Revolutionizing GnRH Antagonist Production: Scalable High-Purity Ganirelix Acetate Manufacturing
Revolutionizing GnRH Antagonist Production: Scalable High-Purity Ganirelix Acetate Manufacturing
The pharmaceutical landscape for fertility treatments is constantly evolving, demanding higher standards of purity and safety for active pharmaceutical ingredients. A significant breakthrough in this domain is detailed in patent CN109879938B, which outlines a novel preparation method for ganirelix acetate, a critical gonadotropin-releasing hormone (GnRH) antagonist. This technology addresses the persistent challenges associated with peptide purification, specifically targeting the removal of isomer impurities and toxic residues that plague conventional synthesis routes. By leveraging a sophisticated dual-stage chromatographic approach, manufacturers can now achieve unprecedented purity levels while streamlining the production workflow. This report analyzes the technical merits of this innovation, offering strategic insights for R&D directors, procurement managers, and supply chain leaders seeking to optimize their API intermediate sourcing strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional manufacturing processes for ganirelix acetate often rely on solid-phase synthesis followed by acid cleavage, typically utilizing trifluoroacetic acid (TFA). While effective for cleavage, the subsequent purification and salt conversion steps frequently introduce significant complications. Prior art methods, such as those disclosed in earlier patents, often employ mobile phases containing TFA or hazardous salts like sodium perchlorate to facilitate purification. The use of perchlorates poses severe safety risks due to their instability and explosive nature, making them unsuitable for large-scale industrial applications. Furthermore, the introduction of these extraneous ions necessitates complex desalting procedures post-purification, adding multiple unit operations that increase production time and cost. The residual presence of TFA is another critical concern, as it requires rigorous removal to meet safety specifications, often resulting in yield loss and inconsistent product quality.
The Novel Approach
In stark contrast, the methodology described in CN109879938B introduces a streamlined, two-step chromatographic process that entirely eliminates the need for hazardous perchlorates and avoids the reintroduction of TFA during purification. The core innovation lies in the exclusive use of an acetic acid aqueous solution and an acetic acid acetonitrile solution system as the eluent throughout the entire process. This strategic choice of mobile phase not only facilitates the simultaneous purification and salt conversion of the crude peptide but also ensures that no new ionic impurities are introduced into the system. By bypassing the need for separate salt exchange steps and dangerous reagents, this approach significantly simplifies the operational workflow. The result is a robust, scalable process that delivers ganirelix acetate with exceptional purity metrics, directly addressing the safety and efficiency bottlenecks inherent in legacy manufacturing techniques.
Mechanistic Insights into Dual-Stage Chromatographic Purification
The technical superiority of this method is rooted in its precise control over chromatographic conditions, utilizing alkylsilane-bonded silica gel, preferably octadecylsilane (C18), as the stationary phase. The process is divided into two distinct stages, each optimized for specific separation goals. In the first stage, the crude ganirelix peptide, often containing TFA from the initial cleavage, is dissolved in a mixture of water and acetic acid. This solution is loaded onto the column and subjected to gradient elution where the volume ratio of the aqueous acetic acid phase to the acetonitrile acetic acid phase shifts from 95:5 to 20:80. This gradient effectively separates the target peptide from major impurities while simultaneously converting the salt form to acetate. The second stage further refines the product using a tighter gradient range, shifting from 85:15 to 65:35, to remove trace isomers and structurally similar impurities that are notoriously difficult to separate in peptide chemistry.
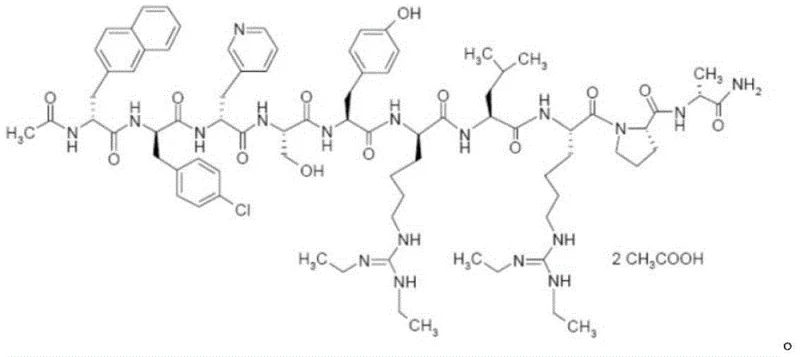
Controlling the impurity profile is paramount for GnRH antagonists, where even minor structural variants can impact biological activity and safety. The described method achieves a maximum single impurity content of merely 0.04%, a testament to the high resolution of the dual-column strategy. The absence of extra ions, such as sodium or phosphate, eliminates the need for downstream ion-exchange treatments, which are often sources of yield degradation. Moreover, the complete absence of trifluoroacetic acid residue in the final product is guaranteed by the exclusive use of acetic acid buffers in the purification train. This mechanistic precision ensures that the final API meets the most stringent pharmacopeial requirements, providing a reliable foundation for formulating fertility medications with consistent therapeutic outcomes.
How to Synthesize Ganirelix Acetate Efficiently
Implementing this synthesis route requires careful attention to solvent ratios and flow rates to maximize the efficiency of the chromatographic separation. The process begins with the preparation of the crude peptide solution, followed by loading onto a pre-equilibrated C18 column. The key to success lies in the precise execution of the gradient elution profiles defined in the patent, which balance resolution with processing time. While the general framework is established, specific operational parameters may require optimization based on equipment scale and column dimensions. For a comprehensive breakdown of the standardized operating procedures, including specific flow rates, equilibration times, and concentration steps, please refer to the technical guide below.
- Dissolve crude ganirelix peptide in an acetic acid and water mixture, filter through a 0.45 μm membrane, and load onto an octadecylsilane-bonded silica column.
- Perform gradient elution using acetic acid aqueous solution and acetic acid acetonitrile solution to convert salts and remove initial impurities.
- Execute a second purification stage with refined gradient parameters to isolate ganirelix acetate with purity exceeding 99.8% before lyophilization.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this purification technology offers substantial benefits that extend beyond mere technical compliance. For procurement managers and supply chain heads, the elimination of hazardous reagents like sodium perchlorate translates directly into reduced regulatory burdens and lower handling costs. The simplified workflow, which merges purification and salt conversion into a seamless continuous process, drastically reduces the number of unit operations required. This consolidation not only shortens the overall manufacturing cycle time but also minimizes the potential for human error and batch-to-batch variability. Consequently, suppliers utilizing this method can offer more competitive pricing structures while maintaining higher margins, creating a win-win scenario for both manufacturers and their downstream pharmaceutical partners.
- Cost Reduction in Manufacturing: The exclusion of expensive and hazardous reagents such as sodium perchlorate eliminates the need for specialized waste disposal protocols and safety infrastructure, leading to significant operational cost savings. Furthermore, by removing the necessity for additional desalting steps and ion-exchange treatments, the process reduces solvent consumption and labor hours. The high recovery rates associated with this gentle acetic acid-based system ensure that valuable peptide material is not lost during purification, maximizing the yield from expensive starting materials. These efficiencies collectively drive down the cost of goods sold, allowing for more aggressive pricing strategies in the competitive API market.
- Enhanced Supply Chain Reliability: Relying on common, non-hazardous solvents like acetic acid and acetonitrile mitigates the risk of supply disruptions often associated with regulated or dangerous chemicals. The robustness of the process ensures consistent batch quality, reducing the likelihood of failed inspections or rejected shipments that can derail production schedules. With a simplified process flow, manufacturers can respond more agilely to fluctuations in demand, scaling production up or down without compromising on purity or safety standards. This reliability is crucial for maintaining uninterrupted supply chains for critical fertility medications, ensuring that patients receive timely access to necessary treatments.
- Scalability and Environmental Compliance: The method is inherently designed for industrial scale-up, avoiding the safety pitfalls of explosive perchlorates that limit batch sizes in traditional processes. The use of volatile organic compounds is optimized through efficient concentration and recycling steps, aligning with modern green chemistry principles and environmental regulations. Waste streams are simpler to treat due to the absence of heavy metals or complex inorganic salts, reducing the environmental footprint of the manufacturing facility. This compliance with stringent environmental standards future-proofs the supply chain against tightening global regulations, ensuring long-term sustainability and operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and sourcing of high-purity ganirelix acetate. These insights are derived directly from the patented methodology, focusing on the practical implications for pharmaceutical development and manufacturing. Understanding these details helps stakeholders make informed decisions about integrating this advanced purification technology into their supply networks.
Q: How does this method eliminate trifluoroacetic acid (TFA) residues?
A: The process utilizes a dedicated acetic acid and acetonitrile mobile phase system throughout both chromatographic stages, completely avoiding the introduction of TFA during the salt conversion and purification steps, thereby ensuring zero TFA residue in the final API.
Q: What purity levels can be achieved with this dual-stage chromatography?
A: By optimizing the gradient elution ratios in two distinct steps, the method consistently achieves a final purity of 99.82% with maximum single impurity content controlled below 0.04%, meeting stringent pharmacopeial standards.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method avoids unstable and explosive reagents like sodium perchlorate, using only common organic solvents and acids, which simplifies waste treatment and enhances operational safety for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ganirelix Acetate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthesis routes to meet the evolving demands of the global pharmaceutical industry. Our team of experts possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the dual-stage chromatographic purification of ganirelix acetate are seamlessly translated into industrial reality. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that validate every batch against the highest international standards. Our capability to handle complex peptide intermediates positions us as a strategic partner for companies seeking to enhance the quality and reliability of their fertility treatment portfolios.
We invite you to collaborate with us to explore how this optimized synthesis route can benefit your specific project requirements. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating the tangible economic advantages of switching to this superior purification method. Please contact us to request specific COA data and route feasibility assessments, and let us help you secure a stable, high-quality supply of ganirelix acetate for your commercial operations.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
