Advanced Selective Nitration Technology for High-Purity 5-Isosorbide Mononitrate Manufacturing
Advanced Selective Nitration Technology for High-Purity 5-Isosorbide Mononitrate Manufacturing
The pharmaceutical industry constantly seeks robust, scalable, and safe methodologies for producing critical cardiovascular intermediates. Patent CN110407846B represents a significant technological leap in the synthesis of 5-isosorbide mononitrate (5-ISMN), a vital active pharmaceutical ingredient (API) used globally for the treatment of angina pectoris and coronary heart disease. This proprietary technology addresses long-standing challenges in nitration chemistry, specifically the issues of regioselectivity and hazardous by-product formation that have plagued traditional manufacturing routes. By leveraging a novel combination of trimethylchlorosilane (TMSCl), nitrates, and aluminum trichloride, this method achieves exceptional purity levels exceeding 99% without the need for complex chromatographic purification. For R&D directors and procurement specialists, understanding the mechanistic advantages of this patent is crucial for securing a reliable 5-ISMN supplier capable of delivering consistent quality while mitigating supply chain risks associated with hazardous chemical processing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 5-isosorbide mononitrate has relied heavily on direct nitration processes using fuming nitric acid, a method first reported in the mid-20th century. As illustrated in the reaction scheme below, this conventional approach suffers from poor regioselectivity, inevitably generating a complex mixture containing the desired 5-mononitrate alongside the unwanted 2-isosorbide mononitrate and isosorbide dinitrate.
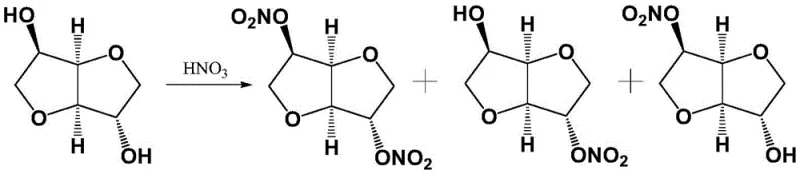
The presence of isosorbide-2,5-dinitrate is particularly concerning from a safety perspective, as this by-product carries a significant risk of explosion under conditions of heating or pressurization, posing severe threats to plant safety and operational continuity. Furthermore, separating the target 5-mononitrate from this hazardous mixture traditionally requires energy-intensive and solvent-heavy column chromatography or multiple recrystallization steps. Alternative routes, such as the esterification-nitration pathway shown in the following figure, attempt to mitigate some selectivity issues but introduce additional synthetic steps involving acylation and subsequent hydrolysis, which drastically increase production costs and environmental waste burdens.
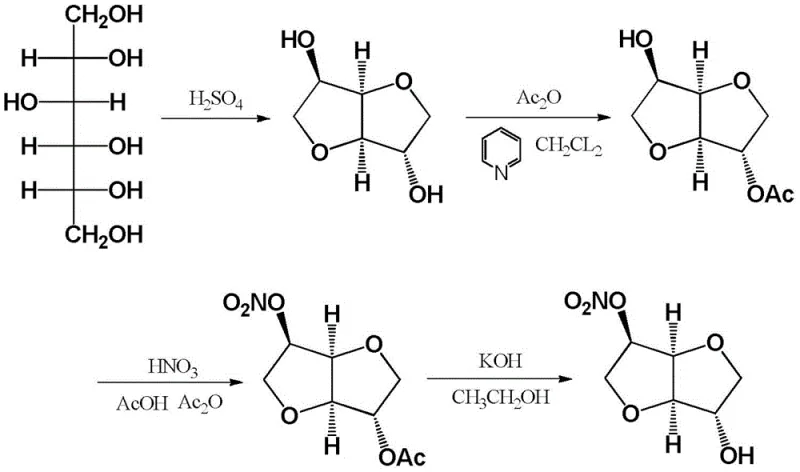
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN110407846B introduces a streamlined, one-pot selective nitration strategy that fundamentally redefines the efficiency of 5-ISMN manufacturing. This innovative process utilizes TMSCl and a nitrate salt in the presence of an organic base and aluminum trichloride to achieve highly specific nitration at the 5-hydroxyl position of the isosorbide backbone. The reaction conditions are remarkably mild, typically operating between -20°C and 10°C, which not only preserves the structural integrity of the sensitive bicyclic sugar alcohol but also minimizes thermal degradation and side reactions. By avoiding the use of fuming nitric acid as the primary nitrating agent, the process inherently eliminates the formation of the explosive dinitrate impurity, thereby resolving the critical safety bottleneck that has limited the scalability of previous methods. The result is a crude product of such high purity that it bypasses the need for column chromatography entirely, requiring only simple extraction and decolorization to meet stringent pharmaceutical standards.
Mechanistic Insights into TMSCl-Mediated Selective Nitration
The core innovation of this patent lies in the sophisticated interplay between the silylating agent (TMSCl) and the Lewis acid catalyst (AlCl3). Initially, the reaction of isosorbide with TMSCl and an organic base, such as triethylamine, serves to transiently protect the hydroxyl groups or activate the substrate through silyl ether formation. This step is critical for modulating the nucleophilicity of the oxygen atoms within the isosorbide ring system. Subsequently, the addition of aluminum trichloride acts as a powerful Lewis acid, coordinating with the nitrate source to generate a highly reactive yet controlled nitronium-like species in situ. This electrophilic complex exhibits a strong preference for attacking the sterically and electronically favored 5-hydroxyl position over the 2-position, driven by the specific conformational constraints of the 1,4:3,6-dianhydro-D-sorbitol structure.
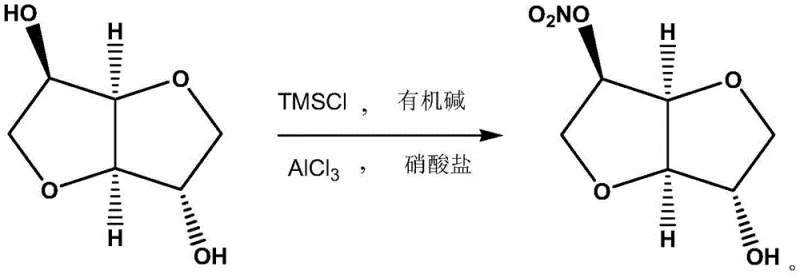
From an impurity control perspective, this mechanism is exceptionally robust. The mild reaction temperature prevents the over-nitration that leads to dinitrate formation, while the specific catalytic environment suppresses the rearrangement or migration of the nitro group. Furthermore, the by-products generated, primarily silicon ethers, are chemically distinct from the product and can be easily separated during the aqueous workup or potentially recycled back into the process. This level of mechanistic precision ensures that the final impurity profile is clean, with HPLC analysis consistently showing purity levels above 99.7%, significantly reducing the burden on downstream quality control laboratories and ensuring batch-to-batch consistency essential for regulatory compliance in global markets.
How to Synthesize 5-Isosorbide Mononitrate Efficiently
The implementation of this synthesis route requires precise control over stoichiometry and temperature to maximize the benefits of the selective nitration mechanism. The process begins with the suspension of isosorbide in an organic solvent, followed by the controlled addition of TMSCl and a base like triethylamine at low temperatures. Once the silylation equilibrium is established, the nitrating system comprising aluminum trichloride and a solid nitrate salt is introduced gradually to maintain thermal stability. The detailed standardized synthesis steps, including specific mass ratios and quenching protocols, are outlined in the guide below.
- Protect the hydroxyl groups of isosorbide by reacting with TMSCl and an organic base like triethylamine at low temperatures (-20°C to 10°C).
- Introduce aluminum trichloride and a nitrate salt (sodium or potassium nitrate) to the mixture to selectively nitrate the 5-hydroxyl position.
- Quench the reaction in ice water, adjust pH to neutral, extract with organic solvent, dry, decolorize with activated carbon, and concentrate to obtain the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology translates into tangible operational improvements and risk mitigation strategies. The elimination of hazardous explosive by-products removes a major regulatory and safety hurdle, allowing for smoother permitting and reduced insurance costs associated with nitration facilities. Moreover, the simplification of the purification process—removing the need for silica gel column chromatography—drastically reduces solvent consumption and waste disposal volumes, aligning with modern green chemistry initiatives and lowering the overall environmental footprint of the manufacturing site.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the drastic simplification of the downstream processing workflow. By eliminating the need for expensive and time-consuming column chromatography, manufacturers save significantly on stationary phase materials, solvent usage, and labor hours required for fraction collection and analysis. Additionally, the high yield reported in the patent examples, often exceeding 90%, ensures that raw material utilization is maximized, directly lowering the cost of goods sold (COGS) per kilogram of active ingredient. The ability to recycle silicon-based by-products further contributes to a circular economy model within the plant, reducing the net consumption of reagents.
- Enhanced Supply Chain Reliability: Safety is a critical component of supply chain continuity. Traditional nitration processes carrying explosion risks are prone to unplanned shutdowns due to safety incidents or rigorous regulatory inspections. This new method, operating under mild conditions without explosive intermediates, offers a much more stable and predictable production environment. The use of common, commercially available reagents like sodium nitrate and TMSCl ensures that the supply of raw materials is robust and not subject to the volatility often seen with specialized nitrating mixtures, thereby securing the continuity of supply for downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling nitration reactions is notoriously difficult due to heat management and safety concerns. The mild exothermic nature of this TMSCl-mediated reaction allows for safer scale-up from pilot plants to multi-ton commercial reactors without requiring exotic cooling infrastructure. The post-treatment involves simple phase separations and filtrations, which are unit operations easily handled in standard stainless steel equipment. Furthermore, the reduction in "three wastes" (wastewater, waste gas, and solid waste) simplifies compliance with increasingly strict environmental regulations, preventing production delays caused by waste treatment bottlenecks.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this selective nitration technology. These answers are derived directly from the experimental data and technical disclosures found within the patent documentation, providing clarity on the feasibility and advantages of adopting this route for commercial production.
Q: Why is the TMSCl/nitrate system superior to traditional fuming nitric acid nitration?
A: Traditional direct nitration with fuming nitric acid often results in a mixture of 2-mononitrate, dinitrate, and unreacted starting material, requiring difficult column chromatography. The TMSCl/nitrate system described in CN110407846B offers high regioselectivity for the 5-position, eliminating explosive by-products and simplifying purification.
Q: What are the safety advantages of this new nitration process?
A: Conventional methods produce isosorbide-2,5-dinitrate, which poses significant explosion risks under heating or pressurization. The novel selective nitration method avoids the formation of this hazardous dinitrate by-product, significantly enhancing operational safety for industrial scale-up.
Q: Does this process require complex post-treatment steps?
A: No. Unlike prior art that necessitates column chromatography or salification, this method allows for simple workup involving phase separation, extraction, and activated carbon decolorization. The silicon ether by-products can be recycled, reducing waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-Isosorbide Mononitrate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced manufacturing technologies requires a partner with deep technical expertise and proven execution capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN110407846B are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of 5-isosorbide mononitrate meets the highest global pharmacopoeia standards, providing our partners with absolute confidence in product quality.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cutting-edge synthesis route. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to evaluate how our optimized manufacturing process can enhance your supply chain resilience and drive down costs for your cardiovascular drug portfolio.
