Scalable Convergent Synthesis of Iguratimod Intermediates for Commercial API Production
Scalable Convergent Synthesis of Iguratimod Intermediates for Commercial API Production
The pharmaceutical industry is constantly seeking robust, scalable, and cost-effective pathways for the production of novel immunosuppressants, and the recent disclosure in patent CN111087356B offers a transformative approach to synthesizing Iguratimod and its hydrochloride salt. This intellectual property details a novel convergent synthesis strategy that fundamentally departs from traditional linear methodologies, addressing critical pain points such as low overall yields, excessive solvent consumption, and complex purification requirements. By leveraging a palladium-catalyzed Suzuki-Miyaura coupling as the cornerstone step, this method connects two advanced intermediates to construct the biphenyl core with exceptional efficiency. For R&D directors and process chemists, this represents a significant opportunity to optimize the manufacturing of S1P1 receptor modulators, ensuring higher purity profiles and reduced operational complexity. The technical breakthrough lies not just in the chemistry itself, but in the strategic disconnection of the molecule, allowing for parallel synthesis of key fragments which dramatically shortens the critical path to the final active pharmaceutical ingredient.
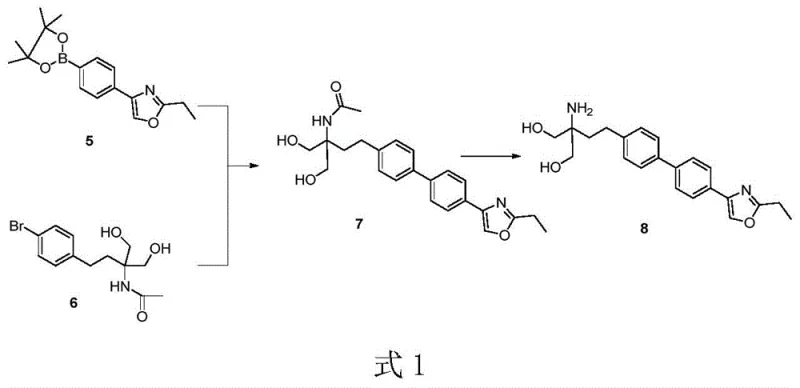
For procurement managers and supply chain heads, the implications of adopting this technology are profound, particularly regarding the reliability of the pharmaceutical intermediate supplier network. Traditional routes often suffer from bottlenecks where a single low-yielding step can jeopardize the entire batch, leading to unpredictable lead times and inflated costs. In contrast, the convergent approach described in this patent mitigates these risks by modularizing the synthesis. The ability to produce key building blocks independently and couple them in a high-yielding final step provides a buffer against supply disruptions. Furthermore, the patent explicitly highlights the reduction in "three wastes" (wastewater, waste gas, and solid waste), which is a crucial metric for modern cost reduction in API manufacturing. By minimizing the reliance on hazardous reagents and energy-intensive purification techniques like column chromatography, manufacturers can achieve a more sustainable and economically viable production model that aligns with increasingly stringent global environmental regulations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of Iguratimod has relied on linear synthetic routes that start from biphenyl and proceed through a tedious sequence of nine distinct reaction steps. These traditional pathways are plagued by inherent inefficiencies, including a dismal total yield of approximately 14.4%, which necessitates the processing of large volumes of starting materials to obtain modest amounts of the final product. A major operational burden in these legacy methods is the extensive use of column chromatography for purification at multiple stages, a technique that is notoriously difficult to scale and generates massive quantities of silica gel waste. Additionally, the conventional process requires repeated applications of strong Lewis acids and harsh reaction conditions, leading to significant safety hazards and complicated downstream processing. The cumulative effect of these factors is a high cost of goods sold (COGS) and a fragile supply chain that struggles to meet the demands of commercial-scale production, making it an unattractive option for large-scale commercial scale-up of complex immunosuppressants.
The Novel Approach
The innovative strategy presented in the patent overturns these limitations by employing a convergent synthesis that effectively halves the longest linear sequence and boosts the overall yield to an impressive 35.7%. Instead of building the molecule step-by-step from one end, this method synthesizes two sophisticated fragments—an oxazole-containing boronate ester and a functionalized amino-diol bromide—separately before joining them. This disconnection allows for the optimization of each fragment's synthesis in isolation, ensuring that impurities are managed early in the process. Crucially, the final coupling step utilizes a palladium catalyst in environmentally benign solvents like water or ethanol-water mixtures, eliminating the need for toxic organic solvents often required in older methods. The elimination of column chromatography throughout the entire route is perhaps the most significant operational improvement, replacing it with simple crystallization and filtration steps that are ideal for industrial reactors. This shift not only enhances the purity of the high-purity Iguratimod intermediate but also drastically simplifies the workflow, making it highly amenable to automation and continuous manufacturing processes.
Mechanistic Insights into Pd-Catalyzed Suzuki Coupling
At the heart of this new synthesis is the Suzuki-Miyaura cross-coupling reaction between intermediate 5 (a pinacol boronate) and intermediate 6 (an aryl bromide). The mechanistic elegance of this transformation lies in the catalytic cycle facilitated by palladium complexes, such as the preferred dibromo-bis(tri-tert-butylphosphine)dipalladium(I). The reaction initiates with the oxidative addition of the aryl bromide to the Pd(0) species, forming an aryl-palladium(II) complex. This is followed by transmetallation with the organoboron species, activated by a base such as potassium carbonate, which transfers the oxazole-containing aryl group to the palladium center. The cycle concludes with reductive elimination, releasing the coupled biphenyl product and regenerating the active Pd(0) catalyst. The patent data indicates that the choice of ligand and base is critical; bulky phosphine ligands enhance the stability of the catalyst and facilitate the difficult oxidative addition step, while the use of inorganic bases in aqueous media promotes the formation of the reactive boronate "ate" complex necessary for transmetallation.
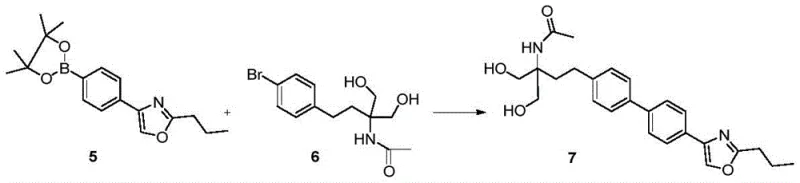
From an impurity control perspective, this mechanism offers distinct advantages over electrophilic aromatic substitution methods used in linear routes. The specificity of the palladium catalyst ensures that coupling occurs exclusively at the designated halide and boronate positions, minimizing the formation of regioisomers that are common in Friedel-Crafts reactions. Furthermore, the mild conditions of the Suzuki coupling (typically 70-90°C) prevent the degradation of sensitive functional groups, such as the acetamide and diol moieties present in intermediate 6. The patent emphasizes that the reaction can be driven to completion with high conversion rates, resulting in a crude product of sufficient purity to proceed directly to hydrolysis without intermediate chromatographic purification. This robustness is essential for reducing lead time for high-purity pharmaceutical intermediates, as it removes the variability associated with purification steps and ensures a consistent quality profile batch after batch, which is a key requirement for regulatory compliance in API manufacturing.
How to Synthesize Iguratimod Intermediates Efficiently
The practical execution of this synthesis involves three main phases: the preparation of the oxazole boronate fragment, the synthesis of the amino-diol bromide fragment, and their subsequent convergence. The first phase transforms simple bromobenzene into the heterocyclic building block through acylation, cyclization, and borylation, utilizing reagents like propionamide and bis(pinacolato)diboron. The second phase constructs the chiral-like amino-diol backbone, either from benzene via malonate chemistry or from p-bromophenethyl alcohol, involving key reduction steps with sodium borohydride to establish the necessary hydroxyl groups. The final phase brings these two complex pieces together in the presence of a palladium catalyst and base, followed by hydrolysis of the acetamide protecting group and salt formation to yield the final Iguratimod hydrochloride. The detailed standardized synthesis steps for implementing this route are outlined below.
- Synthesize the oxazole-boronate intermediate (Compound 5) starting from bromobenzene via Friedel-Crafts acylation, esterification, cyclization, and borylation.
- Prepare the amino-diol intermediate (Compound 6) starting from benzene or p-bromophenethyl alcohol via malonate coupling, reduction, and bromination.
- Perform the key Suzuki-Miyaura coupling between Compound 5 and Compound 6 using a palladium catalyst in aqueous or alcoholic media to form the biphenyl core.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement specialists and supply chain directors, the adoption of this convergent synthesis route offers tangible strategic benefits that extend beyond mere chemical yield. The primary advantage is the drastic simplification of the manufacturing process, which directly correlates to lower operational expenditures and reduced risk. By eliminating the need for column chromatography, the process removes a major bottleneck that typically requires expensive silica gel, large volumes of elution solvents, and significant labor hours for fraction collection and analysis. This streamlining allows for faster batch turnover and higher throughput in existing reactor infrastructure, effectively increasing capacity without capital investment. Moreover, the shift towards using water as a primary solvent for the coupling reaction significantly lowers the cost of solvent procurement and waste disposal, addressing both economic and environmental mandates simultaneously.
- Cost Reduction in Manufacturing: The transition from a linear to a convergent route fundamentally alters the cost structure of Iguratimod production. The patent data indicates a substantial increase in overall yield, moving from roughly 14% to over 35%, which effectively more than doubles the output per unit of raw material input. This efficiency gain is compounded by the reduction in Lewis acid usage; the new method consumes significantly less anhydrous aluminum chloride and other corrosive reagents compared to the traditional nine-step sequence. Consequently, the cost associated with raw material procurement, hazardous waste treatment, and equipment maintenance due to corrosion is markedly decreased, delivering a compelling return on investment for manufacturers adopting this technology.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the modular nature of the convergent synthesis. Since the two key intermediates can be manufactured in parallel or sourced from different suppliers, the risk of a single point of failure disrupting the entire production schedule is minimized. The use of commodity chemicals like bromobenzene, benzene, and diethyl acetamidomalonate as starting materials ensures that the supply base is broad and stable, reducing vulnerability to market fluctuations of exotic reagents. Additionally, the robustness of the palladium-catalyzed coupling step, which tolerates a variety of conditions and solvents, provides flexibility in sourcing and logistics, allowing manufacturers to adapt quickly to regional availability of materials without compromising the quality of the final pharmaceutical intermediate.
- Scalability and Environmental Compliance: Scaling chemical processes often exposes hidden inefficiencies, but this route is designed with scalability in mind. The replacement of chromatographic purification with crystallization and filtration makes the process inherently easier to scale from pilot plant to multi-ton production. Crystallization is a unit operation that scales linearly and predictably, unlike chromatography which becomes exponentially more difficult and costly at large scales. Furthermore, the significant reduction in "three wastes" aligns with global trends towards green chemistry and sustainability. By generating less hazardous waste and utilizing greener solvents, manufacturers can more easily comply with strict environmental regulations, avoiding potential fines and shutdowns, thereby ensuring long-term business continuity and a stronger corporate social responsibility profile.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Iguratimod synthesis pathway. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the feasibility and advantages of the method for potential adopters. Understanding these nuances is critical for making informed decisions about process development and technology transfer.
Q: What is the primary advantage of the convergent synthesis route for Iguratimod?
A: The convergent route significantly improves the total yield from 14.4% in traditional linear methods to 35.7%, while eliminating the need for column chromatography and reducing Lewis acid consumption.
Q: Which catalysts are preferred for the key coupling reaction?
A: The patent highlights dibromo-bis(tri-tert-butylphosphine)dipalladium(I) (Pd-Dimer) as the preferred catalyst, though Pd(dppf)Cl2 and Pd(OAc)2 are also effective under optimized conditions.
Q: How does this method address environmental concerns in API manufacturing?
A: By utilizing water or ethanol-water mixtures as solvents and drastically reducing the usage of anhydrous Lewis acids and silica gel for purification, the process minimizes hazardous waste generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Iguratimod Supplier
As the demand for novel immunosuppressants continues to grow, partnering with a technically proficient CDMO is essential for navigating the complexities of API production. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory bench to full-scale manufacturing. Our rigorous QC labs and commitment to stringent purity specifications guarantee that every batch of Iguratimod intermediate meets the highest international standards, providing you with the confidence needed to advance your clinical or commercial programs. We understand the critical importance of timeline and quality in the pharmaceutical sector and are equipped to handle the specific challenges of palladium-catalyzed reactions and sensitive functional group manipulations.
We invite you to engage with our technical procurement team to discuss how this optimized convergent route can be tailored to your specific production needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits specific to your volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise can drive efficiency and reliability in your supply chain for this vital therapeutic agent.
