Advanced Synthesis of FBDT-IC Non-Fullerene Acceptor for High-Efficiency Organic Photovoltaics
Advanced Synthesis of FBDT-IC Non-Fullerene Acceptor for High-Efficiency Organic Photovoltaics
The rapid evolution of organic photovoltaics (OPV) demands electron acceptor materials that balance high efficiency with manufacturability. Patent CN111333665B introduces a breakthrough conjugated small molecule electron acceptor, FBDT-IC, which utilizes a novel hepta-condensed ring unit based on benzo[1,2-b:5,4-b']dithiophene (aBDT) as its core. This A-D-A structured material addresses critical limitations in current non-fullerene acceptor technologies, specifically targeting the difficulties in material purification and energy level regulation found in earlier generations. The disclosed synthesis route is remarkably efficient, enabling the rapid construction of complex fused-ring systems through a logical sequence of palladium-catalyzed couplings and condensations. By achieving a photoelectric conversion efficiency of 8.63% in device testing, this material validates the potential of aBDT-based cores in next-generation solar applications. For industrial partners, this patent represents a significant opportunity to access high-performance electronic chemicals through a scalable and robust chemical pathway.
The strategic implementation of this synthesis allows for precise control over the molecular architecture, ensuring consistent batch-to-batch quality essential for commercial device fabrication. The integration of electron-withdrawing end groups via Knoevenagel condensation further tunes the LUMO energy level to -4.00 eV, facilitating effective charge transfer when paired with common polymer donors. As a reliable electronic chemical supplier, understanding the nuances of this synthetic pathway is crucial for scaling production to meet the growing demand for sustainable energy materials. The following analysis dissects the technical merits and commercial viability of this patented process.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of organic solar cells has been heavily reliant on fullerene derivatives as electron acceptors, which present inherent challenges in large-scale manufacturing and performance optimization. Traditional fullerene materials often suffer from weak light absorption in the visible region, limiting the overall photon harvesting capability of the active layer. Furthermore, the purification of fullerene derivatives is notoriously difficult and costly, requiring extensive chromatographic separation to remove isomers and impurities that can act as charge traps within the device. The energy levels of fullerenes are also relatively fixed, offering limited tunability for matching with diverse polymer donors, which restricts the design space for high-efficiency tandem or multi-junction cells. These factors collectively contribute to higher production costs and lower power conversion efficiencies compared to emerging non-fullerene alternatives. Consequently, the industry has been actively seeking fused-ring electron acceptors that offer superior solubility, film-forming properties, and adjustable electronic characteristics.
The Novel Approach
The methodology described in CN111333665B overcomes these barriers by employing a benzo[1,2-b:5,4-b']dithiophene (aBDT) core to construct a planar, extended pi-conjugated system that enhances charge carrier mobility. Unlike conventional methods that might rely on complex multi-step functionalizations of unstable intermediates, this novel approach utilizes a modular building block strategy starting from commercially available thiophene and benzene derivatives. The synthesis leverages robust carbon-carbon bond-forming reactions, such as the Stille coupling, to assemble the central scaffold with high fidelity. This results in a material with excellent solubility in common organic solvents like chloroform and dichloromethane, simplifying the solution-processing steps required for thin-film deposition. Moreover, the A-D-A architecture allows for fine-tuning of the bandgap to approximately 1.7-1.8 eV, ensuring strong absorption overlap with the solar spectrum. This strategic design not only improves device performance but also streamlines the manufacturing workflow, making it highly attractive for cost reduction in electronic chemical manufacturing.
Mechanistic Insights into Stille Coupling and Knoevenagel Condensation
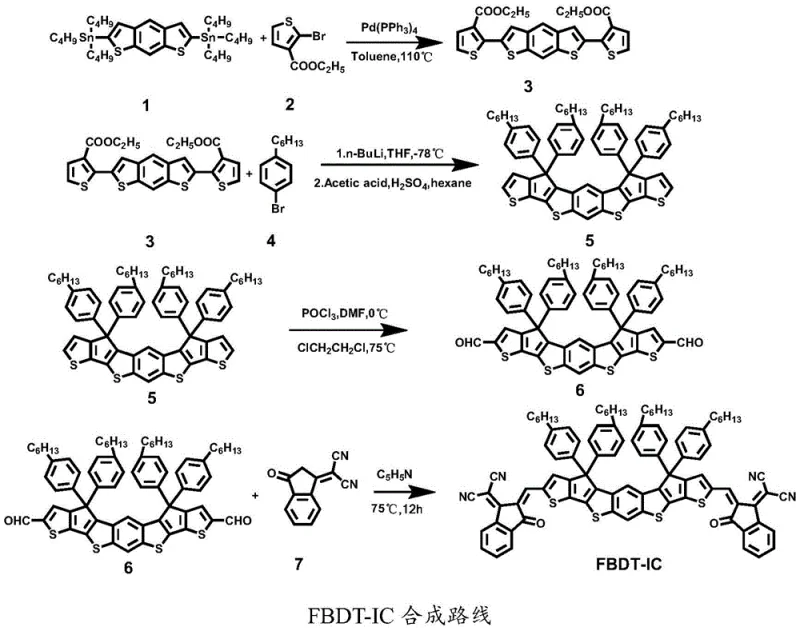
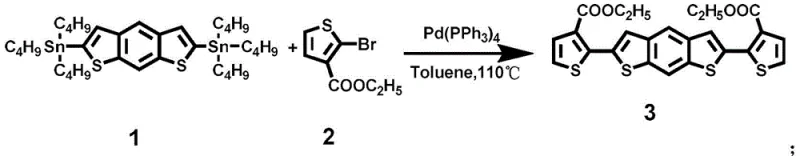
The cornerstone of the FBDT-IC synthesis is the initial construction of the thiophene-substituted intermediate via a palladium-catalyzed Stille coupling reaction. In this critical step, a distannylated benzo[1,2-b:4,5-b']dithiophene derivative reacts with a brominated thiophene ester under inert atmosphere conditions. The use of tetrakis(triphenylphosphine)palladium(0) as the catalyst facilitates the oxidative addition and transmetallation cycles necessary for forming the C-C bonds between the aromatic rings. Maintaining a strict nitrogen environment is paramount to prevent the oxidation of the phosphine ligands and the palladium center, which would otherwise lead to catalyst deactivation and the formation of homocoupling byproducts. The reaction is conducted in toluene at elevated temperatures around 110°C to ensure sufficient kinetic energy for the coupling to proceed to completion. Following the reaction, rigorous purification via column chromatography is employed to remove tin residues and unreacted starting materials, ensuring the high purity required for subsequent steps. This meticulous control over the coupling efficiency directly impacts the yield of the core scaffold, which is vital for the overall economic feasibility of the process.
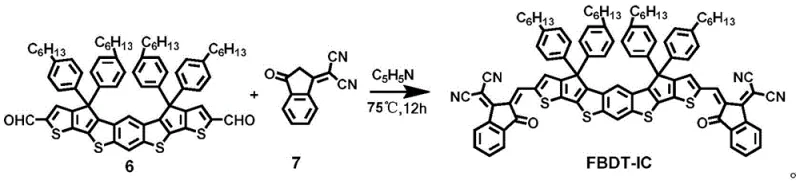
The final transformation involves a Knoevenagel condensation reaction, which installs the electron-deficient 1,1-dicyanomethylene-3-indanone (IC) end groups onto the dialdehyde precursor. This step is catalyzed by a weak base such as pyridine or beta-alanine in a mixed solvent system of chloroform and ethanol. The mechanism proceeds through the deprotonation of the active methylene group on the IC unit, generating a nucleophilic carbanion that attacks the aldehyde carbonyl carbon of the core. Subsequent dehydration leads to the formation of the conjugated double bond, extending the pi-system and lowering the LUMO energy level. The choice of base and solvent ratio is critical; too strong a base can lead to side reactions or decomposition of the sensitive aldehyde groups, while improper solvent polarity can affect the solubility of the growing oligomer. The reaction is typically heated to 75°C for 12 hours to drive the equilibrium towards the product. The resulting FBDT-IC precipitates as a black-purple solid, which is then purified to remove any unreacted end-capping agent. This final condensation step is crucial for defining the optoelectronic properties of the material, as the strength of the electron-withdrawing end groups dictates the open-circuit voltage of the resulting solar cell device.
How to Synthesize FBDT-IC Efficiently
The synthesis of FBDT-IC is designed to be operationally simple while maintaining high chemical precision, making it suitable for scale-up in a GMP-compliant facility. The process begins with the assembly of the central fused-ring core, followed by formylation and final end-capping. Each step has been optimized to maximize yield and minimize impurity formation, utilizing standard laboratory equipment and readily available reagents. The detailed standardized synthesis steps see the guide below, which outlines the specific molar ratios, temperature profiles, and workup procedures required to achieve the reported 8.63% efficiency metrics. Adhering to these parameters ensures the reproducibility of the material's optical and electronic properties.
- Perform Stille coupling between compound 1 and compound 2 using Pd(PPh3)4 catalyst in toluene at 110°C to yield compound 3.
- Execute nucleophilic addition of compound 3 with compound 4 using n-BuLi at -78°C, followed by acid-catalyzed cyclization to form the hepta-fused ring core (compound 5).
- Conduct Vilsmeier-Haack formylation on compound 5 using POCl3 and DMF to introduce aldehyde groups, yielding compound 6.
- Finalize synthesis via Knoevenagel condensation between compound 6 and compound 7 using a base catalyst to produce the target FBDT-IC material.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the FBDT-IC synthesis route offers distinct logistical and financial benefits over traditional fullerene-based supply chains. The reliance on commodity chemicals such as toluene, THF, and DMF, combined with widely available catalysts like palladium tetrakis, reduces the risk of raw material shortages that often plague specialty chemical markets. Furthermore, the elimination of complex purification protocols associated with fullerene derivatives significantly lowers the operational expenditure related to solvent consumption and waste disposal. The robustness of the synthetic steps implies a higher tolerance for minor variations in reaction conditions, which translates to more consistent batch yields and reduced production downtime. This stability is essential for maintaining a continuous supply of high-purity electronic chemicals to downstream device manufacturers.
- Cost Reduction in Manufacturing: The streamlined four-step synthesis eliminates the need for expensive and difficult-to-source fullerene precursors, replacing them with cost-effective thiophene and benzene derivatives. By avoiding the intricate separation processes required for fullerene isomers, the overall processing time and solvent usage are drastically reduced, leading to substantial cost savings per kilogram of produced material. Additionally, the high yields reported in the patent examples suggest that raw material utilization is efficient, minimizing waste generation and further driving down the cost of goods sold. This economic efficiency makes FBDT-IC a competitive alternative for large-area organic photovoltaic modules where material cost is a primary driver.
- Enhanced Supply Chain Reliability: The synthetic pathway relies on stable intermediates that can be stockpiled without significant degradation, providing a buffer against supply chain disruptions. The use of standard organic solvents and common catalysts ensures that sourcing can be diversified across multiple global suppliers, reducing dependency on single-source vendors. This flexibility allows for better negotiation leverage and more secure long-term supply contracts. Moreover, the scalability of the reaction conditions from gram to kilogram scale has been demonstrated through the consistent results across multiple embodiments in the patent, assuring buyers of the technology's readiness for commercial volume production.
- Scalability and Environmental Compliance: The process operates at moderate temperatures and pressures, reducing the energy footprint associated with heating and cooling large reactors. The solvents used, such as toluene and dichloromethane, are well-understood in industrial settings with established recovery and recycling protocols, facilitating compliance with environmental regulations. The absence of heavy metal contaminants in the final product, ensured by rigorous chromatographic purification, simplifies the waste treatment process and aligns with green chemistry principles. This environmental compatibility is increasingly important for meeting the sustainability criteria of international electronics manufacturers and regulatory bodies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the FBDT-IC material and its synthesis. These answers are derived directly from the experimental data and claims presented in patent CN111333665B, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the material's suitability for their specific application requirements and integration strategies.
Q: What is the photoelectric conversion efficiency of FBDT-IC based solar cells?
A: According to patent CN111333665B, organic solar cell devices utilizing FBDT-IC as the acceptor material demonstrate a photoelectric conversion efficiency of 8.63%, with an open-circuit voltage of 0.98V.
Q: How does the FBDT-IC synthesis method reduce production complexity?
A: The patented method utilizes a streamlined four-step sequence involving standard reactions like Stille coupling and Knoevenagel condensation, avoiding the complex purification difficulties often associated with traditional fullerene-based acceptors.
Q: What are the optical properties of the FBDT-IC material?
A: FBDT-IC exhibits strong visible light absorption in the 550-680 nm range with a maximum absorption peak at 660 nm, complementing polymer donors like PBDB-T for broader spectral coverage.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable FBDT-IC Supplier
As the demand for high-efficiency non-fullerene acceptors grows, partnering with an experienced CDMO is essential for translating laboratory success into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including trace metal analysis and thermal stability testing, which are critical for the performance of organic solar cells. We understand the nuances of handling air-sensitive intermediates and optimizing crystallization processes to deliver materials that consistently meet the high standards required by the optoelectronics industry.
We invite you to contact our technical procurement team to discuss how we can support your project with a Customized Cost-Saving Analysis tailored to your volume requirements. By leveraging our expertise in process optimization, we can help you secure specific COA data and route feasibility assessments that validate the economic viability of FBDT-IC for your specific device architecture. Let us collaborate to accelerate the deployment of next-generation solar technologies through reliable supply and technical excellence.
