Revolutionizing Plecanatide Production: Overcoming Solid-Phase Synthesis Barriers for Commercial Scale
Revolutionizing Plecanatide Production: Overcoming Solid-Phase Synthesis Barriers for Commercial Scale
The landscape of polypeptide drug manufacturing is undergoing a significant transformation, driven by the urgent need for more efficient and scalable synthetic routes for complex therapeutic agents. A pivotal advancement in this domain is detailed in Chinese patent CN115403660A, which discloses a highly optimized preparation method for Plecanatide, a 16-amino acid cyclic peptide approved for treating chronic idiopathic constipation. This technology addresses critical bottlenecks inherent in traditional solid-phase peptide synthesis (SPPS), specifically targeting the notorious issues of peptide resin shrinkage and the formation of deletion impurities. By re-engineering the protection strategy at key residues, this innovation offers a robust pathway for producing high-purity pharmaceutical intermediates that meet the stringent demands of global regulatory bodies. For industry leaders seeking a reliable plecanatide supplier, understanding these mechanistic breakthroughs is essential for securing a stable and cost-effective supply chain.
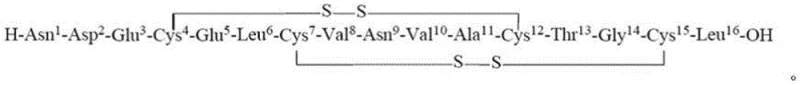
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Plecanatide has been plagued by significant technical hurdles that compromise both yield and purity. Conventional fragment condensation methods, while effective for shorter sequences, become prohibitively expensive and operationally complex when applied to a 16-residue cyclic peptide due to the extensive purification required between fragment couplings. Alternatively, standard stepwise coupling methods often suffer from severe physical changes in the reaction matrix; specifically, as the peptide chain elongates, the peptide resin tends to shrink significantly, particularly after the coupling of Val8 and Glu5. This physical contraction restricts solvent access to the reactive sites, leading to incomplete couplings and the accumulation of difficult-to-remove deletion impurities. Furthermore, previous attempts to mitigate resin shrinkage by protecting Gly14 with bulky groups like Hmb or Dmb inadvertently introduced new problems, such as steric hindrance at the adjacent Thr13 position, resulting in Thr-deleted peptides that drastically lower the overall process efficiency.
The Novel Approach
The methodology outlined in patent CN115403660A represents a paradigm shift by decoupling the relationship between resin swelling and steric hindrance through a precise modification of protecting groups. The core innovation lies in the strategic combination of using Fmoc-(Hmb)Gly-OH or Fmoc-(Dmb)Gly-OH at the 14th position while simultaneously employing an unprotected Fmoc-Thr-OH at the 13th position. This counter-intuitive approach leverages the smaller steric profile of the unprotected threonine side chain to facilitate smooth coupling, even in the presence of the bulky Gly14 protector needed to maintain resin porosity. Additionally, the process opts for Fmoc-Asn-OH without side-chain protection, which not only reduces raw material costs but also circumvents the dehydration side reactions commonly associated with removing stable Trt protecting groups. This holistic optimization ensures that the synthesis proceeds with high coupling efficiency, minimal impurity generation, and superior scalability for commercial manufacturing.
Mechanistic Insights into Optimized Solid-Phase Peptide Synthesis
To fully appreciate the technical superiority of this route, one must examine the intricate interplay of steric effects and solvation dynamics during the elongation phase. In traditional SPPS, the aggregation of peptide chains on the solid support creates a hydrophobic environment that impedes reagent diffusion, a phenomenon exacerbated by the specific sequence of Plecanatide. The introduction of the Hmb or Dmb backbone protection at Gly14 acts as a "solubility tag," disrupting inter-chain hydrogen bonding and maintaining the resin in a swollen, accessible state. However, the critical mechanistic breakthrough is the omission of the t-butyl (tBu) group on the Thr13 hydroxyl moiety. In prior art, the combination of a bulky Gly14 protector and a tBu-protected Thr13 created a "double steric wall" that prevented the incoming activated amino acid from reaching the amine terminus. By leaving the Thr13 side chain free, the local steric density is sufficiently reduced to allow rapid acylation, thereby eliminating the formation of Thr-deletion sequences which are notoriously difficult to separate from the target molecule during downstream purification.
Furthermore, the management of cysteine protecting groups plays a vital role in the fidelity of the final oxidative folding. The patent specifies an orthogonal protection strategy utilizing combinations of Acm (acetamidomethyl), Trt (trityl), or tBu groups to differentiate the two pairs of disulfide bonds required for the cyclic structure. This orthogonality allows for a stepwise oxidation process where the first disulfide bond is formed while the second pair remains protected, preventing the formation of scrambled isomers. The cleavage cocktail, typically comprising TFA with scavengers like Tis or m-cresol, is carefully tuned to remove acid-labile groups while preserving those intended for the second oxidation step. This precise control over the redox states of the cysteine residues ensures that the final cyclization yields the correct native conformation of Plecanatide, minimizing the burden on preparative HPLC and maximizing the recovery of biologically active product.
How to Synthesize Plecanatide Efficiently
The implementation of this advanced synthesis route requires strict adherence to specific operational parameters to replicate the high yields and purity reported in the patent data. The process begins with the preparation of the initial resin loaded with Fmoc-Leu, targeting a substitution value between 0.20 and 0.60 mmol/g to balance loading capacity with steric freedom. Subsequent amino acid couplings utilize standard activation reagents such as HBTU/HOBt/DIEA or HOBt/DIC, but the sequence order and protecting group selection are non-negotiable for success. Specifically, the coupling of Thr13 must be performed without side-chain protection, and the Gly14 residue must carry the Hmb or Dmb modification. Following the assembly of the full linear sequence, the peptide is cleaved from the resin and subjected to a controlled, two-stage oxidation protocol to form the disulfide bridges. For a detailed breakdown of the specific reagent quantities, reaction times, and workup procedures, please refer to the standardized synthesis guide below.
- Couple Fmoc-Leu-OH to resin (Wang or 2-Cl-Trt) to achieve a substitution value of 0.20-0.60 mmol/g.
- Sequentially couple protected amino acids using Fmoc-(Hmb/Dmb)Gly at position 14 and crucially, unprotected Fmoc-Thr-OH at position 13 to prevent resin shrinkage.
- Cleave the peptide resin using TFA cocktails, followed by stepwise oxidation to form the two disulfide bonds and finalize the cyclic structure.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis method translates directly into tangible operational efficiencies and risk mitigation. Traditional methods for producing complex cyclic peptides often suffer from low crude purity, necessitating extensive and costly chromatographic purification steps that consume significant time and resources. By fundamentally improving the quality of the crude peptide through better coupling efficiency and impurity control, this new process drastically simplifies the downstream purification workflow. This reduction in processing complexity means that manufacturing cycles can be shortened, allowing for faster turnaround times and improved responsiveness to market demand fluctuations without compromising on quality standards.
- Cost Reduction in Manufacturing: The economic impact of this technology is driven by the elimination of expensive protecting groups and the reduction of purification losses. By utilizing unprotected Threonine and Asparagine derivatives, the raw material costs are significantly lowered compared to routes requiring heavily protected building blocks. Moreover, the substantial increase in crude purity means that less product is lost during the final HPLC purification stages, leading to a higher overall yield of the finished API. This efficiency gain effectively lowers the cost of goods sold (COGS), providing a competitive pricing advantage in the global marketplace for high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the unpredictability of complex peptide syntheses, where batch failures due to resin shrinkage or coupling errors can cause significant delays. The robust nature of this new method, which effectively solves the resin shrinkage issue, ensures consistent batch-to-batch performance and high reproducibility. This reliability allows suppliers to commit to stricter delivery schedules and maintain safety stock levels with greater confidence. For buyers, this translates to a more secure supply of critical medication ingredients, reducing the risk of production stoppages and ensuring that patient needs are met without interruption.
- Scalability and Environmental Compliance: Scaling peptide synthesis from laboratory to commercial tonnage is fraught with challenges, particularly regarding solvent usage and waste generation. The streamlined nature of this process, with its fewer purification steps and higher yields, inherently reduces the volume of organic solvents and chemical reagents required per kilogram of product. This reduction in chemical intensity not only lowers disposal costs but also aligns with increasingly stringent environmental regulations and sustainability goals. The ability to scale this process from 100 kgs to multi-ton production levels with minimal re-optimization makes it an ideal candidate for long-term commercial partnerships focused on sustainable growth.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Plecanatide using this advanced methodology. These insights are derived directly from the experimental data and claims presented in the underlying patent literature, providing a transparent view of the technology's capabilities. Understanding these details helps stakeholders make informed decisions about integrating this supply source into their broader procurement strategies.
Q: How does the novel method solve the resin shrinkage problem in Plecanatide synthesis?
A: By utilizing Fmoc-(Hmb)Gly or Fmoc-(Dmb)Gly at position 14 combined with an unprotected Thr13 side chain, the method significantly reduces steric hindrance and prevents the peptide resin from shrinking, which traditionally causes difficult couplings and low yields.
Q: What are the purity specifications achievable with this new process?
A: The optimized process described in patent CN115403660A achieves a refined peptide purity greater than 99.5%, with single impurities controlled below 0.1%, significantly outperforming conventional methods that struggle with Thr-deleted peptides.
Q: Why is the removal of the Asn side-chain protecting group beneficial?
A: Using Fmoc-Asn-OH without side-chain protection eliminates the need for stable groups like Trt, which require harsh conditions for removal. This reduces production costs and avoids side reactions such as Asn dehydration, streamlining the purification workflow.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Plecanatide Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of advanced synthesis routes like CN115403660A are fully realized in large-scale operations. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch of Plecanatide meets the highest international standards for identity, potency, and impurity profiles.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic advantages tailored to your volume needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to evaluate the superior quality and reliability that NINGBO INNO PHARMCHEM brings to the global pharmaceutical market.
