Advanced Solid-Liquid Phase Synthesis for High-Purity Polypeptide Drugs with Disulfide Bonds
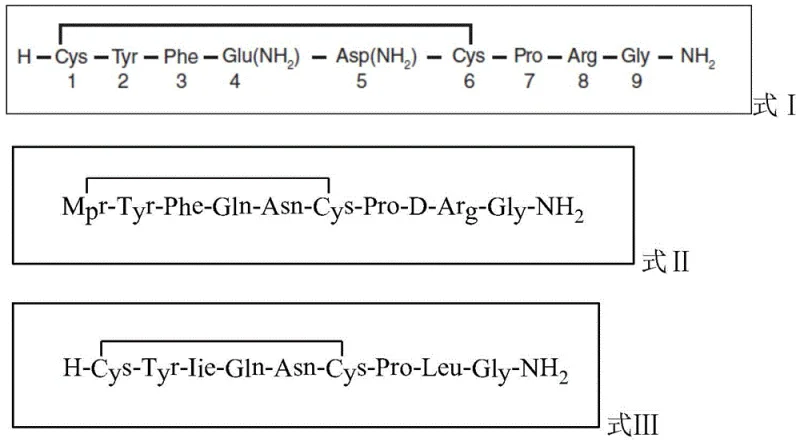
The pharmaceutical industry is constantly seeking more efficient and environmentally friendly pathways for the synthesis of complex polypeptide therapeutics, particularly those stabilized by disulfide bonds. A significant breakthrough in this domain is detailed in patent CN112574285A, which discloses a novel solid-liquid phase synthesis method specifically designed for polypeptide drugs containing a pair of disulfide bonds. This technology represents a paradigm shift from traditional multi-step protocols, offering a streamlined approach that integrates resin cleavage, side-chain deprotection, and cyclization into a unified operation. By leveraging specific weak acid-sensitive protecting groups and a unique oxidation strategy, this method addresses long-standing challenges related to purity, yield, and operational complexity. For R&D directors and process chemists, understanding this innovation is critical, as it promises to enhance the manufacturability of vital hormones such as oxytocin, desmopressin, and arginine vasopressin while simultaneously reducing the environmental footprint associated with harsh acidic reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of disulfide-bonded polypeptides has been plagued by inefficiencies inherent in both liquid-phase fragment condensation and standard solid-phase oxidation strategies. Traditional liquid-phase methods often require the synthesis of separate peptide fragments followed by condensation, a process notorious for its lengthy construction periods, complex purification requirements, and the frequent necessity of using strong acids like hydrobromic acid. Furthermore, conventional solid-phase synthesis typically involves a two-stage process where the peptide is first cleaved from the resin using high concentrations of trifluoroacetic acid (TFA), often around 95%, to yield a linear peptide. This linear intermediate must then undergo a separate oxidation step in an extremely dilute solution to prevent intermolecular dimerization. This requirement for extreme dilution necessitates vast volumes of solvent, creating a massive burden on downstream processing, waste treatment, and overall cost efficiency. Additionally, solid-phase oxidation performed directly on the resin often demands excessive amounts of oxidizing agents, such as 10 to 15 equivalents of iodine, which can lead to undesirable side reactions, including the over-oxidation of sensitive residues like Tyrosine, thereby compromising the purity of the crude peptide.
The Novel Approach
In stark contrast to these cumbersome legacy techniques, the methodology described in patent CN112574285A introduces a sophisticated solid-liquid phase hybrid strategy that fundamentally simplifies the workflow. The core innovation lies in the utilization of Sieber Resin coupled with amino acids protected by weak acid-sensitive groups, such as the Mmt (4-methoxytrityl) group for Cysteine residues. This specific chemical architecture allows for the simultaneous execution of resin cleavage, side-chain deprotection, and disulfide bond formation under mild acidic conditions. Instead of relying on 95% TFA, the process employs a mixture of trifluoroethanol (TFE) and dichloromethane (DCM), or a very low concentration of TFA (1-5%) in DCM. Crucially, the oxidation step is integrated directly into the cleavage phase by adding a dilute iodine solution (0.5-1.5% in DCM) towards the end of the reaction. This "one-pot" capability eliminates the need for a secondary preparation or a separate alkaline oxidation system, drastically reducing the number of unit operations. The result is a process that not only shortens the overall construction period but also significantly improves the separation of impurities from the main peak, facilitating much easier purification and yielding a crude peptide of substantially higher quality.
Mechanistic Insights into Simultaneous Cleavage and Oxidation
The chemical elegance of this synthesis route is rooted in the precise selection of protecting group strategies that enable orthogonal reactivity under mild conditions. The use of Sieber Resin is pivotal, as it provides an acid-labile linkage that can be cleaved under much weaker acidic conditions than the standard Wang or Rink amide resins which typically demand strong TFA cocktails. When combined with Fmoc-amino acids bearing weak acid-sensitive side-chain protections—specifically Fmoc-Cys(Mmt)-OH for the cysteine residues involved in the disulfide bridge—the system creates a synchronized deprotection environment. As the mild acid mixture (TFE/DCM) attacks the resin linkage to release the peptide, it concurrently removes the Mmt groups from the sulfhydryl moieties. This immediate exposure of the free thiol groups in the solution phase, while still in the presence of the cleavage cocktail, sets the stage for rapid intramolecular cyclization. The subsequent addition of a controlled amount of iodine acts as a gentle oxidant that converts the free thiols into the desired disulfide bond without the aggressive conditions that typically degrade sensitive peptide sequences. This mechanistic synergy ensures that the linear peptide does not persist long enough to form intermolecular dimers, a common pitfall in traditional liquid-phase oxidation.
Furthermore, the impurity profile of the resulting crude peptide is markedly superior due to the homogeneity of the reaction phase and the minimized exposure to harsh reagents. In conventional solid-phase oxidation, the diffusion of large oxidant molecules into the resin matrix can be uneven, leading to incomplete cyclization or localized over-oxidation. By transitioning to a liquid-phase oxidation immediately upon cleavage, the reactants are uniformly distributed, ensuring consistent conversion rates across the entire batch. The patent data highlights that this method effectively avoids the side reactions associated with Tyrosine residues, which are prone to iodination under standard solid-phase oxidation conditions using excess iodine. The strategic timing of the iodine addition—introduced only after the initial cleavage period—allows the deprotection to proceed fully before the oxidative stress is applied. This temporal control, combined with the mild acidity of the TFE/DCM system, preserves the integrity of other acid-sensitive functionalities within the peptide sequence, such as the amide side chains of Asparagine and Glutamine, ultimately delivering a product with a cleaner HPLC profile and higher biological activity potential.
How to Synthesize Arginine Vasopressin Efficiently
The practical implementation of this advanced synthesis route offers a clear roadmap for manufacturing high-value polypeptide intermediates with enhanced efficiency. The process begins with the preparation of the amino acid resin using Sieber Resin as the solid support, followed by the sequential coupling of Fmoc-protected amino acids from the C-terminus to the N-terminus. Critical to the success of this route is the incorporation of Cysteine residues protected with the Mmt group, which enables the subsequent mild acid cleavage. Once the full peptide sequence is assembled on the resin, the transformation occurs in a single reaction vessel where the resin is treated with the TFE/DCM cleavage cocktail. After a defined period allowing for resin cleavage and side-chain deprotection, a dilute iodine solution is introduced to drive the cyclization. The detailed standardized synthesis steps, including specific molar ratios, reaction times, and workup procedures required to replicate this high-yield process, are outlined in the guide below.
- Prepare amino acid resin using Sieber Resin and Fmoc-amino acids with weak acid-sensitive protecting groups (e.g., Mmt for Cysteine).
- Perform resin cleavage using a mild acid system (TFE/DCM or low % TFA) followed by the addition of dilute iodine solution to induce simultaneous disulfide bond formation.
- Concentrate the reaction mixture and precipitate the crude peptide using ether to obtain high-purity product ready for purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this solid-liquid phase synthesis technology translates into tangible strategic benefits that extend far beyond simple laboratory curiosity. The primary value driver is the drastic simplification of the manufacturing workflow, which directly correlates to reduced operational expenditures and improved throughput. By eliminating the need for a separate oxidation step and avoiding the use of massive solvent volumes required for dilute liquid-phase oxidation, the process inherently lowers the consumption of raw materials and utilities. This reduction in material intensity not only decreases the direct cost of goods sold but also alleviates the logistical burden associated with the storage, handling, and disposal of hazardous chemicals. Furthermore, the ability to achieve high crude purity means that downstream purification processes, such as preparative HPLC, become more efficient, requiring fewer cycles and less stationary phase, which further drives down the overall production cost.
- Cost Reduction in Manufacturing: The economic impact of this technology is profound, primarily driven by the elimination of expensive and hazardous reagents. Traditional methods rely heavily on high-concentration TFA, a costly and corrosive acid that requires specialized equipment and extensive neutralization protocols. By shifting to a mild acid system based on TFE and DCM, the process significantly reduces the expenditure on acid reagents and the associated waste treatment costs. Moreover, the consolidation of cleavage and oxidation into a single step removes an entire unit operation from the production schedule, saving labor hours and equipment occupancy time. The avoidance of secondary preparation steps means that the yield loss typically incurred during multiple isolation and purification stages is minimized, leading to a higher overall recovery of the final active pharmaceutical ingredient. This atom-economic approach ensures that every gram of starting material contributes more effectively to the final product, optimizing the return on investment for raw material procurement.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the robustness of this synthesis method offers greater predictability and stability in production schedules. Conventional liquid-phase oxidation often suffers from scalability issues due to the requirement for extremely dilute conditions, which necessitates disproportionately large reactors for larger batches, creating bottlenecks in facility utilization. The new method operates at much higher concentrations, allowing manufacturers to maximize the capacity of existing reactor infrastructure without the need for capital-intensive expansion. This efficiency gain translates directly into shorter lead times for order fulfillment, enabling suppliers to respond more agilely to market demands. Additionally, the reduced reliance on specialized, hard-to-source reagents and the simplification of the process flow mitigate the risks associated with supply disruptions, ensuring a more continuous and reliable flow of critical polypeptide intermediates to downstream drug manufacturers.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental sustainability intensify, this synthesis route positions manufacturers favorably by significantly reducing the generation of hazardous waste. The avoidance of 95% TFA cleavage cocktails dramatically lowers the volume of acidic waste streams that require neutralization and safe disposal, aligning production practices with green chemistry principles. The simplified workup procedure, which involves straightforward concentration and ether precipitation, minimizes the use of auxiliary solvents and reduces the energy consumption associated with solvent recovery systems. This environmental advantage is not merely a compliance checkbox but a competitive differentiator that appeals to global pharmaceutical partners who are increasingly prioritizing sustainable supply chains. The process is inherently designed for scale-up, maintaining its efficiency and purity profiles from pilot scales to commercial tonnage, thereby ensuring that the benefits realized in the lab are fully transferable to industrial manufacturing environments.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this solid-liquid phase synthesis technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for evaluating the feasibility of adopting this method for your specific polypeptide projects. Understanding these nuances is essential for making informed decisions about process development and vendor selection.
Q: How does this method avoid the use of high-concentration TFA?
A: The method utilizes Sieber Resin and weak acid-sensitive protecting groups (like Mmt), allowing cleavage and deprotection to occur under mild acidic conditions (e.g., TFE/DCM or 1-5% TFA) rather than the harsh 95% TFA typically required.
Q: What are the advantages regarding impurity control in disulfide bond formation?
A: By performing oxidation simultaneously with cleavage in a homogeneous phase using controlled amounts of iodine, the process minimizes side reactions such as over-oxidation of Tyrosine residues and prevents intermolecular dimerization, resulting in higher crude purity.
Q: Is this synthesis method suitable for large-scale manufacturing?
A: Yes, the process eliminates the need for extremely dilute conditions required in traditional liquid-phase oxidation, significantly reducing solvent consumption and simplifying post-treatment, which makes it highly scalable for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Arginine Vasopressin Supplier
The technological advancements detailed in patent CN112574285A underscore the immense potential for optimizing the production of complex polypeptide therapeutics. At NINGBO INNO PHARMCHEM, we recognize the critical importance of translating such innovative synthetic routes into robust, commercial-scale realities. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of mild acid cleavage and simultaneous oxidation are fully realized in a GMP-compliant environment. Our commitment to quality is unwavering, supported by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify the identity and potency of every batch. We understand that the transition from lab-scale innovation to industrial manufacturing requires not just chemical expertise but also deep engineering knowledge to manage heat transfer, mixing, and safety protocols effectively.
We invite you to collaborate with us to leverage these cutting-edge synthesis strategies for your next project. Our technical team is prepared to conduct a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating exactly how this streamlined process can reduce your overall manufacturing expenses. We encourage you to reach out to our technical procurement team to request specific COA data and route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access to a supply chain that is not only cost-effective and reliable but also at the forefront of polypeptide synthesis technology, ensuring your products reach the market faster and with the highest standards of quality.
