Revolutionizing Diaryl Disulfide Production: A Green Aqueous Catalytic Approach for Commercial Scale
Revolutionizing Diaryl Disulfide Production: A Green Aqueous Catalytic Approach for Commercial Scale
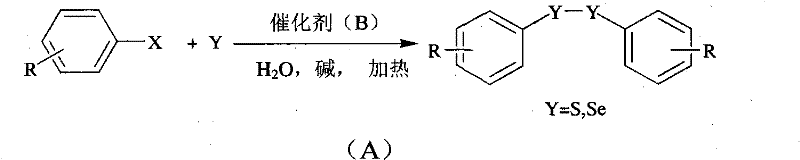
The chemical industry is currently undergoing a paradigm shift towards sustainable manufacturing processes, driven by stringent environmental regulations and the economic necessity of reducing waste disposal costs. Patent CN102010282A introduces a groundbreaking methodology for the preparation of diaryl disulfide and diaryl diselenide compounds, utilizing a water-soluble transition metal catalyst system in a pure aqueous phase. This innovation represents a significant departure from traditional organic solvent-based syntheses, offering a pathway to high-purity intermediates with drastically reduced environmental footprints. By leveraging elemental sulfur or selenium powder directly with halogenated aromatic hydrocarbons, this process eliminates the need for hazardous thiophenol precursors and volatile organic solvents. For R&D directors and procurement managers alike, this technology promises not only enhanced safety profiles but also substantial operational efficiencies in the production of critical pharmaceutical and polymer additives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of diaryl disulfides has relied heavily on the oxidation of thiophenols or the reaction of thiophenols with sulfuryl chloride in organic media. These conventional routes present severe logistical and safety challenges, primarily due to the intense, unpleasant odor and high toxicity of thiophenol, which necessitates specialized containment facilities and rigorous worker protection protocols. Furthermore, the reliance on organic solvents such as dichloromethane or benzene creates significant downstream burdens, including the energy-intensive processes required for solvent recovery and the generation of hazardous waste streams that complicate regulatory compliance. The use of strong oxidants or chlorinating agents often leads to over-oxidation by-products, resulting in lower selectivity and requiring complex purification steps like column chromatography that are difficult to translate to multi-ton commercial scales. Additionally, the synthesis of diaryl diselenides has historically lacked a robust industrial method, limiting their application despite their valuable biological and material properties.
The Novel Approach
In stark contrast, the aqueous catalytic method described in the patent utilizes benign elemental sulfur or selenium powder as the chalcogen source, effectively bypassing the handling issues associated with thiols. The reaction proceeds in water, the most abundant and non-toxic solvent available, which inherently reduces the fire risk and eliminates the emission of volatile organic compounds (VOCs). This novel approach employs a water-soluble transition metal complex, preferably based on copper, iron, or palladium, coordinated with ligands such as phenanthroline to drive the coupling efficiency. The system demonstrates remarkable versatility, accommodating a wide range of functional groups including nitro, cyano, and hydroxyl substituents without the need for protective group strategies. By operating under mild alkaline conditions with inorganic bases like sodium hydroxide, the process achieves high yields, often exceeding 90%, while generating simple inorganic salts as the primary by-products, thereby streamlining the work-up procedure to a simple extraction and crystallization.
Mechanistic Insights into Copper-Catalyzed Aqueous Coupling
The core of this technological advancement lies in the unique behavior of the water-soluble transition metal catalyst within the aqueous medium. The catalyst, typically a copper complex ligated by nitrogen-donor ligands like 1,10-phenanthroline or 2,2'-bipyridine, facilitates the activation of the carbon-halogen bond in the aryl halide substrate. In the presence of a base, the elemental sulfur or selenium is activated to form nucleophilic species that interact with the metal center. The proposed mechanism involves an oxidative addition of the aryl halide to the low-valent metal center, followed by the insertion of the chalcogen species. This is succeeded by a reductive elimination step that forms the stable disulfide or diselenide bond while regenerating the active catalyst species. The aqueous environment plays a crucial role not just as a solvent but as a medium that stabilizes ionic intermediates and enhances the solubility of the inorganic base, ensuring a homogeneous reaction environment that maximizes collision frequency between reactants.
Impurity control in this system is inherently superior due to the high chemoselectivity of the catalyst. Unlike radical-based oxidations which can lead to random coupling and polysulfide formation, this coordination catalysis directs the reaction specifically towards the formation of the disulfide or diselenide linkage. The use of phase transfer catalysts, such as tetrabutylammonium fluoride, further enhances the reaction kinetics by facilitating the transport of ionic species across the interface between the solid sulfur/selenium and the aqueous phase. This ensures that the concentration of reactive intermediates remains optimized, minimizing side reactions such as dehalogenation or homocoupling of the aryl rings without chalcogen insertion. The result is a crude product profile that is significantly cleaner than those obtained from traditional methods, reducing the load on downstream purification units and increasing the overall mass balance efficiency of the manufacturing process.
How to Synthesize Diaryl Disulfides Efficiently
The synthesis protocol derived from this patent offers a straightforward operational framework suitable for both laboratory optimization and plant-scale implementation. The process begins with the precise charging of halogenated aromatic substrates, elemental sulfur, and the copper-ligand catalyst complex into a reactor containing water. The addition of a stoichiometric amount of inorganic base is critical to neutralize the hydrogen halide by-product and drive the equilibrium forward. Detailed standard operating procedures regarding temperature ramping, stirring rates, and quenching protocols are essential to maintain the integrity of the catalyst and ensure consistent batch-to-batch quality. For a comprehensive understanding of the specific molar ratios, temperature profiles, and isolation techniques validated by experimental data, please refer to the standardized synthesis guide below.
- Prepare the reaction mixture by combining halogenated aromatic hydrocarbon, elemental sulfur powder, and a water-soluble transition metal catalyst (preferably copper-based) in an aqueous medium.
- Add an inorganic base such as sodium hydroxide or potassium carbonate and a phase transfer catalyst like tetrabutylammonium fluoride to facilitate the reaction interface.
- Heat the reaction mixture to temperatures between 60°C and 130°C for 12 to 48 hours, then isolate the product via extraction and purification.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this aqueous catalytic technology offers compelling arguments for cost restructuring and supply chain resilience. The elimination of expensive and hazardous organic solvents translates directly into reduced raw material expenditure and lowers the total cost of ownership associated with solvent storage, handling, and recycling infrastructure. Moreover, the substitution of malodorous thiophenols with odorless elemental sulfur significantly improves workplace safety and reduces the likelihood of community complaints or regulatory fines related to air quality, thereby safeguarding the social license to operate. The simplicity of the work-up procedure, which often requires only filtration and extraction, reduces the demand for specialized purification equipment and shortens the overall cycle time per batch. These factors collectively contribute to a more agile manufacturing capability that can respond rapidly to market demands for high-purity intermediates.
- Cost Reduction in Manufacturing: The economic benefits of this process are multifaceted, stemming primarily from the drastic simplification of the input material list. By utilizing commodity chemicals like sulfur powder and sodium hydroxide instead of custom-synthesized thiols, the direct material costs are significantly lowered. Furthermore, the absence of organic solvents removes the substantial energy costs associated with distillation and solvent recovery, which typically account for a large portion of utility expenses in fine chemical plants. The high atom economy of the reaction ensures that the majority of the starting mass is converted into the desired product, minimizing waste disposal fees. Qualitatively, this represents a shift from a high-waste, high-energy process to a lean, efficient manufacturing model that maximizes margin potential.
- Enhanced Supply Chain Reliability: Supply chain stability is greatly improved by the use of globally available, non-restricted raw materials. Elemental sulfur and inorganic bases are produced in massive quantities for other industries, ensuring a steady supply with minimal price volatility compared to specialized organosulfur reagents. The robustness of the catalyst system, which tolerates a wide range of substrates, allows for flexible sourcing of aryl halides, enabling procurement teams to switch suppliers based on availability without requalifying the entire process. Additionally, the non-hazardous nature of the reagents simplifies logistics and transportation, reducing the regulatory burden and insurance costs associated with shipping dangerous goods. This reliability ensures continuous production schedules and mitigates the risk of stockouts for critical downstream customers.
- Scalability and Environmental Compliance: Scaling this process from kilogram to multi-ton production is inherently safer and more straightforward due to the use of water as the reaction medium. Water has a high heat capacity, which aids in temperature control during exothermic steps, reducing the risk of thermal runaway incidents that can plague organic solvent reactions. From an environmental compliance standpoint, the aqueous effluent is easier to treat than mixed organic waste streams, aligning with increasingly strict global discharge standards. The process generates minimal hazardous waste, supporting corporate sustainability goals and enhancing the brand reputation of manufacturers who adopt green chemistry principles. This scalability ensures that the technology remains viable and cost-effective regardless of the production volume required.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aqueous catalytic technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on reaction scope, catalyst performance, and operational parameters. Understanding these nuances is critical for process engineers and technical buyers evaluating the feasibility of integrating this method into existing production lines. The insights provided here aim to bridge the gap between academic innovation and industrial application, ensuring that stakeholders have a realistic expectation of the technology's capabilities and limitations in a commercial setting.
Q: What are the primary advantages of using water as a solvent for disulfide synthesis?
A: Using water eliminates the need for volatile organic compounds (VOCs), significantly reducing environmental impact and fire hazards. It also simplifies product isolation since organic products often separate easily or can be extracted without complex solvent removal steps associated with high-boiling organic solvents.
Q: Can this catalytic method tolerate sensitive functional groups on the aromatic ring?
A: Yes, the patent data indicates high functional group tolerance. Substituents such as nitro, cyano, acetyl, hydroxyl, and alkoxy groups remain intact under the mild alkaline aqueous conditions, making it suitable for complex pharmaceutical intermediate synthesis.
Q: Is this method scalable for industrial production of diaryl diselenides?
A: The method is highly scalable due to the use of inexpensive, commercially available reagents like elemental selenium and simple inorganic bases. The absence of air-sensitive or moisture-sensitive reagents further enhances its viability for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diaryl Disulfide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of green catalytic technologies in modernizing the fine chemical supply chain. Our team of expert process chemists has extensively evaluated the aqueous phase synthesis route described in CN102010282A and confirmed its viability for large-scale manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of diaryl disulfide or diselenide intermediate meets the exacting standards required by the pharmaceutical and agrochemical industries.
We invite you to collaborate with us to optimize your supply chain for these critical intermediates. By leveraging our expertise in catalytic process development, we can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments. Together, we can drive down costs and enhance the sustainability of your manufacturing operations, securing a competitive advantage in the global market through innovative chemistry and reliable execution.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
