Revolutionizing Diaryl Disulfide Production: A Green Aqueous Catalytic Approach for Commercial Scale
The chemical industry is currently witnessing a paradigm shift towards sustainable manufacturing processes, driven by stringent environmental regulations and the economic necessity of reducing waste disposal costs. Patent CN102010282B introduces a groundbreaking methodology for the preparation of diaryl disulfide and diaryl diselenide compounds, utilizing a purely aqueous phase catalytic system that fundamentally alters the traditional approach to organochalcogen synthesis. This innovation leverages water-soluble transition metal catalysts to efficiently mediate the reaction between elemental sulfur or selenium powder and halogenated aromatic hydrocarbons, bypassing the need for hazardous organic solvents entirely. By operating in a pure water phase, this technology not only aligns with the principles of Green Chemistry but also offers substantial improvements in operational safety and cost-efficiency for large-scale production facilities. The ability to tolerate a wide range of functional groups while maintaining high yields positions this method as a superior alternative for producing high-purity pharmaceutical intermediates and agrochemical precursors. For global supply chain leaders, adopting such aqueous-based technologies represents a strategic move towards more resilient and environmentally compliant manufacturing networks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional industrial methods for synthesizing disulfide compounds have long relied on the oxidation of thiophenols or the reaction of thiophenols with sulfuryl chloride in organic solvents, processes that are inherently fraught with significant drawbacks. These conventional routes often necessitate the use of volatile and flammable organic solvents, which pose serious safety hazards in large-scale reactors and require complex, energy-intensive recovery systems to meet environmental standards. Furthermore, the handling of thiophenols is problematic due to their extremely unpleasant odor and high toxicity, creating challenging working conditions and increasing the risk of occupational exposure incidents. Side reactions in these traditional pathways frequently lead to the formation of unwanted by-products, thereby reducing the overall yield and complicating the downstream purification process which is critical for pharmaceutical applications. The reliance on stoichiometric oxidants or chlorinating agents also generates substantial amounts of inorganic salt waste, contributing to a heavier environmental footprint and higher waste treatment costs for manufacturers. Consequently, there is an urgent demand within the fine chemical sector for a cleaner, safer, and more atom-economical synthetic route that can overcome these persistent inefficiencies.
The Novel Approach
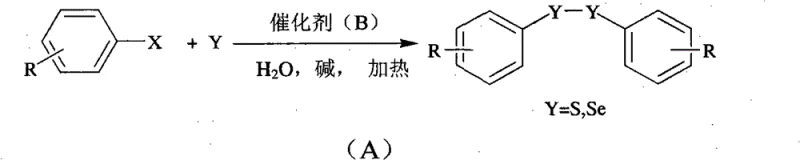
In stark contrast to legacy techniques, the novel aqueous phase catalytic method described in the patent data utilizes elemental sulfur or selenium powder directly as the chalcogen source, eliminating the need for pre-functionalized thiols or selenols. This direct coupling strategy simplifies the raw material supply chain by leveraging inexpensive and commercially abundant elemental powders, which are significantly safer and easier to handle than their volatile thiol counterparts. The use of water as the sole reaction medium not only removes the fire hazard associated with organic solvents but also drastically simplifies the product isolation process, as the organic product can often be easily separated from the aqueous phase without complex distillation steps. The catalytic system demonstrates remarkable versatility, accommodating a broad spectrum of functional groups on the aromatic ring without the need for protective group chemistry, which further streamlines the synthetic sequence. By integrating a phase-transfer catalyst and a water-soluble metal complex, the reaction achieves high conversion rates and selectivity, ensuring that the final product meets the rigorous purity specifications required for high-value applications. This approach effectively transforms the synthesis of organosulfur and organoselenium compounds into a more sustainable and economically viable operation.
Mechanistic Insights into Cu-Catalyzed Aqueous Coupling
The core of this technological breakthrough lies in the sophisticated interplay between the water-soluble transition metal catalyst and the reaction substrates within the aqueous environment. The catalyst, typically a copper complex coordinated with ligands such as phenanthroline or dipyridyl, facilitates the oxidative addition of the halogenated aromatic hydrocarbon, activating the carbon-halogen bond for subsequent nucleophilic attack. In the presence of a strong base like sodium hydroxide or potassium carbonate, the elemental sulfur or selenium is activated to form reactive chalcogenide species that can effectively couple with the activated aryl intermediate. The aqueous medium plays a crucial role not just as a solvent but as a thermal buffer that allows the reaction to proceed at elevated temperatures safely, enhancing the kinetic energy of the molecules without risking thermal runaway. The inclusion of a phase-transfer catalyst, such as tetrabutylammonium fluoride, further enhances the reaction efficiency by facilitating the transport of ionic species between the aqueous phase and the organic substrate interface. This mechanistic pathway ensures that the reaction proceeds with high atom economy, minimizing the generation of waste and maximizing the utilization of the expensive halogenated starting materials.
Understanding the impurity profile is critical for R&D directors aiming to implement this process for GMP-grade production, and the aqueous nature of the reaction inherently suppresses many common organic side reactions. The high selectivity of the copper-catalyzed system minimizes the formation of homocoupled by-products or over-oxidized species that are often encountered in traditional oxidative coupling methods. The use of elemental sulfur in controlled stoichiometric ratios prevents the formation of polysulfides, which can be difficult to separate and often contaminate the final product stream. Moreover, the water-soluble catalyst can potentially be recovered or treated more easily than heterogeneous catalysts that require filtration, reducing the risk of metal contamination in the final active pharmaceutical ingredient. The robustness of the reaction conditions allows for a wider operating window, meaning that minor fluctuations in temperature or pH do not drastically alter the impurity spectrum, providing a more stable and predictable manufacturing process. This level of control over the reaction mechanism is essential for ensuring batch-to-b consistency and meeting the stringent regulatory requirements of the global pharmaceutical market.
How to Synthesize Diaryl Disulfide Efficiently
To implement this synthesis route effectively, operators must adhere to precise stoichiometric ratios and reaction conditions as outlined in the patent examples to ensure optimal yield and purity. The process begins with the careful selection of the halogenated aromatic substrate and the corresponding amount of elemental sulfur or selenium, followed by the addition of the water-soluble catalyst and base in pure water. Detailed standard operating procedures regarding temperature ramping, stirring speeds, and workup protocols are essential to replicate the high success rates observed in the laboratory examples.
- Prepare the reaction system by mixing halogenated aromatic hydrocarbon, elemental sulfur powder, and a water-soluble transition metal catalyst in pure water.
- Add a mineral or organic base such as sodium hydroxide or potassium carbonate to facilitate the deprotonation and activation of the chalcogen source.
- Heat the mixture to 50-150°C for 12-48 hours under stirring, then extract the product with ethyl acetate and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this aqueous catalytic technology offers profound strategic benefits that extend far beyond simple chemical efficiency. The elimination of expensive and regulated organic solvents results in a drastic simplification of the raw material procurement process, reducing dependency on volatile petrochemical supply chains that are subject to frequent price fluctuations. By removing the need for solvent recovery infrastructure, manufacturing facilities can significantly lower their capital expenditure and operational overhead, leading to substantial cost savings in the overall production budget. The use of non-flammable water as the reaction medium enhances plant safety profiles, potentially lowering insurance premiums and reducing the regulatory burden associated with handling hazardous materials. Furthermore, the simplified workup procedure reduces the time and energy required for product isolation, allowing for faster turnaround times and improved throughput in multi-purpose production units. These factors collectively contribute to a more resilient and cost-effective supply chain capable of responding swiftly to market demands without compromising on quality or compliance.
- Cost Reduction in Manufacturing: The replacement of organic solvents with water eliminates the significant costs associated with solvent purchase, storage, and distillation recovery, which traditionally account for a large portion of variable manufacturing expenses. Additionally, the use of elemental sulfur and selenium powder as direct reagents avoids the premium pricing of pre-functionalized thiols or selenols, further driving down the raw material cost basis. The reduction in waste generation also translates to lower disposal fees and reduced environmental compliance costs, creating a leaner and more profitable operational model. By streamlining the purification process, manufacturers can reduce the consumption of silica gel and eluents used in chromatography, adding another layer of cost efficiency to the production cycle.
- Enhanced Supply Chain Reliability: Sourcing elemental sulfur and selenium is generally more stable and less prone to geopolitical disruptions compared to specialized organic reagents or solvents, ensuring a consistent supply of key inputs. The robustness of the aqueous system means that production is less sensitive to variations in raw material quality, reducing the risk of batch failures and supply interruptions. Simplified logistics for non-hazardous aqueous waste streams also streamline the downstream supply chain, removing bottlenecks associated with hazardous waste transportation and treatment. This reliability is crucial for maintaining continuous production schedules and meeting the just-in-time delivery expectations of major pharmaceutical and agrochemical clients.
- Scalability and Environmental Compliance: The inherent safety of water-based chemistry allows for easier scale-up from pilot plants to full commercial production without the need for explosion-proof infrastructure or complex ventilation systems. The process aligns perfectly with increasingly strict global environmental regulations regarding VOC emissions and hazardous waste discharge, future-proofing the manufacturing facility against regulatory changes. The high atom economy of the reaction ensures that raw materials are converted efficiently into product, minimizing the environmental footprint per kilogram of output. This sustainability profile enhances the brand value of the manufacturer, appealing to end-clients who are prioritizing green supply chains and corporate social responsibility in their vendor selection criteria.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this aqueous catalytic technology in industrial settings. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation to ensure accuracy and relevance.
Q: What are the primary advantages of using water as a solvent in this synthesis?
A: Using water eliminates the need for volatile organic compounds (VOCs), significantly reducing environmental impact and solvent recovery costs while improving operational safety by removing flammability risks associated with traditional organic solvents.
Q: Which catalysts are most effective for this aqueous transformation?
A: Water-soluble transition metal complexes, particularly those based on copper, iron, or palladium with ligands like phenanthroline, demonstrate high efficiency in catalyzing the coupling of elemental sulfur with aryl halides in aqueous media.
Q: Is this method suitable for large-scale industrial production?
A: Yes, the process utilizes inexpensive and readily available raw materials like sulfur powder and halogenated aromatics, and the simple workup procedure involving extraction makes it highly amenable to commercial scale-up and continuous manufacturing processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Diaryl Disulfide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this aqueous catalytic technology and have integrated similar green chemistry principles into our own CDMO operations. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods can be successfully translated into robust industrial processes. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of diaryl disulfide or diselenide intermediate meets the highest international standards. Our commitment to sustainability and efficiency mirrors the advantages offered by this patent, making us an ideal partner for companies looking to modernize their supply chain.
We invite you to collaborate with us to explore how this advanced synthesis method can be tailored to your specific project requirements. Please contact our technical procurement team to request a Customized Cost-Saving Analysis and discuss the feasibility of implementing this route for your target molecules. We are ready to provide specific COA data and route feasibility assessments to support your R&D and sourcing decisions, ensuring a seamless transition to a more efficient and sustainable manufacturing partner.
