Advanced Solid-Phase Synthesis of Plecanatide: Enhancing Purity and Commercial Viability
The pharmaceutical industry continuously seeks robust manufacturing pathways for complex peptides like Plecanatide, a guanylate cyclase-C agonist used for treating chronic idiopathic constipation. Patent CN115991738A introduces a significant technological advancement in this domain by detailing a highly efficient solid-phase synthesis method that addresses the critical challenge of regioselective disulfide bond formation. Unlike traditional approaches that often struggle with mispaired cysteine residues, this invention employs a strategic combination of orthogonal protecting groups, specifically trimethoxyphenylthio (S-Tmp) and acetamidomethyl (Acm), to dictate the precise order of cyclization. The process begins with the assembly of the peptide backbone on a solid support, followed by a controlled on-resin cyclization to form the first disulfide bridge between Cys4 and Cys12. Subsequent cleavage and liquid-phase oxidation complete the structure, yielding a product with exceptional purity levels exceeding 99.5 percent. This technical breakthrough offers a compelling value proposition for reliable peptide suppliers aiming to streamline their production of gastrointestinal therapeutics while minimizing downstream purification burdens.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Plecanatide has been plagued by inefficiencies inherent in random oxidation strategies and complex hybrid liquid-solid phase couplings. Conventional methods often utilize identical protecting groups for all cysteine residues, relying on statistical oxidation to form the necessary disulfide bonds, which inevitably leads to a heterogeneous mixture of correctly paired and mismatched isomers. Furthermore, prior art such as CN108003222A attempted to mitigate this by combining liquid-phase hexapeptide synthesis with solid-phase elongation, but this introduced significant solubility issues and coupling inefficiencies as the fragment length increased. The reliance on harsh conditions for deprotection, such as repeated trifluoroacetic acid treatments for Mmt groups, frequently resulted in partial resin cleavage and the generation of truncated impurities that are notoriously difficult to separate. These technical bottlenecks not only compromise the final yield but also escalate the cost of goods due to the extensive chromatographic purification required to meet stringent regulatory specifications for single impurity content.
The Novel Approach
The methodology disclosed in CN115991738A fundamentally reengineers the synthesis workflow by implementing a stepwise, orthogonal protection strategy that decouples the formation of the two distinct disulfide bonds. By assigning the S-Tmp group to the Cys4 and Cys12 positions and the Acm group to Cys7 and Cys15, the process enables the selective formation of the first disulfide bridge directly on the solid support under mild reducing conditions. This on-resin cyclization step effectively locks in the correct conformation early in the synthesis, preventing the scrambling that occurs in solution-phase random oxidations. Following this, the peptide is cleaved from the resin, and the second disulfide bond is formed in the liquid phase using iodine-mediated deprotection of the Acm groups. This separation of cyclization events into distinct phases—solid for the first bond and liquid for the second—dramatically simplifies the impurity profile, ensuring that the crude product already possesses high purity before final polishing, thereby enhancing the overall feasibility of cost reduction in API manufacturing.
Mechanistic Insights into Orthogonal Disulfide Bond Formation
The core innovation of this synthesis lies in the chemical orthogonality between the S-Tmp and Acm protecting groups, which allows for independent manipulation of the cysteine side chains without cross-reactivity. The S-Tmp group is uniquely susceptible to nucleophilic attack by thiol reducing agents such as dithiothreitol (DTT) in the presence of a base like N-methylmorpholine, conditions that leave the acid-stable Acm group completely intact. Once the S-Tmp groups are removed on the resin, the resulting free thiols are immediately oxidized by N-chlorosuccinimide (NCS) to form the 4-12 disulfide bond. This intramolecular reaction is favored by the proximity of the residues on the solid support, driving the equilibrium towards the desired cyclic product. The stability of the Acm group during the subsequent acidic cleavage step (using TFA cocktails) is crucial, as it preserves the Cys7 and Cys15 thiols in a protected state until the peptide is fully liberated into solution. This precise temporal control over reactivity is what enables the high fidelity of the final structure.
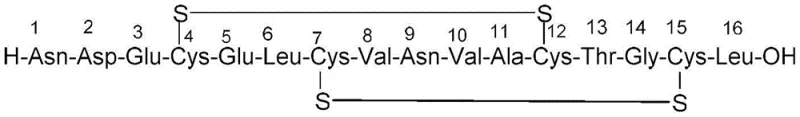
Following the resin cleavage, the linear peptide containing the first disulfide bond and protected Acm groups undergoes the second cyclization in the liquid phase. The use of iodine in acetic acid serves a dual purpose: it acts as an oxidant to remove the Acm groups and simultaneously facilitates the formation of the second disulfide bond between Cys7 and Cys15. The reaction conditions are carefully optimized, typically maintaining temperatures around 15 degrees Celsius initially to control the reaction rate and prevent over-oxidation or side reactions on sensitive residues like methionine or tryptophan, although Plecanatide does not contain these, the principle of gentle oxidation holds. Monitoring via UPLC ensures that the starting material is consumed to below 0.1 percent before quenching with ascorbic acid. This mechanistic precision ensures that the final product contains minimal mismatched isomers, directly addressing the primary purity concerns faced by R&D directors in the development of complex peptide therapeutics.
How to Synthesize Plecanatide Efficiently
The practical implementation of this synthesis route involves a series of well-defined operational steps that leverage standard solid-phase peptide synthesis (SPPS) equipment, making it accessible for most contract development and manufacturing organizations. The process initiates with the anchoring of the C-terminal amino acid to a Wang or CTC resin, followed by the sequential coupling of dipeptide fragments such as Fmoc-Thr(tBu)-Gly-OH and Fmoc-Cys(Acm)-Val-OH to accelerate chain elongation. Critical to the success of the protocol is the strict adherence to the orthogonal deprotection sequence, where the S-Tmp groups are targeted first on-resin before the global acid cleavage. Detailed standard operating procedures regarding reagent concentrations, reaction times, and washing protocols are essential to maintain the integrity of the growing peptide chain and ensure high coupling efficiencies at each step. For a comprehensive breakdown of the specific experimental parameters and stoichiometry required for each stage of this synthesis, please refer to the standardized guide below.
- Perform solid-phase synthesis using Wang or CTC resin, incorporating S-Tmp protected cysteine at positions 4 and 12, and Acm protected cysteine at positions 7 and 15.
- Deprotect S-Tmp groups on-resin using a thiol reducing agent like DTT, followed by cyclization with N-chlorosuccinimide (NCS) to form the first disulfide bond.
- Cleave the peptide from the resin using TFA cocktail, then perform liquid-phase deprotection of Acm groups using iodine in acetic acid to form the second disulfide bond.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, the adoption of this patented synthesis method offers substantial strategic benefits by mitigating risks associated with yield loss and purification bottlenecks. Traditional peptide synthesis often suffers from low crude purity, necessitating multiple rounds of preparative HPLC which consumes significant amounts of solvent and stationary phase, driving up both operational costs and environmental waste. By achieving a crude purity that is significantly higher through controlled cyclization, this process reduces the load on purification columns and shortens the overall production cycle time. Furthermore, the use of commercially available dipeptide fragments and standard resins ensures that raw material sourcing remains stable and不受 geopolitical constraints that might affect exotic reagents. This reliability is paramount for supply chain heads who must guarantee continuous availability of high-purity peptide intermediates to meet global demand without interruption.
- Cost Reduction in Manufacturing: The elimination of complex liquid-phase fragment couplings and the reduction in mismatched isomers directly translate to lower manufacturing costs. By avoiding the need for extensive recycling of off-spec material and minimizing the consumption of expensive chromatography media, the overall cost of goods sold is drastically simplified. The ability to perform the first cyclization on-resin also reduces the volume of solvents required for intermediate isolation, contributing to substantial cost savings in utility and waste disposal expenses.
- Enhanced Supply Chain Reliability: The reliance on robust, standard chemical reagents such as N-chlorosuccinimide and iodine, rather than specialized enzymes or unstable catalysts, enhances the resilience of the supply chain. This method reduces the lead time for high-purity peptide intermediates by streamlining the synthesis workflow, allowing for faster batch turnover. Procurement managers can benefit from a more predictable production schedule, as the process is less prone to the variability and failure rates associated with random oxidation techniques.
- Scalability and Environmental Compliance: The solid-phase nature of the initial steps facilitates easy scale-up from laboratory to commercial production scales, as the reaction kinetics are governed by the resin loading rather than solution concentration. Additionally, the reduced solvent usage and higher atom economy of the stepwise cyclization approach align with green chemistry principles, helping manufacturers meet increasingly stringent environmental regulations. This scalability ensures that the commercial scale-up of complex peptide APIs can be achieved without compromising on quality or sustainability goals.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis method is critical for stakeholders evaluating its potential for integration into existing manufacturing portfolios. The following questions address common inquiries regarding the chemical stability, purification requirements, and regulatory compliance of the process described in CN115991738A. These insights are derived directly from the patent's experimental data and comparative examples, providing a factual basis for decision-making. For further technical clarification or to discuss specific customization needs, our team is available to provide detailed route feasibility assessments.
Q: Why is the S-Tmp protecting group preferred for the first cyclization step?
A: The S-Tmp (trimethoxyphenylthio) group allows for mild deprotection conditions using thiol reducing agents on the solid phase, preventing premature cleavage of the peptide from the resin or damage to other acid-labile protecting groups.
Q: How does this method improve purity compared to random oxidation?
A: By using orthogonal protection (S-Tmp and Acm), the method enforces a specific order of disulfide bond formation (4-12 first, then 7-15), significantly reducing the formation of mismatched disulfide isomers common in random oxidation processes.
Q: What are the scalability advantages of this solid-phase approach?
A: The process utilizes standard solid-phase peptide synthesis equipment and dipeptide fragments, which simplifies the workflow and reduces the accumulation of deletion sequences, making it highly suitable for kilogram-to-ton scale production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Plecanatide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has extensively evaluated the orthogonal protection strategy outlined in CN115991738A and possesses the technical capability to implement this route effectively. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale optimization to full-scale manufacturing is seamless. Our facility is equipped with state-of-the-art rigorous QC labs and analytical instruments capable of verifying stringent purity specifications, guaranteeing that every batch of Plecanatide meets the highest international standards for safety and efficacy.
We invite potential partners to engage with our technical procurement team to explore how this optimized synthesis route can benefit your specific supply chain requirements. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic advantages of switching to this high-efficiency process. We encourage you to request specific COA data and route feasibility assessments to validate the performance metrics discussed in this report. Let us partner with you to deliver high-quality peptide solutions that drive your business forward.
