Advanced Synthesis of Aprepitant: A Technical Breakthrough for Commercial Scale-up
Advanced Synthesis of Aprepitant: A Technical Breakthrough for Commercial Scale-up
The pharmaceutical industry continuously seeks robust and scalable methodologies for the production of high-value active pharmaceutical ingredients (APIs), particularly for complex molecules like Aprepitant, a potent NK1 receptor antagonist approved for the prevention of chemotherapy-induced nausea and vomiting. Patent CN113582982B introduces a transformative preparation method that addresses longstanding challenges in stereochemical control and process efficiency. This technical disclosure outlines a sophisticated two-step synthesis involving a novel chiral resolution strategy utilizing calcium chloride additives and a subsequent catalytic coupling reaction. By shifting away from harsh hydrogenation conditions and cumbersome purification techniques, this innovation offers a compelling pathway for manufacturers aiming to secure a reliable NK1 receptor antagonist supplier status while optimizing their production economics.
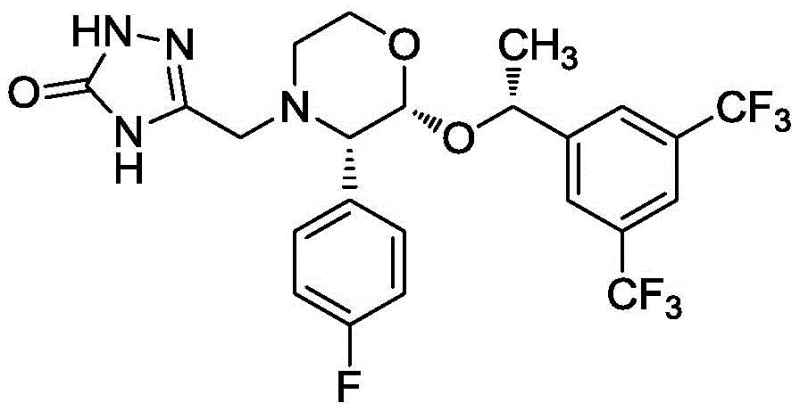
Aprepitant, chemically known as 5-[2(R)-[1(R)-[3,5-bis(trifluoromethyl)phenyl]ethoxy]-3(S)-(4-fluorophenyl)morpholin-4-ylmethyl]-3,4-dihydro-2H-1,2,4-triazol-3-one, possesses three distinct chiral centers that dictate its biological activity. The structural complexity, as illustrated in the molecular diagram, necessitates precise stereochemical control during synthesis to ensure therapeutic efficacy and regulatory compliance. The presence of multiple trifluoromethyl groups and the specific morpholine-triazole linkage requires a synthesis route that minimizes racemization and impurity formation. The patent data highlights that achieving high optical purity is not merely a quality metric but a fundamental requirement for the commercial viability of this antiemetic agent, driving the need for advanced chiral resolution technologies over traditional racemic synthesis followed by separation.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historical approaches to synthesizing the critical chiral intermediates for Aprepitant have been plagued by significant operational hurdles that hinder large-scale manufacturing. Literature reports frequently describe the use of asymmetric hydrogenation catalysis requiring high-pressure hydrogen sources, which introduces severe safety risks and necessitates specialized, capital-intensive reactor infrastructure. Furthermore, existing patents, such as CN112174909A, disclose methods relying on specific chiral catalysts that are not only difficult to source commercially but also mandate complex purification steps like column chromatography. These chromatographic processes are notoriously inefficient for industrial scale-up due to high solvent consumption, low throughput, and difficulties in solvent recovery, ultimately inflating the cost of goods sold (COGS) and extending production lead times beyond acceptable limits for global supply chains.
In addition to equipment and purification challenges, conventional alkylation strategies for the final coupling step often suffer from sluggish kinetics and poor yields. Prior art methods typically employ halogenated precursors like 5-chloromethyl or 5-bromomethyl triazolones, which require extended reaction times exceeding 10 hours even at elevated temperatures of 30-35°C. Such prolonged exposure to reactive conditions increases the likelihood of side reactions and degradation, resulting in crude products that demand further refining to meet the stringent purity specifications of >99.8% required for API manufacture. The cumulative effect of these inefficiencies—harsh conditions, difficult purification, and long cycle times—creates a bottleneck that compromises the reliability of the supply chain for this essential medication.
The Novel Approach
The methodology disclosed in patent CN113582982B represents a paradigm shift by introducing a mild, crystallization-driven chiral resolution followed by an efficient catalytic coupling. The core innovation lies in the use of a mixed solvent system comprising acetone and methyl tert-butyl ether, augmented by the strategic addition of calcium chloride (CaCl2) in an alkaline environment. Unlike traditional methods where additives might participate chemically, CaCl2 here serves a physical role in enhancing crystallization efficiency without being consumed, allowing for the isolation of the chiral intermediate (Compound II) with exceptional optical purity (ee value > 99.6%) and high yield (>90%). This eliminates the need for expensive chiral catalysts and column chromatography, drastically simplifying the workflow.
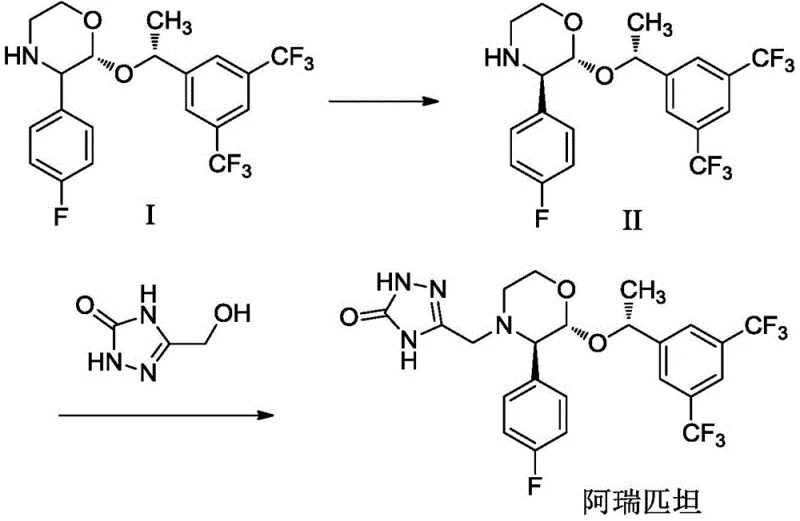
Furthermore, the final coupling reaction utilizes 5-hydroxymethyl-2,4-dihydro-[1,2,4]triazol-3-one directly, catalyzed by 1,8-diazabicyclo[5.4.0]undec-7-ene (DBU) in a ternary solvent mixture of ethyl acetate, acetone, and methanol. This approach allows the reaction to proceed under relatively mild conditions, with the patent noting the potential for room temperature operation or mild reflux, significantly reducing energy consumption compared to high-pressure alternatives. The direct use of the hydroxymethyl precursor avoids the handling of hazardous halogenated intermediates, improving process safety. The result is a streamlined synthesis that delivers Aprepitant with HPLC purity exceeding 99.9% and yields approaching 90%, demonstrating superior scalability and cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Chiral Resolution and DBU-Catalyzed Coupling
The chiral resolution step is mechanistically fascinating due to the synergistic effect of the solvent system and the inorganic salt additive. The process begins with the dissolution of the racemic precursor (Compound I) in an acetone-methyl tert-butyl ether mixture, where D-(-)-diethyl tartrate acts as the resolving agent. The addition of CaCl2 in the presence of a base like N,N-diisopropylethylamine creates a specific ionic environment that favors the nucleation and growth of the desired enantiomer crystals. The patent elucidates that CaCl2 does not chemically react but modifies the solubility profile and crystal lattice formation, effectively acting as a crystallization promoter. Post-reaction, the addition of water quenches the system and simultaneously dissolves the CaCl2, facilitating the isolation of the pure chiral intermediate (Compound II) through simple filtration, a mechanism that ensures consistent batch-to-batch reproducibility.
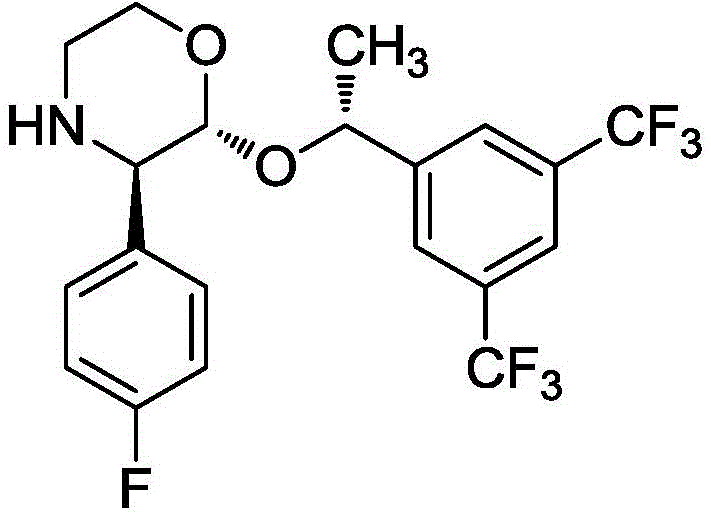
Following the isolation of the chiral morpholine intermediate, the synthesis proceeds via a nucleophilic substitution mechanism catalyzed by DBU. The base activates the hydroxyl group of the triazole derivative or facilitates the displacement reaction with the morpholine nitrogen, forming the critical C-N bond that links the two major fragments of the molecule. The use of a mixed solvent system (ethyl acetate-acetone-methanol) is crucial here, as it balances the solubility of both the polar triazole component and the lipophilic morpholine intermediate, ensuring a homogeneous reaction phase that maximizes collision frequency. This mechanistic optimization prevents the formation of oligomeric byproducts and ensures that the stereochemical integrity of the three chiral centers is maintained throughout the coupling, resulting in a final product with minimal impurity profiles suitable for direct crystallization.
How to Synthesize Aprepitant Efficiently
Implementing this novel synthesis route requires precise control over solvent ratios and additive concentrations to replicate the high yields and purity reported in the patent data. The process is divided into two distinct stages: the chiral resolution of the morpholine core and the subsequent coupling with the triazole moiety. Operators must adhere strictly to the specified volume-to-mass ratios, such as maintaining an acetone to methyl tert-butyl ether ratio between 1:1 and 1:4 during the resolution phase, to ensure optimal crystallization kinetics. The following guide outlines the standardized operational parameters derived from the patent examples, providing a clear roadmap for technical teams aiming to adopt this high-purity pharmaceutical intermediates production method.
- Dissolve the racemic precursor (Compound I) in an acetone-methyl tert-butyl ether mixed solvent, add D-(-)-diethyl tartrate and CaCl2 under alkaline conditions to induce chiral crystallization of Compound II.
- Filter and purify the chiral intermediate (Compound II) using water quenching and recrystallization techniques to achieve high optical purity (ee value > 99%).
- React Compound II with 5-hydroxymethyl-2,4-dihydro-[1,2,4]triazol-3-one in an ethyl acetate-acetone-methanol solvent system using DBU as a catalyst to form Aprepitant.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the synthesis method described in CN113582982B translates into tangible strategic advantages beyond mere technical metrics. The elimination of column chromatography and high-pressure hydrogenation equipment significantly lowers the barrier to entry for manufacturing, allowing for more flexible production scheduling and reduced capital expenditure. By replacing hazardous halogenated reagents with safer hydroxymethyl precursors and utilizing common organic solvents, the process mitigates regulatory risks associated with hazardous material handling and disposal. This shift not only enhances the safety profile of the manufacturing site but also ensures greater supply chain continuity by reducing dependency on specialized, hard-to-source catalysts and equipment.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the simplification of the purification workflow. Traditional methods relying on column chromatography consume vast quantities of silica gel and solvents, generating significant waste disposal costs and extending cycle times. By switching to a crystallization-based purification enabled by the CaCl2 additive, manufacturers can drastically reduce solvent usage and eliminate the cost of chromatographic media. Furthermore, the avoidance of expensive chiral metal catalysts, which often require complex recovery systems or are single-use, results in substantial raw material cost savings. The higher yields reported (>90% for both steps) mean less starting material is wasted, directly improving the overall atom economy and reducing the cost per kilogram of the final API.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of readily available, commodity-grade chemicals rather than bespoke catalysts. The reliance on D-(-)-diethyl tartrate and DBU, which are commercially abundant, reduces the risk of supply disruptions caused by vendor shortages of specialized reagents. Additionally, the milder reaction conditions (room temperature to mild reflux) reduce the energy load on the facility and decrease the wear and tear on reactor vessels, leading to higher equipment availability and lower maintenance downtime. This operational robustness ensures that production targets can be met consistently, reducing lead time for high-purity pharmaceutical intermediates and allowing for more responsive inventory management.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, the process is inherently greener and more adaptable to large-volume production. The removal of heavy metal catalysts and halogenated byproducts simplifies wastewater treatment and aligns with increasingly stringent global environmental regulations. The crystallization steps are easily scalable from pilot plant to commercial tonnage without the engineering complexities associated with high-pressure hydrogenation reactors. This ease of scale-up facilitates the commercial scale-up of complex pharmaceutical intermediates, allowing manufacturers to rapidly respond to market demand surges without compromising on quality or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical implications for industrial adoption. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating this method into their existing manufacturing portfolios.
Q: What is the key advantage of the CaCl2 additive in the chiral resolution step?
A: According to patent CN113582982B, CaCl2 does not participate chemically in the reaction but is critical for improving crystallization efficiency and yield during the chiral resolution process when added in an alkaline environment.
Q: How does this method improve upon traditional hydrogenation routes?
A: Traditional methods often require high-pressure hydrogenation and complex column chromatography purification. This novel route operates under milder conditions (often room temperature or mild reflux) and utilizes crystallization for purification, significantly simplifying the operational complexity.
Q: What represents the primary cost driver reduction in this synthesis?
A: The elimination of expensive chiral catalysts that are difficult to recover, combined with the avoidance of energy-intensive high-pressure equipment and time-consuming chromatographic separation, leads to substantial operational cost reductions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aprepitant Supplier
As the global demand for effective antiemetic therapies continues to rise, securing a stable supply of high-quality Aprepitant is paramount for pharmaceutical companies. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging deep expertise in chiral chemistry and process optimization to deliver superior intermediates and APIs. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous volume requirements of multinational clients. We maintain stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of Aprepitant or its intermediates meets the highest international pharmacopeial standards.
We invite procurement leaders and R&D directors to collaborate with us to explore how this advanced synthesis technology can be integrated into your supply chain. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific production needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of technical excellence and commercial reliability.
