Advanced Chiral Zinc Complex Catalyst for High-Purity Pharmaceutical Intermediate Manufacturing
Introduction to Next-Generation Chiral Catalysis
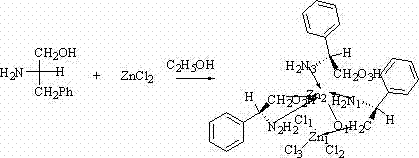
The landscape of asymmetric synthesis is continuously evolving, driven by the relentless demand for high-purity chiral intermediates in the pharmaceutical and agrochemical sectors. A pivotal advancement in this domain is documented in patent CN103342710A, which discloses a novel preparation method for a chiral zinc complex, specifically identified as tris[(S)-phenylalaninol] zinc chloride. This compound represents a significant leap forward in ligand design, utilizing abundant amino alcohol derivatives to create a robust catalytic system capable of inducing chirality in carbon-carbon bond-forming reactions. Unlike traditional noble metal catalysts that often suffer from toxicity issues and prohibitive costs, this zinc-based system offers a greener, more economically viable alternative for generating optically active compounds. The utility of this complex is particularly evident in nucleophilic addition reactions, such as the cyanosilylation of aldehydes, which are fundamental steps in constructing complex molecular architectures required for modern drug discovery.

Furthermore, the structural integrity of this catalyst, confirmed through rigorous X-ray single crystal diffraction analysis, ensures consistent performance across multiple batches, a critical factor for industrial reliability. By leveraging the inherent chirality of L-phenylalaninol, a readily available chiral pool material, the synthesis avoids the need for resolution steps, thereby streamlining the supply chain for downstream manufacturers. This report delves deep into the technical nuances of this innovation, providing R&D directors and procurement specialists with a comprehensive understanding of its potential to revolutionize current manufacturing protocols for chiral intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of chiral catalysts for asymmetric induction has been plagued by several inherent inefficiencies that hinder widespread industrial adoption. Traditional methods often rely on sophisticated, multi-step ligand syntheses that require stringent anhydrous conditions, cryogenic temperatures, and the use of expensive transition metals such as palladium, rhodium, or ruthenium. These precious metal catalysts not only inflate the raw material costs significantly but also introduce severe regulatory hurdles regarding residual metal limits in final pharmaceutical products, necessitating costly purification stages. Moreover, many conventional chiral ligands are derived from rare natural sources or require complex asymmetric synthesis themselves, leading to supply chain vulnerabilities and inconsistent availability. The operational complexity of these legacy systems often results in lower overall yields and extended production cycles, creating bottlenecks that compromise the agility of chemical manufacturing operations.
The Novel Approach
In stark contrast, the methodology outlined in patent CN103342710A presents a paradigm shift towards simplicity and economic efficiency through a direct, one-pot coordination strategy. The novel approach utilizes a straightforward reaction between (L)-phenylalaninol and anhydrous zinc chloride in a precise 3:1 molar ratio, eliminating the need for complex ligand pre-functionalization. This process is conducted in absolute ethyl alcohol, a benign and inexpensive solvent, under atmospheric pressure and moderate reflux conditions, drastically reducing energy consumption and safety risks associated with high-pressure reactors. The resulting complex precipitates as faint yellow crystals upon slow volatilization, indicating a high degree of thermodynamic stability and ease of isolation without the need for chromatographic purification. This streamlined synthetic route not only minimizes waste generation but also ensures that the final catalyst is free from the contaminants typically associated with multi-step organic syntheses.
By adopting this novel approach, manufacturers can transition from fragile, high-maintenance catalytic systems to a robust, shelf-stable solid that retains its stereochemical fidelity over time. The simplicity of the workup procedure, involving merely hot filtration and solvent exchange, allows for seamless integration into existing pilot plant infrastructure, facilitating rapid technology transfer from laboratory bench to commercial scale production lines.
Mechanistic Insights into Zn-Amino Alcohol Coordination
Understanding the precise three-dimensional arrangement of atoms within the chiral zinc complex is paramount for predicting its catalytic behavior and optimizing reaction parameters for specific substrates. The crystallographic data reveals a dinuclear zinc structure where the metal centers are bridged by oxygen atoms from the phenylalaninol ligands, creating a rigid chiral pocket essential for enantioselective discrimination. Specifically, the bond length analysis indicates a Zn(1)-O(1) distance of 1.928(4) Å and a Zn(2)-N(3) distance of 2.052(5) Å, suggesting a strong coordination environment that stabilizes the transition state during nucleophilic attack. The spatial orientation of the phenyl rings from the phenylalaninol moieties creates significant steric bulk around the active zinc sites, which is hypothesized to block one face of the approaching aldehyde substrate, thereby directing the incoming cyanide group to the opposite face with high selectivity.
![X-ray crystal structure of the chiral tris[(S)-phenylalaninol] zinc chloride complex](/insights/img/chiral-zinc-complex-catalyst-pharma-supplier-20260306161534-01.png)
Furthermore, the presence of chloride ions in the coordination sphere, with bond lengths ranging from 2.2258(16) Å to 2.3348(13) Å, plays a crucial role in modulating the Lewis acidity of the zinc centers, fine-tuning the electrophilicity of the coordinated carbonyl group. This delicate balance between steric hindrance and electronic activation is what grants the complex its superior catalytic performance compared to simple zinc salts. For R&D teams, this structural insight provides a blueprint for further ligand modification; by altering the substituents on the phenyl ring of the phenylalaninol, it is possible to systematically tune the steric environment to accommodate bulkier substrates or enhance reaction rates. The robustness of this coordination geometry, evidenced by the sharp melting point range of 174-176 °C, suggests that the catalyst maintains its integrity even under the thermal stress of prolonged reaction times, ensuring consistent product quality throughout the manufacturing campaign.
How to Synthesize Tris[(S)-phenylalaninol] Zinc Chloride Efficiently
The practical implementation of this catalytic technology begins with a reproducible and scalable synthesis protocol that adheres strictly to the stoichiometric and thermal parameters defined in the patent literature. To achieve the highest purity and crystallinity required for X-ray grade standards and subsequent catalytic applications, operators must exercise precise control over the molar ratios and solvent conditions during the reflux stage. The following guide outlines the critical operational steps necessary to replicate the successful preparation of this high-value chiral complex, ensuring that the final material meets the stringent specifications demanded by fine chemical applications. Detailed standardized synthesis steps are provided below to facilitate immediate technology transfer.
- Accurately weigh (L)-phenylalaninol and anhydrous zinc chloride in a strict 3: 1 molar ratio using an electronic balance.
- Combine reagents in a round-bottom flask with absolute ethyl alcohol and heat under reflux with stirring for 48 hours.
- Perform hot filtration, concentrate the filtrate, and allow slow volatilization to crystallize faint yellow crystals suitable for X-ray diffraction.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this chiral zinc complex technology translates directly into tangible strategic advantages that bolster the bottom line and mitigate operational risks. The primary driver of value lies in the drastic simplification of the raw material portfolio; by replacing expensive, imported chiral ligands and precious metal salts with commodity chemicals like L-phenylalaninol and zinc chloride, organizations can achieve significant cost reduction in fine chemical manufacturing. This shift not only lowers the direct material costs but also insulates the production schedule from the volatility of the precious metals market, ensuring stable pricing and reliable budget forecasting for long-term projects. Furthermore, the elimination of toxic heavy metals from the catalyst formulation simplifies waste disposal protocols and reduces the environmental compliance burden, aligning perfectly with modern green chemistry initiatives and corporate sustainability goals.
- Cost Reduction in Manufacturing: The economic benefits of this process are profound, stemming from the use of earth-abundant zinc instead of scarce noble metals, which inherently lowers the cost basis of the catalyst per kilogram. Additionally, the one-step synthesis eliminates multiple purification and isolation stages typically required for complex ligand preparation, resulting in substantial savings in labor, energy, and solvent consumption. The high atom economy of the reaction ensures that nearly all input materials are incorporated into the final product or easily recoverable byproducts, minimizing waste disposal fees and maximizing overall process efficiency.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical concern for global supply chains, and this technology addresses it by relying on L-phenylalaninol, a mass-produced amino acid derivative available from multiple global suppliers. This diversification of the supply base reduces the risk of single-source dependency and ensures continuity of supply even during geopolitical disruptions or logistics bottlenecks. The robust nature of the final crystalline product also allows for extended storage periods without significant degradation, enabling manufacturers to build strategic inventory buffers that protect against sudden spikes in demand or temporary production halts.
- Scalability and Environmental Compliance: The scalability of this process is exceptional due to the use of standard unit operations such as reflux, filtration, and crystallization, which are well-understood and easily replicated at the multi-ton scale. The absence of hazardous reagents and the use of ethanol as a primary solvent significantly reduce the safety risks associated with scale-up, lowering insurance premiums and facility maintenance costs. Moreover, the simplified waste stream, devoid of heavy metal contamination, facilitates easier treatment and discharge, ensuring full compliance with increasingly stringent environmental regulations across different jurisdictions.
Frequently Asked Questions (FAQ)
To assist technical decision-makers in evaluating the feasibility of integrating this chiral zinc complex into their existing workflows, we have compiled a set of frequently asked questions based on the specific technical data and experimental results presented in the patent documentation. These answers address common concerns regarding structural stability, catalytic scope, and operational parameters, providing a clear framework for assessing the technology's fit for specific synthetic challenges. The information provided here is derived directly from the empirical data to ensure accuracy and reliability for your internal review processes.
Q: What are the primary structural characteristics of this chiral zinc complex?
A: The complex features a unique coordination geometry where zinc centers are bridged by oxygen and nitrogen atoms from the phenylalaninol ligands, confirmed by X-ray single crystal diffraction analysis showing specific bond lengths such as Zn-O at approximately 1.928 Å.
Q: Is this catalyst suitable for large-scale industrial production?
A: Yes, the synthesis utilizes inexpensive, commercially available starting materials like L-phenylalaninol and zinc chloride, and employs a simple reflux procedure in ethanol, making it highly amenable to scale-up without requiring exotic equipment.
Q: What specific reactions does this complex catalyze effectively?
A: The complex has demonstrated catalytic activity in the asymmetric cyanosilylation of aldehydes, such as the conversion of benzaldehyde to 2-phenyl-2-(trimethylsiloxy)acetonitrile, serving as a key intermediate in organic synthesis.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Zinc Complex Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the tris[(S)-phenylalaninol] zinc chloride complex in driving innovation within the pharmaceutical and fine chemical industries. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from laboratory discovery to market-ready product is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation capable of verifying stringent purity specifications, guaranteeing that every batch of catalyst delivered meets the exacting standards required for sensitive asymmetric synthesis applications.
We invite you to collaborate with our technical team to explore how this cost-effective catalytic solution can optimize your specific manufacturing processes. Please contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your production volume, along with specific COA data and route feasibility assessments that demonstrate the tangible benefits of partnering with us for your chiral intermediate needs.
