Scalable Synthesis and Commercial Supply of High-Efficiency FBDT-IC Electron Acceptors
Scalable Synthesis and Commercial Supply of High-Efficiency FBDT-IC Electron Acceptors
The rapid evolution of organic photovoltaics (OPV) has necessitated the development of novel non-fullerene acceptor materials that offer superior tunability and processing characteristics compared to traditional fullerene derivatives. Patent CN111333665B discloses a breakthrough conjugated small molecule electron acceptor material, designated as FBDT-IC, which features an A-D-A (Acceptor-Donor-Acceptor) architecture centered around a hepta-fused ring unit of benzo[1,2-b:5,4-b']dithiophene (aBDT). This material represents a significant advancement in the field of electronic chemical manufacturing, providing a robust platform for high-performance organic solar cells. The molecular design strategically incorporates electron-withdrawing end groups to optimize energy levels, resulting in a moderate optical band gap of 1.7-1.8 eV and excellent film-forming properties. As a reliable electronic chemical supplier, understanding the structural nuances of such materials is critical for ensuring batch-to-batch consistency and performance reliability in downstream device fabrication.
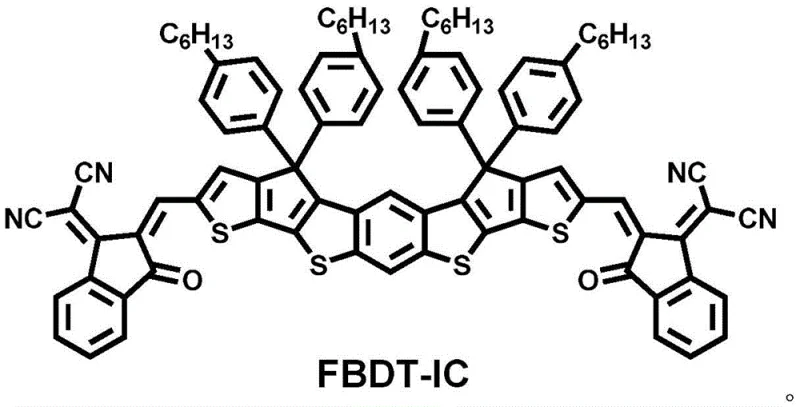
The structural integrity of FBDT-IC is paramount for its function as an electron transporter within the bulk heterojunction active layer. The core aBDT unit provides a rigid, planar backbone that facilitates efficient charge transport, while the peripheral alkyl chains ensure sufficient solubility in common organic solvents for solution processing. This balance between crystallinity and solubility is often a bottleneck in the commercial scale-up of complex conjugated small molecules, yet the design outlined in the patent addresses this by utilizing hexyl side chains that prevent excessive aggregation without compromising electronic coupling. For procurement managers, this translates to a material that is not only high-performing but also manageable within standard coating and printing workflows used in roll-to-roll manufacturing environments.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the domain of organic solar cells was dominated by fullerene-based acceptors, such as PCBM, which suffered from inherent limitations including weak absorption in the visible region, difficult purification processes, and limited tunability of energy levels. The synthesis of high-purity fullerene derivatives often involves harsh conditions and complex separation techniques, leading to elevated production costs and inconsistent impurity profiles that can degrade device longevity. Furthermore, the fixed electronic properties of fullerenes restrict the ability to match energy levels with emerging polymer donors, thereby capping the theoretical efficiency limits of the devices. These factors collectively hindered the widespread adoption of OPVs in cost-sensitive applications, creating a demand for alternative acceptor materials that could be synthesized through more controllable and economically viable pathways.
The Novel Approach
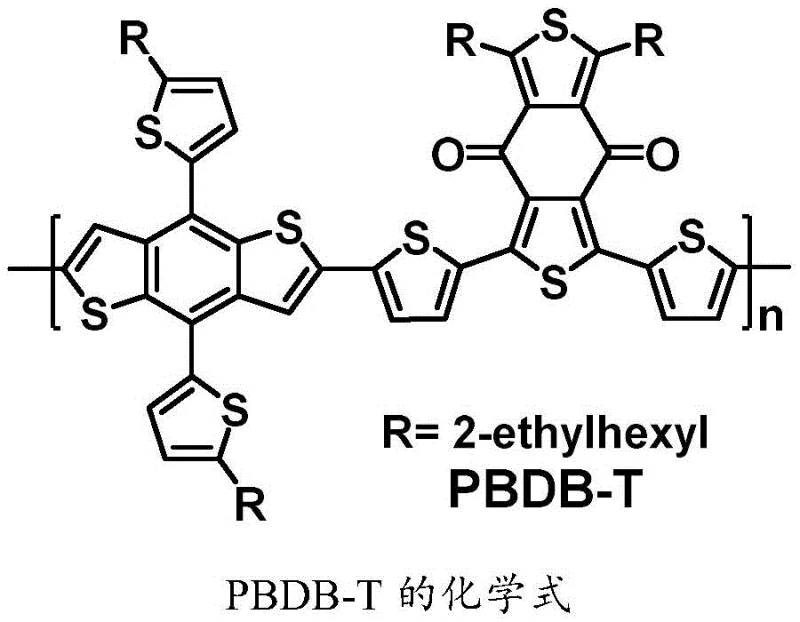
The synthetic strategy presented in patent CN111333665B offers a transformative solution by employing a modular A-D-A design that allows for precise tuning of optoelectronic properties. Unlike the rigid and isotropic nature of fullerenes, the FBDT-IC molecule is engineered to have complementary spectral absorption with common polymer donors like PBDB-T, as illustrated by the donor structure compatibility. This novel approach leverages a convergent synthesis where the central fused-ring core is constructed first, followed by the attachment of end groups, allowing for easier purification of intermediates and better control over the final product quality. By shifting away from fullerene chemistry, manufacturers can achieve cost reduction in organic solar cell material manufacturing through the use of abundant petrochemical feedstocks and established coupling reactions, significantly lowering the barrier to entry for large-area module production.
Mechanistic Insights into Pd-Catalyzed Coupling and Acid-Cyclization
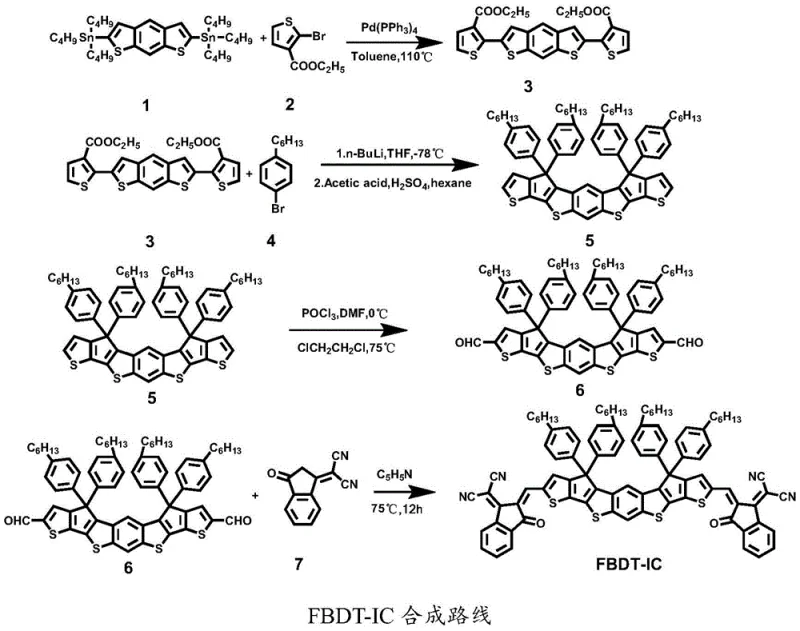
The construction of the FBDT-IC backbone relies heavily on a sequence of well-understood yet highly optimized organometallic transformations, beginning with a palladium-catalyzed Stille cross-coupling reaction. In this critical step, a distannylated benzo[1,2-b:4,5-b']dithiophene derivative reacts with a brominated thiophene ester in the presence of tetrakis(triphenylphosphine)palladium(0) at elevated temperatures (110°C) in toluene. This reaction forms the carbon-carbon bonds necessary to extend the conjugation length, which is essential for narrowing the bandgap and enhancing charge mobility. The choice of catalyst and ligand system is crucial here, as it ensures high turnover numbers and minimizes the formation of homocoupling byproducts, which are common impurities that can act as charge traps in the final semiconductor film. Following this, the synthesis proceeds through a lithiation-mediated cyclization, where n-butyllithium generates a reactive nucleophile that attacks the ester groups, followed by an acid-catalyzed dehydration and aromatization step using a mixture of acetic acid and sulfuric acid to fuse the rings into the rigid aBDT core.
Subsequent functionalization involves a Vilsmeier-Haack formylation to introduce aldehyde handles at the terminal positions of the fused core, which are then subjected to a Knoevenagel condensation with an electron-deficient indanone derivative. This final condensation step extends the conjugation further and installs the strong electron-withdrawing cyano and carbonyl groups that define the 'A' blocks of the A-D-A structure. The entire pathway is designed to minimize side reactions; for instance, the use of mild bases like piperidine or beta-alanine in the final step prevents the hydrolysis of sensitive functional groups while driving the equilibrium towards the desired alkene product. Understanding these mechanistic details allows our technical team to rigorously monitor critical process parameters, such as temperature gradients and reagent stoichiometry, ensuring that the resulting high-purity non-fullerene acceptor meets the stringent specifications required for high-efficiency photovoltaic applications.
How to Synthesize FBDT-IC Efficiently
The preparation of FBDT-IC follows a logical four-step sequence that balances yield with purity, starting from commercially available thiophene and benzodithiophene precursors. The process begins with the assembly of the ester-functionalized intermediate, followed by the rigorous ring-closing steps to form the hepta-fused core, and concludes with the installation of the end-capping groups. Each stage requires careful control of atmospheric conditions, typically under nitrogen protection, to prevent oxidation of sensitive organometallic intermediates. The detailed standardized synthesis steps, including specific molar ratios, solvent choices, and workup procedures, are outlined below to guide process engineers in replicating this high-value material.
- Perform Stille coupling between compound 1 and compound 2 using Pd(PPh3)4 in toluene at 110°C to yield compound 3.
- Execute nucleophilic addition with n-BuLi followed by acid-catalyzed cyclization using acetic acid and sulfuric acid to form the fused ring compound 5.
- Conduct Vilsmeier-Haack formylation on compound 5 using POCl3 and DMF in 1,2-dichloroethane to introduce aldehyde groups, yielding compound 6.
- Finalize the synthesis via Knoevenagel condensation between compound 6 and compound 7 using a base catalyst like piperidine to obtain FBDT-IC.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the synthesis of FBDT-IC offers distinct advantages over legacy acceptor materials due to its reliance on scalable organic synthesis techniques rather than exotic or scarce resources. The raw materials, such as thiophene derivatives and simple alkyl halides, are commodity chemicals with stable global supply chains, reducing the risk of procurement bottlenecks. Furthermore, the reaction conditions, while requiring precision, do not necessitate extreme pressures or cryogenic temperatures beyond standard laboratory capabilities, making the transition from gram-scale research to kilogram-scale pilot production significantly smoother. This accessibility translates directly into enhanced supply chain reliability, as manufacturers are not dependent on single-source suppliers for specialized precursors, thereby mitigating the risk of production stoppages.
- Cost Reduction in Manufacturing: The streamlined four-step synthetic route eliminates the need for complex multi-step functionalizations often required in fullerene processing, leading to substantial cost savings in terms of labor, energy, and solvent consumption. By avoiding the use of expensive transition metals beyond the catalytic amounts of palladium, and utilizing recyclable solvents like toluene and dichloromethane, the overall cost of goods sold (COGS) is drastically optimized. Additionally, the high yields reported in the patent examples for key steps, such as the formylation and condensation reactions, minimize material waste and reduce the burden on downstream purification processes, further driving down the unit cost of the final active material.
- Enhanced Supply Chain Reliability: The use of robust chemical transformations like Stille coupling and Knoevenagel condensation ensures that the manufacturing process is resilient to minor fluctuations in reaction conditions, which is vital for maintaining consistent output in a commercial setting. Since the intermediates are stable solids that can be isolated and stored, production can be decoupled into distinct campaigns, allowing for flexible inventory management and just-in-time delivery models. This modularity means that reducing lead time for high-purity OSC materials is achievable by parallelizing the synthesis of different intermediates, ensuring that customer demands for rapid prototyping and volume scaling are met without compromising quality.
- Scalability and Environmental Compliance: The synthetic pathway avoids the generation of highly toxic byproducts associated with some inorganic semiconductor processes, aligning with modern green chemistry principles and simplifying waste treatment protocols. The purification methods described, primarily involving column chromatography and recrystallization, are readily adaptable to industrial-scale simulated moving bed (SMB) chromatography or continuous crystallization units. This scalability ensures that the commercial scale-up of complex conjugated small molecules can be achieved with minimal environmental footprint, satisfying the increasingly strict regulatory requirements for electronic chemical manufacturing facilities globally.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the FBDT-IC material, derived directly from the experimental data and beneficial effects described in the patent documentation. These insights are intended to clarify the material's performance metrics and processing advantages for potential partners evaluating its integration into their photovoltaic architectures.
Q: What is the photoelectric conversion efficiency of FBDT-IC based solar cells?
A: According to patent CN111333665B, organic solar cells utilizing FBDT-IC as the acceptor material demonstrate a photoelectric conversion efficiency of 8.63%, with a high open-circuit voltage of 0.98V.
Q: What are the optical absorption properties of the FBDT-IC material?
A: FBDT-IC exhibits strong visible light absorption in the 550-680 nm range, with a maximum absorption peak at 660 nm, complementing polymer donors like PBDB-T effectively.
Q: Is the synthesis of FBDT-IC suitable for large-scale production?
A: Yes, the patented method utilizes standard organic reactions such as Stille coupling and Knoevenagel condensation with commercially available reagents, facilitating scalable manufacturing and cost reduction.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable FBDT-IC Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of non-fullerene acceptors like FBDT-IC in driving the next generation of renewable energy technologies. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab-scale discovery to market-ready products is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including trace metal analysis and isomer separation, which are critical for maximizing the power conversion efficiency of organic solar cells. We are committed to delivering high-purity FBDT-IC that meets the exacting standards of the optoelectronics industry.
We invite you to collaborate with us to optimize your supply chain and accelerate your product development cycles. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing expertise can support your strategic goals in the rapidly evolving field of organic photovoltaics.
