Scalable Electrochemical Synthesis of 2-Substituted Benzoxazole Derivatives for Commercial API Production
Scalable Electrochemical Synthesis of 2-Substituted Benzoxazole Derivatives for Commercial API Production
The pharmaceutical industry is currently witnessing a paradigm shift towards greener, more sustainable synthetic methodologies, driven by the urgent need to reduce environmental footprints and lower production costs for complex active pharmaceutical ingredients (APIs). A groundbreaking development in this arena is detailed in Chinese Patent CN113818033B, which discloses a novel electrochemical synthesis method for 2-substituted benzoxazole derivatives. This technology represents a significant departure from traditional transition-metal catalyzed processes, utilizing electric current as a clean reagent to drive oxidative cyclization. By employing an undivided electrolytic cell with specific electrode configurations and electrolyte systems, this method achieves high-efficiency conversion of o-hydroxyphenyl amine derivatives into valuable benzoxazole scaffolds without the need for external chemical oxidants. The implications for industrial manufacturing are profound, offering a pathway to high-purity intermediates with simplified workup procedures and reduced hazardous waste generation.
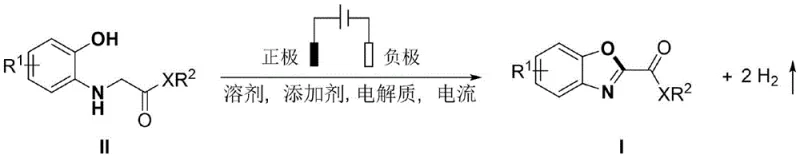
For R&D directors and process chemists, the ability to access these privileged heterocyclic structures through such a mild and atom-economical route opens new avenues for drug discovery and process optimization. The patent explicitly highlights the versatility of this approach, accommodating a wide range of substituents on the aromatic ring and various ester or amide side chains, which are critical features for tuning the pharmacological properties of the final drug molecule. As a reliable pharmaceutical intermediate supplier, understanding and adopting such cutting-edge electrochemical technologies allows us to offer clients not just a product, but a superior, future-proof supply chain solution that aligns with global sustainability goals while maintaining rigorous quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-substituted benzoxazole derivatives has relied heavily on traditional chemical oxidation methods that present significant challenges for large-scale manufacturing and environmental compliance. Conventional routes often involve the condensation of o-aminophenols with aldehydes or carboxylic acid derivatives under the influence of strong oxidants or transition metal catalysts, such as copper or iron salts. These methods frequently require harsh reaction conditions, including high temperatures and the use of corrosive reagents like thionyl chloride or benzoyl peroxide, which pose serious safety hazards and operational risks in a plant setting. Furthermore, the reliance on stoichiometric amounts of metal catalysts introduces a critical bottleneck in the purification process, necessitating expensive and time-consuming steps to remove trace heavy metals to meet stringent regulatory limits for pharmaceutical products. The generation of substantial amounts of chemical waste from these oxidants and catalysts also complicates waste treatment protocols, driving up the overall cost of production and negatively impacting the environmental profile of the manufacturing process.
The Novel Approach
In stark contrast, the electrochemical synthesis method described in the patent offers a transformative solution by replacing chemical oxidants with electrons, effectively turning electricity into a clean and tunable reagent. This novel approach operates under remarkably mild conditions, typically between 20°C and 60°C, using constant current electrolysis in an undivided cell to facilitate the oxidative dehydrogenation cyclization. The reaction system is elegantly simple, requiring only a suitable organic solvent, a supporting electrolyte like tetrabutylammonium tetrafluoroborate, and a proton source additive such as acetic acid to proceed efficiently. One of the most compelling advantages of this method is that the only byproduct generated is hydrogen gas, which can be easily vented or captured, thereby eliminating the formation of toxic or difficult-to-dispose-of chemical waste streams. This metal-free and oxidant-free strategy not only simplifies the downstream processing by removing the need for metal scavenging but also significantly enhances the safety and sustainability of the entire production lifecycle, making it an ideal candidate for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Electrochemical Oxidative Cyclization
The core of this innovative synthesis lies in the anodic oxidation mechanism, where the o-hydroxyphenyl amine derivative undergoes a controlled electron transfer at the electrode surface to initiate the cyclization process. In this electrocatalytic system, the substrate is oxidized at the anode, likely forming a radical cation intermediate that facilitates the intramolecular nucleophilic attack of the phenolic oxygen onto the adjacent carbonyl or imine carbon. The presence of the additive, such as acetic acid, plays a crucial role in modulating the proton concentration and stabilizing the reaction intermediates, ensuring that the electron transfer proceeds smoothly without competing side reactions that could lead to polymerization or decomposition. The choice of electrode material is also critical; the patent specifies the use of reticular glassy carbon or graphite felt as the anode and platinum as the cathode, a combination that provides a high surface area for efficient electron transfer while maintaining chemical stability under the applied potential. This precise control over the electrochemical environment allows for the selective formation of the benzoxazole ring with high fidelity, minimizing the formation of regioisomers or over-oxidized byproducts that often plague traditional thermal methods.
Furthermore, the scope of this electrochemical transformation is exceptionally broad, demonstrating remarkable tolerance to various functional groups which is essential for the synthesis of diverse drug candidates. For instance, the method successfully converts substrates with electron-donating groups, such as the tert-butyl group in the synthesis of ethyl 4-tert-butyl-2-benzoxazole carboxylate, achieving yields as high as 79% under optimized conditions. Similarly, substrates bearing electron-withdrawing groups like chlorine are also compatible, yielding products like ethyl 5-chloro-2-benzoxazole carboxylate with respectable efficiency, proving that the electronic nature of the aromatic ring does not severely inhibit the anodic oxidation process.
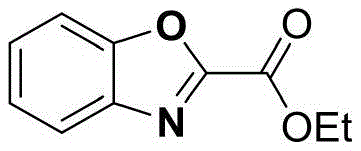
Beyond simple esters, the methodology extends seamlessly to amide derivatives, as evidenced by the successful synthesis of N-methyl-2-benzoxazole carboxamide and N,N-dimethyl variants. This versatility indicates that the mechanism is robust enough to handle different nucleophilicities and steric environments around the reaction center, providing medicinal chemists with a powerful tool for late-stage functionalization. The ability to access both ester and amide substituted benzoxazoles from similar glycine-derived precursors in a single electrochemical setup underscores the flexibility of this platform technology for generating diverse chemical libraries.
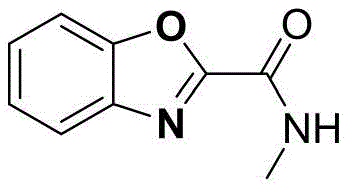
How to Synthesize Ethyl 2-Benzoxazole Carboxylate Efficiently
To implement this electrochemical synthesis in a laboratory or pilot plant setting, operators must adhere to specific parameters regarding current density, electrolyte concentration, and reaction time to maximize yield and purity. The process begins with the preparation of the reaction mixture in an undivided cell, where the precise molar ratios of the o-hydroxyphenyl amine derivative to the electrolyte and additive are critical for maintaining conductivity and reaction kinetics. Detailed standard operating procedures regarding electrode spacing, stirring rates, and temperature control are essential to ensure reproducibility and safety, particularly when scaling up from milligram to kilogram quantities. For a comprehensive guide on the exact experimental conditions and step-by-step execution of this synthesis, please refer to the standardized protocol below.
- Prepare the undivided electrolytic cell by adding the o-hydroxyphenyl amine derivative, electrolyte (e.g., tetrabutylammonium tetrafluoroborate), additive (e.g., acetic acid), and organic solvent.
- Insert the anode (e.g., reticular glassy carbon) and cathode (e.g., platinum) into the solution and apply a constant current of 2-50 mA.
- Stir the reaction mixture at 20-60°C for 1-48 hours under nitrogen protection, then isolate the product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this electrochemical synthesis method offers substantial strategic advantages for procurement managers and supply chain leaders who are constantly under pressure to reduce costs and mitigate supply risks. The elimination of expensive transition metal catalysts, such as palladium, copper, or rhodium, directly translates to a significant reduction in raw material costs, as these metals are subject to volatile market prices and geopolitical supply constraints. Moreover, the removal of these metals from the process flow eliminates the need for specialized metal scavenging resins and the associated validation testing required to prove low residual metal levels in the final API, thereby streamlining the quality control workflow and reducing the overall cycle time for batch release. This simplification of the manufacturing process also leads to a drastic reduction in the volume of hazardous chemical waste, lowering disposal costs and minimizing the environmental compliance burden on the manufacturing facility.
- Cost Reduction in Manufacturing: The primary driver for cost savings in this electrochemical route is the substitution of chemical reagents with electricity, which is generally cheaper and more consistent in price than specialized oxidants or catalysts. By avoiding the use of stoichiometric oxidants like benzoyl peroxide or thionyl chloride, the process inherently reduces the material bill of goods, while the mild reaction conditions lower energy consumption associated with heating and cooling cycles. Additionally, the simplified workup procedure, which avoids complex extraction and purification steps needed to remove metal residues, results in higher overall process efficiency and reduced labor costs per kilogram of product produced.
- Enhanced Supply Chain Reliability: Relying on electricity as the primary "reagent" decouples the production process from the supply chain volatility of critical raw materials, ensuring a more stable and predictable manufacturing schedule. The starting materials, such as o-hydroxyphenyl glycine derivatives, are readily available and do not require complex multi-step synthesis, further securing the upstream supply chain against disruptions. This robustness allows for better inventory management and shorter lead times for high-purity pharmaceutical intermediates, enabling our partners to respond more agilely to market demands and regulatory changes without the fear of raw material shortages.
- Scalability and Environmental Compliance: The electrochemical reactor design is inherently scalable, moving from small undivided cells in the lab to larger flow electrolysis reactors in production without significant changes to the reaction chemistry. This scalability ensures that the process can meet commercial demand volumes ranging from hundreds of kilograms to multi-ton annual production capacities while maintaining consistent product quality. Furthermore, the generation of hydrogen gas as the sole byproduct aligns perfectly with green chemistry principles, facilitating easier permitting and environmental impact assessments, which is increasingly becoming a prerequisite for doing business with top-tier global pharmaceutical companies committed to sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this electrochemical technology, derived directly from the patent specifications and practical application scenarios. Understanding these details helps stakeholders evaluate the feasibility of integrating this method into their existing supply chains and R&D pipelines. The answers provided reflect the specific advantages and operational parameters outlined in the intellectual property documentation.
Q: What are the primary advantages of this electrochemical method over traditional catalytic oxidation?
A: The primary advantage is the elimination of stoichiometric metal catalysts and harsh chemical oxidants. This method uses electricity as the reagent, producing only hydrogen gas as a byproduct, which significantly simplifies downstream purification and reduces heavy metal contamination risks in the final API.
Q: Can this synthesis method tolerate diverse functional groups on the aromatic ring?
A: Yes, the patent demonstrates excellent substrate scope. It successfully synthesizes derivatives with electron-donating groups like tert-butyl and methyl, as well as electron-withdrawing groups like chloro, indicating robustness for various pharmaceutical scaffold modifications.
Q: Is this process suitable for large-scale commercial manufacturing?
A: The process utilizes mild reaction conditions (20-60°C) and common solvents like acetonitrile, making it highly adaptable for scale-up. The absence of expensive metal catalysts further enhances its economic viability for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Substituted Benzoxazole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of electrochemical synthesis in modernizing the production of complex pharmaceutical intermediates like 2-substituted benzoxazole derivatives. Our team of expert process chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are equipped with state-of-the-art electrochemical reactors and stringent purity specifications managed by our rigorous QC labs, guaranteeing that every batch meets the highest international standards for impurity profiles and residual solvent content. Our commitment to technological advancement allows us to offer this greener, more cost-effective synthetic route to our global partners, positioning them for success in an increasingly competitive and regulated market.
We invite you to collaborate with us to leverage this advanced electrochemical technology for your next project, whether it involves custom synthesis or large-scale commercial manufacturing. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how switching to this metal-free electrochemical process can optimize your supply chain and enhance your product's market competitiveness.
