Advanced Synthesis of Prucalopride Intermediates: A Safe and Scalable Ethylene Carbonate Route
Introduction to Patent CN114591278A
The pharmaceutical industry continuously seeks robust synthetic pathways for complex heterocyclic intermediates, particularly for gastrointestinal prokinetic agents like Prucalopride. Patent CN114591278A introduces a groundbreaking methodology for preparing methyl 4-acetamido-5-chloro-2,3-dihydrobenzofuran-7-carboxylate, a critical building block in the synthesis of Prucalopride Succinate. This novel approach leverages ethylene carbonate as a key reagent to construct the benzofuran ring system through a streamlined two-step sequence, effectively bypassing the hazardous and costly limitations of legacy technologies. By shifting away from cryogenic organolithium chemistry and toxic heavy metal oxidants, this invention offers a safer, more environmentally compliant, and economically viable solution for large-scale manufacturing. The strategic implementation of intramolecular alkylation ensures high regioselectivity and minimizes impurity profiles, addressing key pain points for R&D teams focused on process robustness. As a reliable pharmaceutical intermediates supplier, understanding such technological shifts is vital for maintaining competitive advantage in the global supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of the 2,3-dihydrobenzofuran moiety has relied on chemically aggressive and operationally complex strategies that pose significant challenges for industrial scale-up. Traditional routes, such as those described in US5374637, depend heavily on butyllithium-mediated ortho-lithiation at cryogenic temperatures of -78°C, requiring specialized equipment and posing severe safety risks due to the pyrophoric nature of the reagents. Furthermore, alternative pathways utilizing osmium tetroxide for oxidative cleavage or zinc powder for reductive cyclization introduce substantial environmental burdens through heavy metal waste generation and difficult purification protocols. These legacy methods often suffer from low overall yields, sometimes below 30%, and require tedious column chromatography to remove isomeric impurities, driving up production costs and extending lead times. The reliance on hazardous reagents like hydrazine hydrate or Raney Nickel in other variations further complicates regulatory compliance and worker safety, making these processes increasingly untenable for modern green chemistry standards.
The Novel Approach
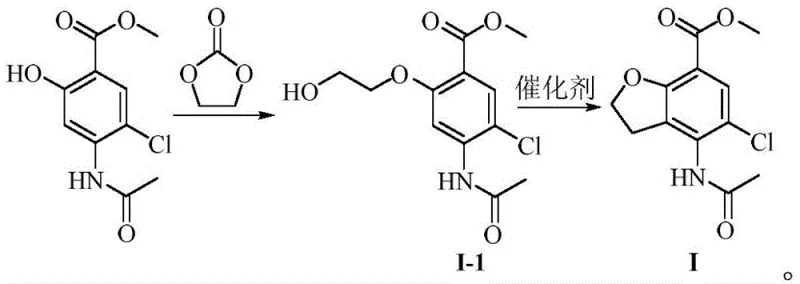
In stark contrast, the methodology disclosed in CN114591278A utilizes ethylene carbonate to introduce the hydroxyethyl side chain under mild basic conditions, followed by a direct intramolecular cyclization to close the furan ring. This strategy effectively eliminates the need for cryogenic operations and toxic oxidants, replacing them with a thermally controlled process that operates between 90°C and 120°C for the etherification step. The subsequent cyclization is catalyzed by accessible Lewis acids such as boron trifluoride etherate, proceeding efficiently at near-ambient temperatures to deliver the target scaffold with exceptional purity. By avoiding the formation of disubstituted byproducts common in halogen-coupling strategies, this route simplifies downstream processing and significantly enhances material throughput. The transition to this cleaner chemistry represents a paradigm shift in cost reduction in API manufacturing, allowing producers to achieve higher yields without the burden of extensive waste treatment or expensive reagent procurement.
Mechanistic Insights into Lewis Acid-Catalyzed Cyclization
The core innovation of this synthesis lies in the mechanistic elegance of the intramolecular alkylation step, where the phenolic oxygen attacks the activated hydroxyethyl group to form the five-membered furan ring. In the presence of a Lewis acid catalyst like BF3·Et2O, the hydroxyl group of the intermediate is activated, facilitating the departure of water or a coordinated species and promoting nucleophilic attack by the adjacent phenoxide. This cyclization is highly regioselective due to the pre-organization of the side chain, which minimizes the entropic penalty typically associated with ring-closing reactions. The choice of catalyst is critical; while strong protic acids can induce cyclization, Lewis acids offer superior control over reaction kinetics, preventing unwanted side reactions such as ester hydrolysis or amide cleavage that could compromise product integrity. Detailed kinetic studies suggest that maintaining the reaction temperature between 15°C and 30°C optimizes the balance between reaction rate and selectivity, ensuring that the delicate acetamido protecting group remains intact throughout the transformation.
Impurity control is another pivotal aspect of this mechanism, as the mild conditions prevent the formation of polymeric byproducts or over-alkylated species often seen in harsher Friedel-Crafts type reactions. The use of ethylene carbonate in the preceding step ensures a clean introduction of the two-carbon bridge without generating halogenated waste, which simplifies the impurity profile of the crude intermediate. Analytical data from the patent indicates that the final product consistently achieves purity levels exceeding 99% after simple recrystallization, demonstrating the high fidelity of the cyclization process. For R&D directors, this level of control translates to reduced analytical burden and faster technology transfer, as the process is less sensitive to minor fluctuations in reagent quality or mixing efficiency. The robustness of this catalytic cycle underscores its suitability for continuous manufacturing environments where consistent product quality is paramount.
How to Synthesize Methyl 4-acetamido-5-chloro-2,3-dihydrobenzofuran-7-carboxylate Efficiently
The practical execution of this synthesis involves a straightforward two-step protocol that begins with the etherification of methyl 4-acetamido-5-chlorosalicylate using ethylene carbonate in a polar aprotic solvent such as DMF. Following the isolation of the hydroxyethoxy intermediate, the material is subjected to cyclization conditions using a stoichiometric amount of Lewis acid in anhydrous THF, followed by a standard aqueous workup and recrystallization. This streamlined workflow minimizes unit operations and solvent exchanges, thereby reducing the overall processing time and resource consumption. The detailed standardized synthesis steps, including specific molar ratios, temperature ramping profiles, and quenching procedures, are outlined in the guide below to ensure reproducibility and safety during scale-up operations.
- React methyl 4-acetamido-5-chlorosalicylate with ethylene carbonate in an organic solvent (e.g., DMF) using a base like potassium carbonate at 90-120°C to form the hydroxyethoxy intermediate.
- Treat the intermediate with a Lewis acid catalyst (e.g., BF3·Et2O) in anhydrous THF at mild temperatures (15-30°C) to induce intramolecular cyclization.
- Perform workup by quenching in ice water, extracting with dichloromethane, and recrystallizing from petroleum ether/ethyl acetate to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ethylene carbonate-based route offers transformative benefits that extend far beyond simple yield improvements. By eliminating the dependency on scarce and volatile reagents like butyllithium or osmium tetroxide, manufacturers can secure a more stable supply of raw materials, insulating production schedules from market fluctuations and geopolitical supply disruptions. The removal of heavy metals from the process stream drastically reduces the cost and complexity of wastewater treatment and solid waste disposal, aligning production facilities with increasingly stringent environmental regulations without requiring capital-intensive infrastructure upgrades. Furthermore, the high purity of the crude product minimizes the need for expensive chromatographic purification steps, leading to substantial cost savings in solvent usage and labor hours. These efficiencies collectively enhance the overall economic viability of the intermediate, allowing for more competitive pricing strategies in the final API market.
- Cost Reduction in Manufacturing: The substitution of expensive and hazardous reagents with commodity chemicals like ethylene carbonate and potassium carbonate directly lowers the bill of materials, while the high yield reduces the effective cost per kilogram of the active intermediate. The avoidance of cryogenic cooling systems also results in significant energy savings, as the process operates at moderate temperatures compatible with standard jacketed reactors. Additionally, the simplified workup procedure reduces solvent consumption and waste generation, further driving down operational expenditures associated with utility and disposal fees. These cumulative factors create a leaner manufacturing process that maximizes margin potential for both the intermediate supplier and the downstream API producer.
- Enhanced Supply Chain Reliability: Sourcing ethylene carbonate and common Lewis acids is far more reliable than procuring specialized organometallic reagents that often have long lead times and strict storage requirements. The robustness of the reaction conditions means that production can be maintained across multiple geographic locations without the need for highly specialized technical expertise or exotic equipment. This flexibility allows supply chain managers to diversify their vendor base and mitigate risks associated with single-source dependencies or regional logistical bottlenecks. Consequently, the continuity of supply for this critical intermediate is significantly strengthened, ensuring that downstream drug production schedules remain uninterrupted even during periods of global market volatility.
- Scalability and Environmental Compliance: The intrinsic safety of the process, characterized by the absence of pyrophoric materials and toxic heavy metals, facilitates easier regulatory approval and smoother scale-up from pilot plant to commercial tonnage. Facilities can operate with lower insurance premiums and reduced safety overheads, as the risk profile of the chemistry is markedly lower than that of traditional lithiation or hydrogenation routes. The generation of carbon dioxide as the primary byproduct simplifies effluent management, ensuring compliance with green chemistry principles and reducing the environmental footprint of the manufacturing site. This alignment with sustainability goals not only meets current regulatory standards but also future-proofs the supply chain against evolving environmental legislation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic route, drawing directly from the experimental data and comparative analysis provided in the patent documentation. These insights are designed to clarify the operational advantages and feasibility of adopting this technology for large-scale production of prucalopride intermediates. Understanding these nuances is essential for stakeholders evaluating the transition from legacy processes to this improved methodology.
Q: What are the primary safety advantages of the ethylene carbonate route over traditional butyllithium methods?
A: The ethylene carbonate route eliminates the need for cryogenic conditions (-78°C) and pyrophoric reagents like butyllithium, significantly reducing operational hazards and fire risks associated with traditional benzofuran ring construction.
Q: How does this method address environmental concerns related to heavy metal oxidants?
A: Unlike prior art methods utilizing toxic osmium tetroxide or large quantities of zinc powder, this process uses ethylene carbonate which generates carbon dioxide as a benign byproduct, drastically lowering heavy metal waste and pollution control costs.
Q: What purity levels can be achieved with the described Lewis acid cyclization?
A: The optimized protocol using catalysts like BF3·Et2O allows for the production of the target intermediate with purity exceeding 99%, minimizing the need for extensive chromatographic purification and ensuring suitability for downstream API synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methyl 4-acetamido-5-chloro-2,3-dihydrobenzofuran-7-carboxylate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to ensure the consistent supply of high-quality pharmaceutical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of innovations like the ethylene carbonate route are fully realized at an industrial level. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of methyl 4-acetamido-5-chloro-2,3-dihydrobenzofuran-7-carboxylate meets the exacting standards required for API synthesis. Our commitment to process excellence means we can reliably deliver this complex intermediate with the consistency and reliability your supply chain demands.
We invite you to engage with our technical procurement team to discuss how this optimized route can benefit your specific project requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this greener, more efficient process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your production needs, ensuring a seamless partnership driven by technical expertise and commercial value.
