing pharmaceutical manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the depsipeptide synthesis strategy for Liraglutide. These insights are derived directly from the patent specifications and are intended to clarify the operational benefits and chemical feasibility of this advanced manufacturing route for potential partners and technical stakeholders evaluating the technology.
Q: How does the Depsipeptide Unit strategy improve Liraglutide synthesis?
A: By temporarily replacing difficult amide bonds with ester bonds at specific sites (e.g., Val10-Ser11), the method disrupts the secondary structure aggregation that typically hinders long peptide synthesis, significantly improving coupling efficiency and crude purity.
Q: What is the role of the ester bond-transamidation reaction?
A: This final step converts the temporary ester linkages back into the native amide peptide bonds under mild alkaline conditions (pH 7.5-9.5), ensuring the final product matches the natural Liraglutide sequence without requiring complex enzymatic ligation.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the patent demonstrates high total synthesis yields (over 86%) and crude purity nearing 80%, which drastically reduces the burden on downstream purification and makes the process economically viable for commercial scale-up compared to traditional fragment condensation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Liraglutide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the depsipeptide synthesis method in meeting the global demand for high-quality anti-diabetic therapeutics. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a GMP-compliant manufacturing environment. Our facility is equipped with state-of-the-art peptide synthesizers and rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Liraglutide intermediate meets the highest international standards for safety and efficacy.
We invite pharmaceutical companies and biotech firms to collaborate with us to leverage this innovative synthesis technology for their pipeline projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to discuss specific COA data and route feasibility assessments, ensuring that your supply chain is optimized for both performance and profitability in the competitive landscape of peptide drug manufacturing.
