Advanced Selective Nitration of Sorbitan for High-Purity 5-Isosorbide Mononitrate Production
The pharmaceutical industry continuously seeks robust synthetic pathways for cardiovascular therapeutics, particularly for anti-anginal agents like 5-isosorbide mononitrate (5-ISMN). A pivotal advancement in this domain is detailed in patent CN107011354B, which discloses a novel preparation method utilizing a silver nitrate and thionyl chloride system. This technology represents a significant departure from conventional multi-step syntheses, offering a direct, selective nitration route starting from sorbitan. For R&D directors and procurement specialists, this patent outlines a pathway that not only enhances chemical purity to levels exceeding 99% but also drastically simplifies the downstream processing infrastructure. By leveraging a heterogeneous reaction system where byproducts precipitate out, the method addresses critical pain points regarding waste management and yield optimization that have long plagued the manufacturing of this vital API intermediate.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 5-isosorbide mononitrate has been fraught with challenges related to selectivity and safety. Traditional direct nitration processes, as illustrated in early literature, involve treating isosorbide with concentrated nitric acid. This approach invariably generates a complex mixture containing 2-isosorbide mononitrate, isosorbide dinitrate, and unreacted starting materials. The separation of these closely related isomers typically requires energy-intensive column chromatography, which is economically unviable for ton-scale production. Furthermore, the presence of isosorbide dinitrate poses a severe safety hazard due to its explosive nature under heating or pressurization conditions.
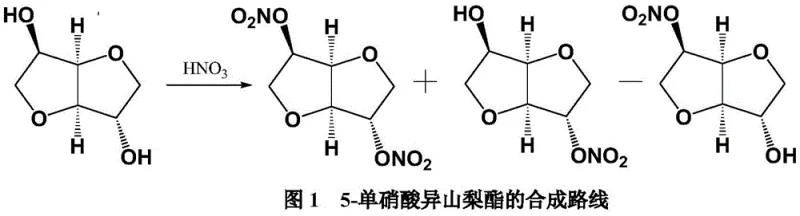
Alternative strategies involving selective reduction of isosorbide dinitrate have also been explored, utilizing reagents such as zinc powder, hydrazine hydrate, or expensive transition metal catalysts like ruthenium complexes. While some of these methods achieve moderate selectivity, they introduce substantial environmental burdens. For instance, zinc powder reduction generates large quantities of zinc salt waste, complicating effluent treatment. Similarly, catalytic hydrogenation methods often suffer from poor catalyst recyclability and the risk of over-reduction to isosorbide, necessitating rigorous purification steps that erode overall process efficiency and increase the cost of goods sold.
The Novel Approach
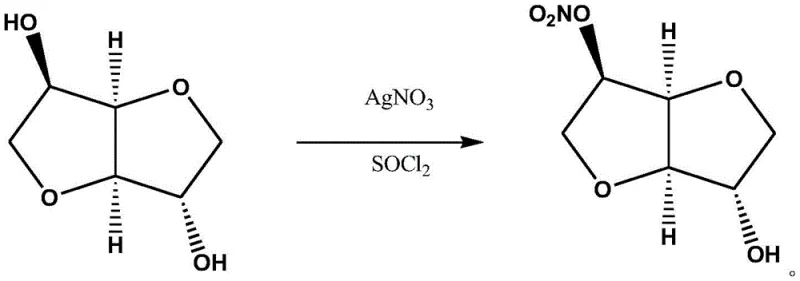
In stark contrast to these legacy methods, the technology described in CN107011354B introduces a streamlined one-pot nitration strategy. By employing sorbitan as the substrate and a silver nitrate/thionyl chloride system as the nitrating agent, the process achieves exceptional regioselectivity for the 5-hydroxyl group. The reaction proceeds under mild conditions, typically between -20°C and 10°C, in common dry organic solvents such as ethyl acetate or dichloromethane. A key feature of this novelty is the in-situ formation of silver chloride precipitate, which can be effortlessly removed via simple suction filtration. This physical separation mechanism bypasses the need for complex aqueous workups or chromatographic purification, directly yielding a crude product of high purity that requires only decolorization and concentration to meet pharmaceutical standards.
Mechanistic Insights into AgNO3/SOCl2 Mediated Nitration
The efficacy of this synthetic route lies in the unique reactivity of the thionyl chloride and silver nitrate couple. Thionyl chloride acts as a potent activating agent, likely converting the hydroxyl group of the sorbitan into a reactive chlorosulfite intermediate or facilitating the formation of a nitronium ion equivalent in the presence of the silver salt. Silver nitrate serves a dual purpose: it provides the nitrate source for the esterification and acts as a halide scavenger. As the reaction progresses, the chloride ions released from thionyl chloride react immediately with silver ions to form insoluble silver chloride (AgCl). This precipitation drives the equilibrium forward according to Le Chatelier's principle, ensuring high conversion rates without the need for a large excess of reagents.
From an impurity control perspective, the choice of sorbitan (1,4-anhydro-D-sorbitol) is mechanistically superior to isosorbide (1,4:3,6-dianhydro-D-sorbitol). Sorbitan possesses a primary and a secondary hydroxyl group, but the specific steric and electronic environment created by the 1,4-anhydro ring structure favors nitration at the secondary 5-position under these specific conditions. The mild temperature window of -5°C to 0°C further kinetically suppresses the nitration of the less reactive positions or the formation of the dinitrate species. Consequently, the impurity profile is significantly cleaner, with HPLC data from the patent examples indicating purities consistently above 99.7%, effectively eliminating the costly burden of isomer separation that plagues direct nitration of isosorbide.
How to Synthesize 5-Isosorbide Mononitrate Efficiently
The operational protocol derived from this patent is designed for scalability and safety, minimizing the handling of hazardous intermediates. The process begins with the dissolution of sorbitan in a dry organic solvent, followed by the controlled addition of reagents at low temperatures to manage exotherms. The simplicity of the workup—filtration, washing, drying, and concentration—makes this an ideal candidate for technology transfer from lab to pilot plant. For detailed standard operating procedures and specific stoichiometric ratios optimized for your facility, please refer to the technical guide below.
- Dissolve sorbitan in a dry organic solvent such as ethyl acetate or dichloromethane and cool the mixture to a temperature range of -20°C to 10°C.
- Add solid silver nitrate followed by the slow dropwise addition of thionyl chloride, maintaining the low temperature to ensure selective nitration at the 5-hydroxyl position.
- Filter off the silver chloride precipitate, wash the filtrate with water to neutrality, dry the organic phase, decolorize with activated carbon, and concentrate to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this AgNO3/SOCl2 mediated synthesis offers transformative economic benefits. The most immediate impact is the elimination of column chromatography, a unit operation that is notoriously difficult to scale and expensive to maintain in terms of silica gel consumption and solvent usage. By replacing this with a simple filtration step, the process drastically reduces cycle times and solvent recovery costs. Furthermore, the avoidance of heavy metal catalysts like ruthenium or palladium removes the need for expensive metal scavenging resins and the associated regulatory testing for residual metals in the final API, streamlining the quality control workflow.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the high atom economy and the use of commodity chemicals. Silver nitrate, while a premium reagent, is used in stoichiometric amounts and the resulting silver chloride byproduct has scrap value, allowing for potential recovery of silver content which offsets raw material costs. Additionally, the high yield reported in the patent examples, often exceeding 85%, minimizes the loss of valuable starting materials. The reduction in processing steps—from multi-step reduction sequences to a single nitration event—lowers labor costs and utility consumption, contributing to a substantially lower cost of goods sold compared to traditional reduction-based routes.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on widely available raw materials. Sorbitan is a bulk chemical derived from sorbitol, ensuring a stable upstream supply base unlike specialized chiral catalysts which may have long lead times. The robustness of the reaction conditions, which tolerate a range of common solvents including ethyl acetate and dichloromethane, provides flexibility in sourcing. This adaptability ensures that production schedules are not disrupted by shortages of specific niche reagents, making the supply of high-purity 5-isosorbide mononitrate more predictable and reliable for downstream formulation partners.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this method offers a cleaner profile. The absence of zinc waste or hydrazine derivatives simplifies wastewater treatment protocols, reducing the load on effluent treatment plants. The mild reaction temperatures reduce the energy footprint associated with heating or cryogenic cooling. Moreover, the process avoids the accumulation of energetic dinitrate intermediates in the reaction mass, enhancing operational safety during scale-up. These factors collectively facilitate easier regulatory approval for new manufacturing sites and support sustainable manufacturing goals.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this nitration technology. They are derived from the specific experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational feasibility and quality attributes of the resulting 5-ISMN.
Q: What are the primary advantages of the AgNO3/SOCl2 nitration system over traditional reduction methods?
A: The AgNO3/SOCl2 system eliminates the need for expensive transition metal catalysts like Ruthenium or Palladium and avoids the generation of heavy metal waste associated with Zinc powder reduction. It offers a simpler workup via filtration of insoluble AgCl and achieves high purity without column chromatography.
Q: How does this method control the formation of the 2-isosorbide mononitrate isomer?
A: By using sorbitan (1,4-anhydro-D-sorbitol) as the starting material instead of isosorbide or isosorbide dinitrate, the reaction inherently targets the secondary hydroxyl group at the 5-position. The mild reaction conditions (-5°C to 0°C) further suppress isomerization and over-nitration.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process is highly scalable due to its mild temperature requirements, the use of common organic solvents like ethyl acetate, and the absence of complex purification steps like silica gel column chromatography, which are bottlenecks in traditional synthesis routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Isosorbide Mononitrate Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting next-generation synthetic routes like the AgNO3/SOCl2 nitration system. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our engineering teams are adept at optimizing the filtration and solvent recovery units essential for this specific chemistry, ensuring that the theoretical benefits of high purity and yield are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs equipped to detect trace isomers, guaranteeing that every batch meets the exacting standards required for cardiovascular pharmaceuticals.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. By switching to this more efficient synthesis, you can achieve significant operational efficiencies and cost savings. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are ready to provide specific COA data from our pilot runs and comprehensive route feasibility assessments to support your regulatory filings and product launches.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
