Revolutionizing Disulfide-Bonded Polypeptide Manufacturing with Advanced Solid-Liquid Phase Synthesis
The pharmaceutical industry continuously seeks robust methodologies for the synthesis of complex polypeptide therapeutics, particularly those stabilized by intramolecular disulfide bonds. Patent CN112574285B introduces a groundbreaking solid-liquid phase synthesis method that addresses critical bottlenecks in the production of hormones such as Arginine Vasopressin, Oxytocin, and Desmopressin. Unlike conventional approaches that separate resin cleavage from oxidation, this innovation integrates both steps into a unified process using weak acid-sensitive resins and controlled iodine oxidation. This strategic shift not only enhances the chemical integrity of sensitive amino acid residues but also streamlines the downstream processing required for high-purity active pharmaceutical ingredients (APIs). 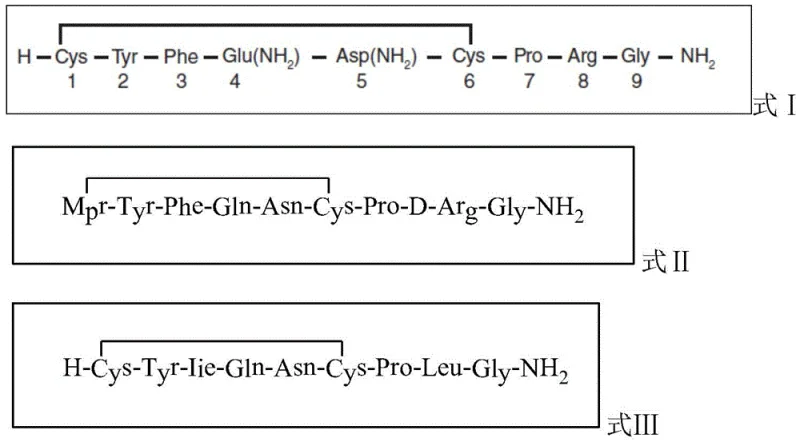
For global procurement and supply chain leaders, the implications of this technology extend far beyond the laboratory bench. By eliminating the need for harsh cleavage conditions and separate oxidation vessels, the method offers a pathway to significantly reduced operational expenditures and improved safety profiles. As a reliable polypeptide supplier, understanding these mechanistic advantages is essential for securing a competitive edge in the market for high-value hormonal intermediates. The following analysis details how this patented route transforms the manufacturing landscape for disulfide-bonded peptides.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis strategies for disulfide-containing polypeptides are generally bifurcated into liquid-phase oxidation and solid-phase oxidation, both of which suffer from significant inefficiencies at scale. In liquid-phase oxidation, the linear peptide must first be cleaved from the resin using high concentrations of trifluoroacetic acid (typically 95% TFA), isolated, and then dissolved in extremely dilute solutions (often as low as 0.5 mg/ml) to prevent intermolecular dimerization during oxidation. This requirement for extreme dilution necessitates massive solvent volumes, leading to prohibitive energy costs for solvent recovery and creating a substantial burden on waste treatment facilities. Furthermore, the multi-step nature of isolating the linear intermediate before oxidation increases the risk of yield loss and extends the overall production lead time considerably.
Conversely, conventional solid-phase oxidation attempts to form the disulfide bond while the peptide is still attached to the resin, typically using large excesses of iodine. While this avoids the dilution issue, it introduces severe chemical selectivity problems. The high local concentration of oxidant on the solid support often leads to the over-oxidation of susceptible side chains, particularly Tyrosine and Methionine residues, resulting in complex impurity profiles that are difficult to purify. Additionally, the reliance on strong acid systems for the final cleavage step remains a constant safety and environmental hazard, generating large quantities of corrosive waste that complicate regulatory compliance and increase the total cost of ownership for the manufacturing process.
The Novel Approach
The patented solid-liquid phase synthesis method fundamentally reengineers this workflow by utilizing Sieber Resin, which is sensitive to weak acids, thereby enabling cleavage under much milder conditions than standard Rink Amide resins. The core innovation lies in the simultaneous execution of resin cleavage and disulfide bond formation within a single reaction vessel. By employing a solvent system such as TFE/DCM (1:4 ratio) or low-percentage TFA in DCM, the peptide is released from the resin into a homogeneous solution where a controlled amount of dilute iodine (0.5-1.5% in DCM) is introduced. This ensures that the oxidation occurs in a liquid phase environment that favors intramolecular cyclization without the need for the extreme dilution required in traditional liquid-phase methods.
This hybrid approach effectively mitigates the drawbacks of both predecessor technologies. It avoids the excessive iodine usage and side reactions associated with solid-phase oxidation while eliminating the massive solvent volumes and isolation steps of liquid-phase oxidation. The result is a streamlined process where deprotection, cleavage, and cyclization converge, yielding a crude peptide with superior purity and a simplified impurity profile. For manufacturers, this translates to a drastic reduction in unit operations, lower solvent consumption, and a more robust process capable of delivering consistent quality for critical hormonal therapies like Arginine Vasopressin and Oxytocin.
Mechanistic Insights into Weak Acid-Mediated Cleavage and Oxidation
The success of this synthesis route hinges on the precise selection of protecting groups and the kinetics of the cleavage-oxidation cascade. The method employs Fmoc-solid phase synthesis strategies where the Cysteine residues, crucial for disulfide formation, are protected with weak acid-labile groups such as Mmt (4-methoxytrityl). Unlike the robust Trt (trityl) or Pbf groups used in conventional synthesis which require 95% TFA for removal, the Mmt group can be cleaved under the mild acidic conditions provided by the TFE/DCM mixture or low-concentration TFA. This orthogonality allows the side-chain protecting groups to be removed concurrently with the cleavage of the peptide from the Sieber Resin linker, ensuring that the free thiol groups are generated in situ exactly when the oxidant is introduced.
From an impurity control perspective, the homogeneous nature of the reaction medium during the oxidation phase is critical. In solid-phase oxidation, diffusion limitations within the resin matrix can lead to uneven oxidant distribution, causing localized over-oxidation. In this novel method, the peptide is solvated, allowing for uniform exposure to the dilute iodine solution. The controlled addition of iodine during the late stage of cleavage ensures that the concentration of free thiols and oxidant is balanced to favor the formation of the monomeric cyclic disulfide over polymeric dimers or higher-order aggregates. This mechanistic precision results in a crude product where the main peak is well-separated from impurities in HPLC analysis, significantly reducing the load on preparative chromatography during the final purification stage.
How to Synthesize Arginine Vasopressin Efficiently
The synthesis of Arginine Vasopressin using this patented methodology offers a clear roadmap for process chemists aiming to optimize yield and purity. The procedure begins with the loading of Fmoc-Gly onto Sieber Resin, followed by the sequential coupling of amino acids using standard Fmoc chemistry, with specific attention paid to using Fmoc-Cys(Mmt)-OH to enable the subsequent weak-acid cleavage. Once the full sequence is assembled, the resin is treated with the cleavage cocktail containing the oxidant, merging two critical steps into one. This consolidation not only saves time but also minimizes the handling of reactive intermediates. For detailed operational parameters, stoichiometry, and safety protocols, please refer to the standardized synthesis guide below.
- Prepare the peptide resin using Sieber Resin and Fmoc-amino acids with weak acid-sensitive protecting groups (e.g., Mmt for Cysteine).
- Perform resin cleavage using a weak acid system (TFE/DCM or low % TFA) while simultaneously adding a dilute iodine solution to form the disulfide bond.
- Concentrate the reaction mixture and precipitate the crude peptide with diethyl ether to obtain high-purity product ready for final purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this solid-liquid phase synthesis technology represents a strategic opportunity to optimize the cost structure and reliability of polypeptide sourcing. The elimination of separate oxidation reactors and the reduction in solvent volumes directly correlate to lower utility costs and reduced dependency on volatile solvent markets. Furthermore, the simplified workflow reduces the number of quality control checkpoints required between steps, accelerating the release of batches for final formulation. This efficiency gain is particularly valuable in the context of cost reduction in peptide manufacturing, where margin pressures are increasingly driven by complex purification requirements.
- Cost Reduction in Manufacturing: The primary driver for cost savings in this process is the consolidation of unit operations. By combining cleavage and oxidation, the method eliminates the need for a dedicated oxidation vessel, the associated stirring and monitoring equipment, and the labor hours required to transfer and isolate the linear peptide intermediate. Additionally, the avoidance of high-concentration TFA reduces the consumption of this expensive reagent and lowers the costs associated with neutralizing and disposing of hazardous acidic waste. The use of weaker acids also permits the potential use of less corrosion-resistant equipment materials, further lowering capital expenditure requirements for production facilities.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by complex processes that have multiple points of failure. This streamlined synthesis reduces the number of critical process steps, thereby statistically lowering the risk of batch failures due to operational errors or equipment malfunctions. The robustness of the crude peptide quality means that purification columns have a longer lifespan and require less frequent regeneration or replacement, ensuring a smoother flow of finished goods. This reliability is essential for maintaining the inventory levels required to meet the demands of global pharmaceutical clients without unexpected disruptions.
- Scalability and Environmental Compliance: Scaling peptide synthesis is notoriously difficult due to heat transfer and mixing issues in large oxidation tanks. This method mitigates those challenges by removing the need for large-volume dilute oxidation steps, making the transition from pilot scale to commercial tonnage significantly more predictable. From an environmental standpoint, the reduction in solvent usage and the elimination of heavy acidic waste streams align with increasingly stringent global environmental regulations. This proactive compliance reduces the risk of regulatory shutdowns and positions the supply chain as a sustainable partner for eco-conscious pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this solid-liquid phase synthesis technology. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering a transparent view of the method's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this solid-liquid phase method improve crude peptide purity compared to traditional solid-phase oxidation?
A: Traditional solid-phase oxidation often requires excessive iodine equivalents on the resin, leading to side reactions like the over-oxidation of Tyrosine residues. The patented method utilizes a homogeneous liquid-phase oxidation environment during cleavage with controlled, dilute iodine concentrations, significantly reducing side products and improving the separation of impurities from the main peak.
Q: What are the environmental benefits of avoiding high-concentration TFA in this synthesis route?
A: Conventional methods rely on 95% trifluoroacetic acid (TFA) for cleavage, generating hazardous acidic waste that is costly and difficult to treat. This novel approach employs weak acid conditions (such as TFE/DCM mixtures or 1-5% TFA), drastically reducing the load of corrosive waste and simplifying the post-reaction workup and environmental compliance procedures.
Q: Why is the simultaneous cleavage and oxidation strategy considered more scalable for commercial production?
A: By merging the cleavage and cyclization steps into a single operational unit, the process eliminates the need for isolating linear peptides and setting up separate, highly dilute oxidation reactors. This reduction in unit operations shortens the overall production cycle, minimizes solvent consumption, and reduces the risk of material loss during transfer, making it highly suitable for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Arginine Vasopressin Supplier
The technological advancements detailed in patent CN112574285B underscore the complexity and sophistication required to produce high-quality disulfide-bonded polypeptides. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is backed by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify the identity and potency of every batch, guaranteeing that our clients receive materials that meet the highest international pharmacopeial standards.
We invite pharmaceutical partners to leverage our technical expertise to optimize their supply chains for critical hormonal intermediates. By collaborating with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that drive efficiency and value in your peptide drug development programs.
