Advanced Synthesis of Chiral Zinc-Nitrogen Coordination Compounds for Industrial Catalysis
The landscape of asymmetric catalysis is undergoing a significant transformation, driven by the urgent need for sustainable and cost-effective chiral auxiliaries in the pharmaceutical and fine chemical sectors. Patent CN102690279A introduces a breakthrough in this domain by disclosing a novel chiral zinc-nitrogen coordination compound, specifically the [6-methyl-3-(4-isobutyl-4,5-dihydro)-2-oxazolinyl-pyridine-2-phenol] Zn complex. This innovation addresses the longstanding challenges associated with the high cost and toxicity of traditional precious metal catalysts, offering a robust alternative rooted in earth-abundant zinc chemistry. The patent details a streamlined synthetic pathway that leverages readily available starting materials, such as L-leucinol and 6-methyl-2-cyano-3-hydroxypyridine, to construct a sophisticated chiral environment capable of inducing high stereoselectivity in downstream organic transformations. For R&D directors and procurement strategists alike, this technology represents a pivotal shift towards greener, more economically viable manufacturing processes that do not compromise on the stringent purity requirements demanded by modern drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of high-performance chiral catalysts has been dominated by complexes utilizing expensive transition metals such as rhodium, ruthenium, or iridium, which impose severe economic burdens on large-scale industrial operations. These conventional methods often necessitate intricate ligand designs that require multi-step organic syntheses involving hazardous reagents and cryogenic conditions, leading to poor atom economy and significant waste generation. Furthermore, the removal of trace heavy metal residues from the final active pharmaceutical ingredients (APIs) is a regulatory nightmare, requiring additional purification steps that drastically increase production lead times and operational costs. The reliance on scarce precious metals also introduces substantial supply chain volatility, making long-term production planning difficult for manufacturers who require consistent access to catalytic materials. Consequently, there is a critical industry demand for alternative catalytic systems that can deliver comparable enantioselectivity without the associated financial and environmental liabilities of noble metal chemistry.
The Novel Approach
The methodology outlined in CN102690279A presents a paradigm shift by utilizing zinc chloride as the central metal source, coordinated with a chiral ligand derived from the natural amino alcohol L-leucinol. This approach simplifies the synthetic architecture significantly, replacing complex organometallic procedures with a straightforward reflux reaction in chlorobenzene. The use of L-leucinol not only provides a ready-made chiral pool but also ensures that the resulting complex possesses a well-defined stereochemical configuration essential for asymmetric induction. By operating under relatively mild thermal conditions compared to high-pressure hydrogenation systems, this novel route enhances process safety and reduces energy consumption. The structural integrity of the resulting complex, as evidenced by single-crystal diffraction data, confirms a stable coordination geometry that is crucial for maintaining catalytic activity over extended reaction cycles, thereby offering a reliable solution for continuous manufacturing environments.
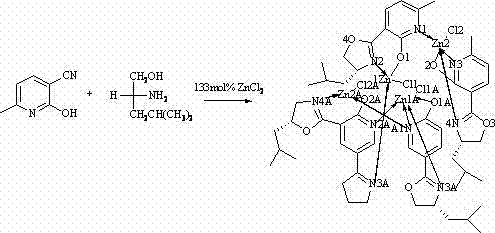
Mechanistic Insights into Zn-N Coordination and Chiral Induction
The core of this technology lies in the precise formation of the zinc-nitrogen coordination bond, which creates a rigid chiral pocket around the metal center. In this complex, the zinc ion acts as a Lewis acid, coordinating with the nitrogen atoms of the oxazoline ring and the phenolic oxygen, locking the ligand into a specific conformation that dictates the approach of substrates during catalysis. The steric bulk provided by the isobutyl group of the leucinol moiety plays a critical role in shielding one face of the reactive center, thereby enforcing high facial selectivity during nucleophilic attacks. This mechanistic feature is particularly advantageous for reactions such as the cyanosilylation of aldehydes, where the spatial arrangement of the ligand directly correlates with the enantiomeric excess of the product. Understanding this coordination geometry allows chemists to fine-tune reaction parameters, such as solvent polarity and temperature, to maximize the turnover number and selectivity of the catalyst in various synthetic applications.
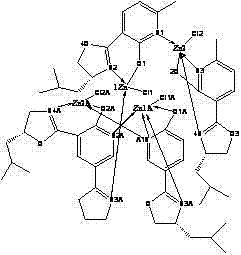
Furthermore, the stability of the Zn-N bond under the described reaction conditions ensures that the catalyst does not decompose prematurely, which is a common failure mode in less robust metal-organic frameworks. The patent data indicates that the complex maintains its structural integrity even after prolonged exposure to reaction media, suggesting a high degree of kinetic stability. This durability is essential for minimizing the formation of metal-containing impurities that could contaminate the final product stream. From a quality control perspective, the predictable nature of this coordination chemistry simplifies the impurity profiling process, allowing analytical teams to focus on organic byproducts rather than unpredictable metal degradation species. The ability to crystallize the complex into high-quality single crystals further facilitates rigorous structural characterization, providing a solid foundation for computational modeling and future ligand optimization efforts.
How to Synthesize [6-methyl-3-(4-isobutyl-4,5-dihydro)-2-oxazolinyl-pyridine-2-phenol] Zn Complex Efficiently
The synthesis protocol described in the patent offers a reproducible pathway for generating this high-value chiral catalyst, emphasizing the importance of strict moisture and oxygen exclusion to prevent hydrolysis of the zinc precursor. The process begins with the combination of anhydrous zinc chloride and the organic precursors in a chlorobenzene solvent system, followed by a prolonged reflux period to ensure complete complexation. Subsequent workup involves careful solvent exchange and extraction techniques to isolate the crude product, which is then subjected to column chromatography for final purification. This standardized approach minimizes batch-to-batch variability, a key concern for process chemists aiming to scale up production.
- Reflux 6-methyl-2-cyano-3-hydroxypyridine and L-leucinol in chlorobenzene with 133 mol% anhydrous ZnCl2 for 48 hours under water and oxygen-free conditions.
- Remove chlorobenzene solvent, dissolve residue in water, and extract the aqueous phase using chloroform to isolate the organic layer.
- Purify the extraction phase via column chromatography using petroleum ether and dichloromethane to obtain the final white crystal complex.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this zinc-based catalytic technology offers profound strategic advantages that extend beyond simple raw material costs. The substitution of precious metals with zinc eliminates the exposure to volatile commodity markets associated with rhodium and palladium, stabilizing the cost of goods sold (COGS) over long-term contracts. Additionally, the use of L-leucinol, a derivative of a naturally occurring amino acid, ensures a secure and sustainable supply of the chiral source material, mitigating risks related to geopolitical instability or resource scarcity. The simplified purification workflow, which avoids the need for specialized heavy metal scavengers, further reduces the operational expenditure associated with waste management and regulatory compliance. These factors collectively contribute to a more resilient and cost-efficient supply chain, enabling manufacturers to offer competitive pricing to their downstream clients while maintaining healthy profit margins.
- Cost Reduction in Manufacturing: The elimination of expensive noble metals like ruthenium or rhodium from the catalyst formulation results in a drastic reduction in direct material costs. Since zinc salts are commodity chemicals available in bulk quantities at a fraction of the price of precious metals, the overall catalyst loading cost is significantly lowered. Moreover, the simplified downstream processing removes the need for costly metal removal resins or additional filtration steps, streamlining the entire production line and reducing labor and utility expenses associated with extended purification cycles.
- Enhanced Supply Chain Reliability: Sourcing L-leucinol and basic zinc salts is far more reliable than securing specialized organometallic precursors, which often have limited suppliers and long lead times. This abundance of raw materials ensures that production schedules can be maintained without interruption, even during periods of global supply chain disruption. The robustness of the synthesis method also means that the catalyst can be produced in larger batches and stored for extended periods without significant degradation, providing a buffer against sudden spikes in demand.
- Scalability and Environmental Compliance: The use of chlorobenzene and standard reflux conditions makes this process highly scalable using existing reactor infrastructure, avoiding the need for capital-intensive equipment upgrades. From an environmental standpoint, the lower toxicity profile of zinc compared to heavy transition metals simplifies effluent treatment and reduces the regulatory burden associated with hazardous waste disposal. This alignment with green chemistry principles not only lowers compliance costs but also enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral zinc complex technology in industrial settings. These insights are derived directly from the experimental data and structural analysis provided in the patent documentation, offering clarity on performance metrics and operational requirements.
Q: What are the critical reaction conditions for synthesizing this chiral zinc complex?
A: The synthesis requires strictly anhydrous and oxygen-free conditions. The reaction involves refluxing the precursors in chlorobenzene for 48 hours with a significant excess of anhydrous ZnCl2 (133 mol%) to drive the coordination equilibrium.
Q: Why is this zinc-based catalyst preferred over traditional transition metal complexes?
A: Zinc is significantly more abundant and less toxic than precious metals like Ruthenium or Rhodium. This complex offers a cost-effective alternative for asymmetric catalysis while maintaining high enantioselectivity potential due to the rigid chiral environment provided by the L-leucinol moiety.
Q: How is the purity of the final complex ensured during purification?
A: The protocol utilizes a rigorous purification sequence involving solvent removal, liquid-liquid extraction with chloroform, and final separation via column chromatography using a petroleum ether/dichloromethane gradient, ensuring high chemical purity suitable for catalytic applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Zinc Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the chiral zinc-nitrogen coordination compounds described in CN102690279A for advancing asymmetric synthesis in the pharmaceutical industry. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to full-scale manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of catalyst or intermediate we produce adheres to the highest international standards. We understand that consistency is key in catalytic applications, and our dedicated process engineering team works tirelessly to optimize reaction parameters for maximum yield and enantioselectivity.
We invite you to collaborate with us to leverage this innovative technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes, demonstrating exactly how switching to this zinc-based system can improve your bottom line. Please contact us today to request specific COA data and comprehensive route feasibility assessments, and let us help you build a more sustainable and profitable supply chain for your chiral intermediates.
