Scalable Electrochemical Synthesis of 2-Substituted Benzoxazole Derivatives for Commercial API Production
The pharmaceutical and fine chemical industries are constantly seeking sustainable and efficient pathways to access complex heterocyclic scaffolds, particularly those serving as critical building blocks for bioactive molecules. A groundbreaking development in this domain is detailed in patent CN113818033A, which discloses a novel electrochemical synthesis method for 2-substituted benzoxazole derivatives. This technology represents a paradigm shift from traditional stoichiometric oxidation methods to a catalytic electrocatalytic dehydrogenation process. By utilizing electricity as the primary reagent, this method achieves the cyclization of o-hydroxyphenyl amine derivatives under mild conditions, producing hydrogen gas as the sole byproduct. This approach not only aligns with green chemistry principles but also offers substantial operational advantages for large-scale manufacturing. The core transformation involves the direct anodic oxidation of the substrate in an undivided cell, facilitating the formation of the benzoxazole ring without the need for external chemical oxidants or transition metal catalysts. 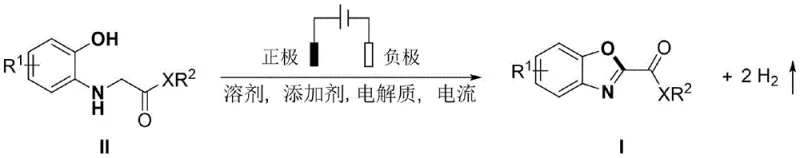
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-substituted benzoxazole formates and formamides has relied heavily on methodologies that present significant challenges for industrial scale-up and environmental compliance. Traditional routes often involve the condensation of o-aminophenol with benzaldehyde derivatives under the influence of strong oxidants, or the use of hazardous reagents such as thionyl chloride and benzoyl peroxide to effect cyclization dehydrogenation. These conventional processes frequently necessitate the use of transition metal catalysts, such as copper complexes, which introduce the risk of heavy metal contamination in the final product. Furthermore, the reliance on stoichiometric chemical oxidants generates substantial amounts of toxic waste, complicating disposal procedures and increasing the overall environmental footprint of the manufacturing process. The harsh reaction conditions often required, including high temperatures and aggressive reagents, can also lead to poor selectivity and the formation of difficult-to-remove impurities, thereby compromising the purity profile essential for pharmaceutical applications.
The Novel Approach
In stark contrast to these legacy methods, the electrochemical synthesis strategy outlined in the patent data offers a streamlined and environmentally benign alternative that directly addresses the aforementioned pain points. This novel approach utilizes an undivided electrolytic cell where the o-hydroxyphenyl amine derivative undergoes anodic oxidation to form the desired benzoxazole ring system. The elimination of metal catalysts and chemical oxidants means that the reaction mixture is inherently cleaner, reducing the burden on downstream purification steps such as chromatography or recrystallization. The process operates under mild conditions, typically between 20-60°C, which enhances safety and energy efficiency compared to high-temperature thermal methods. Moreover, the generation of hydrogen gas as the only byproduct signifies a zero-waste oxidation process, drastically reducing the cost and complexity associated with waste treatment. This method effectively transforms electrical energy into chemical potential, providing a precise and controllable means of driving the reaction forward with high atom economy.
Mechanistic Insights into Electrochemical Oxidative Cyclization
The mechanistic pathway of this electrochemical transformation is rooted in the anodic oxidation of the nitrogen and oxygen functionalities within the o-hydroxyphenyl amine substrate. Upon application of a constant current, the substrate loses electrons at the anode surface, generating reactive radical cation intermediates that facilitate the intramolecular nucleophilic attack of the phenolic oxygen onto the adjacent carbon center. This electron transfer process is mediated by the electrolyte system, which typically comprises salts like tetrabutylammonium tetrafluoroborate or tetrabutylammonium hexafluorophosphate to ensure sufficient conductivity in the organic solvent medium. The presence of acidic additives, such as acetic acid or p-toluenesulfonic acid, plays a crucial role in proton management, helping to stabilize intermediates and drive the equilibrium towards the cyclized product. Unlike chemical oxidants that may react non-selectively with various functional groups, the electrode potential can be tuned to selectively oxidize the specific moiety required for ring closure, thereby minimizing side reactions. This precise control over the oxidation state is a key factor in achieving high yields and selectivity, as evidenced by the successful synthesis of various derivatives with different electronic properties on the aromatic ring.
From an impurity control perspective, the absence of transition metals is a decisive advantage for the production of high-purity pharmaceutical intermediates. In traditional metal-catalyzed reactions, trace amounts of catalyst residues can persist through multiple purification steps, posing regulatory risks and potential toxicity issues in the final drug substance. The electrochemical method completely bypasses this issue, as the "catalyst" is essentially the electrode surface and the electric current, neither of which leaves residual contaminants in the product stream. Furthermore, the mild reaction conditions help to prevent the degradation of sensitive functional groups, such as esters or amides, which might otherwise hydrolyze or decompose under the harsh acidic or basic conditions often required by conventional cyclization protocols. The result is a cleaner crude reaction profile, which simplifies the isolation process and ensures that the final product meets stringent quality specifications with minimal effort. This inherent purity advantage translates directly into cost savings and reduced time-to-market for new drug candidates relying on this scaffold.
How to Synthesize Ethyl 2-Benzoxazole Carboxylate Efficiently
To implement this cutting-edge synthesis in a laboratory or pilot plant setting, operators must carefully optimize the electrochemical parameters to maximize yield and efficiency. The process begins with the preparation of the reaction mixture, where the o-hydroxyphenyl glycine derivative is dissolved in a suitable organic solvent such as acetonitrile along with the supporting electrolyte and an acidic additive. The choice of electrode material is also critical, with reticulated glassy carbon anodes and platinum cathodes demonstrating superior performance in terms of current efficiency and stability. Once the cell is assembled and the constant current is applied, the reaction proceeds via the oxidative dehydrogenation mechanism described previously, converting the linear precursor into the cyclic benzoxazole structure. Detailed standard operating procedures regarding specific molar ratios, current densities, and work-up protocols are essential for reproducible results.
- Prepare the undivided electrolytic cell by adding the o-hydroxyphenyl amine derivative substrate, electrolyte such as tetrabutylammonium tetrafluoroborate, acidic additive like acetic acid, and organic solvent.
- Insert the anode and cathode electrodes into the reaction solution and apply a constant current between 1-200 mA while maintaining the temperature between 10-100°C.
- Stir the reaction mixture for 1-48 hours under nitrogen protection, then perform column chromatography separation to isolate the pure 2-substituted benzoxazole derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this electrochemical technology presents a compelling value proposition centered on cost reduction and supply security. The elimination of expensive noble metal catalysts and hazardous oxidants directly lowers the raw material costs associated with the synthesis of these valuable intermediates. Additionally, the simplified work-up procedure, driven by the absence of metal residues and toxic byproducts, reduces the consumption of solvents and stationary phases required for purification, further driving down the cost of goods sold. From a supply chain reliability standpoint, the use of electricity as a reagent decouples the production process from the volatility of the chemical reagent market, ensuring a more stable and predictable manufacturing workflow. The scalability of electrochemical reactors is well-established, allowing for seamless transition from gram-scale optimization to multi-ton commercial production without the need for significant process re-engineering. This robustness ensures that supply commitments can be met consistently, even in the face of fluctuating demand from downstream pharmaceutical clients.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the drastic simplification of the reaction setup and the removal of costly reagents. By replacing stoichiometric oxidants and metal catalysts with electricity, the variable costs per kilogram of product are significantly reduced. Furthermore, the generation of hydrogen gas as a clean byproduct eliminates the need for expensive waste disposal services associated with heavy metal sludge or toxic organic waste streams. The energy efficiency of the process, operating at moderate temperatures and currents, also contributes to lower utility costs compared to energy-intensive thermal processes. These cumulative savings allow for a more competitive pricing structure for the final intermediate, enhancing the margin potential for both the manufacturer and the end-user.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of readily available and stable starting materials, such as o-hydroxyphenyl amine derivatives, which are commercially accessible from multiple sources. The electrochemical nature of the reaction reduces dependency on specialized reagents that may have long lead times or supply constraints. Moreover, the continuous flow potential of electrochemical synthesis offers opportunities for intensified production, enabling faster turnaround times and reduced inventory holding costs. This agility allows suppliers to respond more rapidly to urgent orders or changes in production schedules, thereby strengthening the partnership between the chemical supplier and the pharmaceutical developer. The consistent quality of the product, free from batch-to-batch variations caused by catalyst degradation, further reinforces trust and reliability in the supply relationship.
- Scalability and Environmental Compliance: Scaling this electrochemical process to industrial levels is facilitated by the modular nature of electrolytic cells, which can be stacked or paralleled to increase capacity without altering the fundamental reaction kinetics. This modularity supports flexible manufacturing strategies, allowing production volumes to be adjusted dynamically based on market demand. From an environmental compliance perspective, the process aligns perfectly with increasingly stringent global regulations regarding emissions and waste discharge. The absence of heavy metals and toxic oxidants simplifies the permitting process for new manufacturing facilities and reduces the liability associated with environmental incidents. This "green" credential is increasingly valued by major pharmaceutical companies who are committed to sustainability goals, making suppliers of such intermediates preferred partners in their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical synthesis route. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the technology's capabilities and limitations. Understanding these details is crucial for R&D teams evaluating the feasibility of this route for their specific pipeline projects.
Q: What are the primary advantages of this electrochemical method over traditional synthesis?
A: This method eliminates the need for expensive transition metal catalysts and harsh chemical oxidants, resulting in a cleaner process with hydrogen gas as the only byproduct, which significantly simplifies downstream purification and waste treatment.
Q: What is the substrate scope for this electrochemical cyclization?
A: The process demonstrates broad compatibility with various substituents on the aromatic ring, including alkyl, alkoxy, and halogen groups, as well as different ester and amide side chains, allowing for diverse structural modifications.
Q: How does this process impact impurity profiles in pharmaceutical intermediates?
A: By avoiding metal catalysts, the risk of heavy metal contamination is completely removed, ensuring high-purity specifications that meet stringent regulatory requirements for active pharmaceutical ingredient manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Substituted Benzoxazole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in modernizing the production of complex pharmaceutical intermediates. Our team of expert chemists and engineers possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods like the one described in CN113818033A can be successfully translated into robust industrial processes. We are equipped with state-of-the-art electrochemical reactors and rigorous QC labs capable of meeting stringent purity specifications required by global regulatory bodies. Our commitment to technological advancement allows us to offer clients not just a product, but a optimized manufacturing solution that balances cost, quality, and sustainability. By leveraging our expertise in green chemistry and process intensification, we help our partners accelerate their drug development timelines while minimizing their environmental impact.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific project needs. Whether you require clinical trial materials or commercial quantities of 2-substituted benzoxazole derivatives, our technical procurement team is ready to provide specific COA data and route feasibility assessments to support your decision-making. Partnering with us means gaining access to a reliable supply chain backed by deep technical knowledge and a dedication to excellence. Contact us today to discuss how our advanced electrochemical capabilities can enhance your production strategy and drive value for your organization.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
