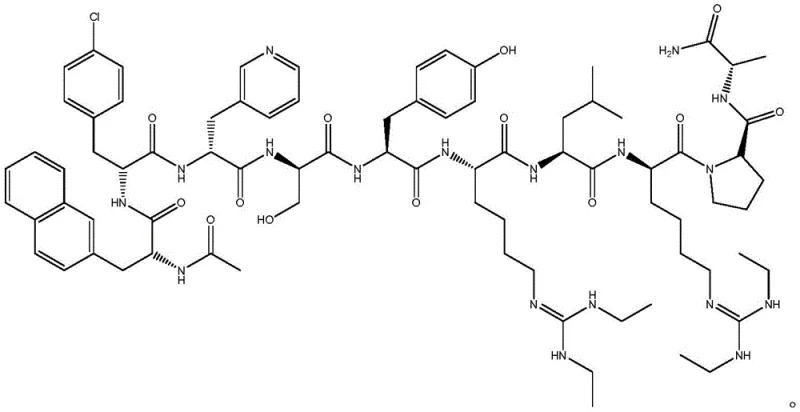
Advanced 8+2 Fragment Solid-Phase Synthesis Strategy for High-Purity Ganirelix Manufacturing
The pharmaceutical landscape for GnRH antagonists is undergoing a significant transformation driven by the need for greener, more efficient manufacturing processes. Patent CN114478708B introduces a groundbreaking 8+2 fragment solid-phase synthesis method for ganirelix, a critical decapeptide drug used in assisted reproductive technology. This innovation addresses the longstanding challenges of traditional peptide synthesis, specifically targeting the reduction of toxic reagent usage and the enhancement of crude peptide purity. By strategically dividing the ten-amino-acid sequence into an octapeptide fragment A[3-10] and a dipeptide intermediate B[1-2], the process drastically simplifies the purification burden. This technical breakthrough is not merely a laboratory curiosity but represents a viable pathway for industrial-scale production, offering a robust solution for reliable polypeptide intermediate supplier networks seeking to optimize their portfolios. The method ensures that the final active pharmaceutical ingredient meets stringent quality standards while mitigating the environmental hazards associated with legacy synthesis routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex peptides like ganirelix has been plagued by inefficient strategies that compromise both yield and environmental safety. Traditional approaches often relied on BOC (tert-butyloxycarbonyl) solid-phase synthesis, which necessitates the use of trifluoroacetic acid (TFA) for deprotection at every step and, more critically, highly toxic hydrogen fluoride (HF) or trifluoromethanesulfonic acid (TFMSA) for final cleavage. These hazardous materials pose severe risks to operator safety and require expensive waste treatment infrastructure, significantly inflating the cost reduction in peptide manufacturing efforts. Furthermore, alternative Fmoc-based strategies found in prior art, such as 5+5 or 6+4 fragment condensations, often suffer from lower crude purity levels, typically hovering around 90-94%. These methods frequently involve multiple side-chain deprotection and modification steps, such as the use of hydrazine hydrate to remove Dde protecting groups, which extends the reaction timeline and introduces additional impurities like deletion peptides and isomers that are difficult to separate.
The Novel Approach
In stark contrast, the novel 8+2 fragment synthesis method disclosed in the patent data revolutionizes the production workflow by eliminating the need for extremely toxic cleavage reagents and minimizing side-chain manipulation. By synthesizing the shorter dipeptide fragment B[1-2] separately on a 2-Chlorotrityl Chloride Resin, the process allows for rigorous purification of this intermediate via simple ether recrystallization before it is ever coupled to the main chain. This strategic decoupling ensures that impurities from the N-terminal region do not propagate through the entire synthesis, thereby elevating the crude peptide purity to an impressive 95%. The method utilizes standard Fmoc chemistry with piperidine for deprotection and a mild TFA/TIS/EDT cleavage cocktail that avoids water, preventing product stickiness and hydrolysis. This streamlined approach not only shortens the overall reaction period but also significantly enhances the commercial scale-up of complex polypeptide intermediates by reducing the load on downstream purification columns.
Mechanistic Insights into Fmoc Solid-Phase Peptide Synthesis
The core of this technological advancement lies in the precise control of reaction conditions during the coupling phases, particularly the implementation of ice-bath pre-activation. In standard peptide synthesis, the activation of carboxyl groups can lead to racemization, especially with sterically hindered or sensitive amino acids. The patent specifies that Fmoc-protected amino acids must be pre-activated using condensing agents like HOBT/DIC or HATU/DIEA under ice-bath conditions (0-5°C) for 30 minutes prior to addition to the resin. This low-temperature environment kinetically suppresses the formation of oxazolone intermediates, which are the primary precursors to racemization and epimerization. By filtering the pre-activated solution before it contacts the resin, insoluble urea byproducts (such as DIU from DIC) are removed, preventing pore blockage within the resin matrix and ensuring uniform reagent diffusion. This meticulous attention to activation thermodynamics is crucial for maintaining the stereochemical integrity of the chiral centers within the ganirelix sequence.
Furthermore, the impurity control mechanism is deeply embedded in the choice of the cleavage reagent system. Unlike conventional methods that may introduce water or scavengers that lead to side reactions, this protocol employs a specific mass ratio of TFA:TIS:EDT (95:3:2). The absence of water in the cleavage cocktail is a critical design choice that prevents the hydrolysis of sensitive amide bonds and reduces the formation of sticky residues that can trap impurities. The use of triisopropylsilane (TIS) and 1,2-ethanedithiol (EDT) acts as effective scavengers for carbocations generated during the acidolysis of side-chain protecting groups, such as the t-butyl groups on Serine and Tyrosine. This results in a crude product profile that is remarkably clean, with a maximum single impurity content as low as 0.02% after purification, demonstrating the efficacy of this mechanistic approach in producing high-purity OLED material grade chemicals, although here applied to pharma.
How to Synthesize Ganirelix Efficiently
The synthesis of ganirelix via this patented route requires strict adherence to the 8+2 fragment strategy to maximize yield and purity. The process begins with the assembly of the C-terminal octapeptide on a Rink Amide resin, followed by the independent synthesis of the N-terminal dipeptide. The convergence of these two fragments is the critical step where the efficiency of the entire process is realized. Detailed operational parameters, including resin substitution degrees and specific molar equivalents of activators, are essential for reproducibility. For a comprehensive understanding of the standardized operating procedures required to implement this technology in a GMP environment, please refer to the technical guide below.
- Prepare Ganirelix peptide resin fragment A[3-10] using Fmoc-D-Ala-Rink AM Amide resin and sequential coupling of protected amino acids.
- Synthesize intermediate fragment B[1-2] (Ac-D-Nal-D-Cpa-OH) on 2-Chlorotrityl Chloride Resin and purify via ether recrystallization.
- Couple fragment B to fragment A, cleave the resin using TFA/TIS/EDT, and purify the crude peptide to achieve >99.9% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this 8+2 synthesis methodology translates into tangible strategic benefits that extend beyond simple unit pricing. The elimination of hazardous reagents like HF and hydrazine hydrate removes the need for specialized containment equipment and costly waste disposal protocols, leading to substantial cost savings in facility operations. Moreover, the significant increase in crude peptide purity from typical levels of 90% to over 95% means that the burden on preparative HPLC purification is drastically reduced. This efficiency gain prolongs the service life of expensive chromatography columns and reduces solvent consumption, directly impacting the bottom line. By simplifying the synthetic route and reducing the number of reaction steps, manufacturers can achieve faster batch turnover times, enhancing the reliability of the supply chain for critical fertility treatments.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven by the qualitative elimination of expensive and dangerous reagents. By avoiding the use of hydrogen fluoride and complex side-chain modification steps involving hydrazine, the process reduces the capital expenditure required for safety infrastructure and waste treatment. Additionally, the high purity of the intermediate fragment B[1-2], achieved through simple recrystallization, minimizes the loss of valuable materials during the final coupling stage. This efficiency ensures that raw material utilization is optimized, driving down the cost of goods sold (COGS) without compromising on the quality of the final active pharmaceutical ingredient.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of peptide synthesis and the scarcity of specialized reagents. This method utilizes widely available Fmoc-protected amino acids and standard condensing agents, reducing the risk of supply bottlenecks. The robustness of the 8+2 fragment approach means that production schedules are less susceptible to delays caused by failed coupling steps or extensive rework. Consequently, partners can rely on a more predictable lead time for high-purity polypeptide intermediates, ensuring that downstream formulation and packaging operations proceed without interruption.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental sustainability intensify, the ability to scale production without increasing the toxic footprint is paramount. This synthesis route is inherently greener, generating less hazardous waste and avoiding the release of volatile toxic gases associated with HF cleavage. The process is designed for industrial mass production, with reaction conditions that are easily transferable from pilot scale to multi-ton commercial batches. This scalability ensures that manufacturers can meet growing global demand for ganirelix while maintaining compliance with increasingly stringent environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis protocol. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, highlighting the specific advantages over prior art methods. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this new standard for peptide manufacturing.
Q: How does the 8+2 fragment method improve purity compared to conventional 5+5 synthesis?
A: The 8+2 strategy minimizes the accumulation of deletion sequences and racemization by reducing the number of coupling cycles on the longer fragment, resulting in crude peptide purity exceeding 95%.
Q: What are the environmental advantages of this synthesis route?
A: This method eliminates the use of highly toxic reagents such as hydrogen fluoride (HF) and hydrazine hydrate, replacing them with safer Fmoc chemistry and TFA-based cleavage systems.
Q: Why is ice-bath pre-activation critical in this process?
A: Pre-activating amino acids at 0-5°C significantly reduces racemization and the formation of difficult-to-remove isomer impurities, ensuring higher final product quality.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ganirelix Supplier
The technical potential of the 8+2 fragment synthesis method represents a significant leap forward in the field of polypeptide pharmaceuticals, offering a pathway to superior product quality and operational efficiency. NINGBO INNO PHARMCHEM stands at the forefront of this innovation, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced purification systems capable of meeting the stringent purity specifications required for clinical and commercial-grade ganirelix. We understand that the transition to a new synthesis route requires a partner with deep technical expertise and a commitment to quality assurance.
We invite global pharmaceutical partners to collaborate with us to unlock the full value of this patented technology. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to determine how this advanced manufacturing process can enhance your supply chain resilience and product competitiveness in the global market.
