Advanced Organometallic Synthesis of Methyl Dihydrojasmonate for Commercial Scale-up
Advanced Organometallic Synthesis of Methyl Dihydrojasmonate for Commercial Scale-up
The global demand for high-purity fragrance ingredients continues to drive innovation in synthetic organic chemistry, particularly for established molecules like methyl dihydrojasmonate, a cornerstone of the modern perfume industry. Patent CN101519355B introduces a transformative methodology that leverages organozinc chemistry and transition metal catalysis to streamline the production of this valuable compound. Unlike traditional multi-step condensations that suffer from poor atom economy and harsh conditions, this novel approach utilizes a direct 1,4-Michael addition strategy followed by a selective oxidative cleavage. The process begins with the construction of the core cyclopentanone scaffold and proceeds through a highly controlled organometallic coupling, ensuring exceptional regioselectivity. By integrating these advanced catalytic cycles, the technology offers a pathway to significantly reduce manufacturing complexity while maintaining the stringent quality standards required by top-tier flavor and fragrance houses.
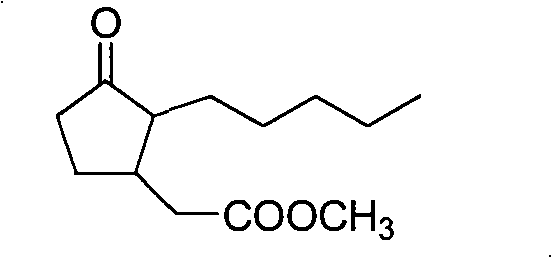
This technical breakthrough is particularly relevant for procurement and supply chain leaders seeking to diversify their sources of high-purity Flavor & Fragrance Intermediates. The patent details a route that avoids the use of extremely hazardous reagents often associated with older methodologies, replacing them with more manageable catalytic systems. The final product, characterized by its fresh, floral jasmine scent with woody undertones, is critical for both fine fragrances and functional perfumery applications. Understanding the underlying chemical architecture of this synthesis allows stakeholders to appreciate the robustness of the supply chain, as the reliance on stable, commercially available starting materials mitigates the risk of production bottlenecks. Furthermore, the high yields reported in the experimental data suggest a process that is not only chemically elegant but also economically viable for commercial scale-up of complex fragrance molecules.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of methyl dihydrojasmonate has relied on convoluted pathways that involve the condensation of dimethyl malonate with various halides, followed by decarboxylation and ring-closing reactions. These classical approaches are fraught with inefficiencies, including the generation of substantial quantities of salt waste during the decarboxylation steps and the requirement for high-temperature conditions that can degrade sensitive intermediates. Another common route involves the synthesis of 2-amyl-1,3-cyclopentanedione, which necessitates multiple protection and deprotection steps, drastically increasing the operational time and capital expenditure. The cumulative effect of these lengthy sequences is a lower overall yield and a higher impurity load, which complicates the purification process and increases the cost of goods sold. Additionally, the use of strong bases and aggressive alkylating agents in these traditional methods poses significant safety risks and environmental compliance challenges, making them less attractive in the context of modern green chemistry initiatives.
The Novel Approach
In stark contrast, the methodology described in CN101519355B employs a convergent strategy that dramatically shortens the synthetic timeline. The core innovation lies in the use of 2-pentylcyclopentenone as a versatile electrophile, which undergoes a smooth conjugate addition with an allyl zinc bromide species. This reaction is facilitated by transition metal catalysts such as nickel acetylacetonate or palladium complexes, which activate the organozinc reagent for efficient carbon-carbon bond formation. Following the construction of the carbon skeleton, the terminal alkene is selectively oxidized to a carboxylic acid using a ruthenium-catalyzed system with sodium periodate, a transformation that preserves the integrity of the ketone functionality. This streamlined sequence eliminates the need for tedious decarboxylation steps and reduces the number of isolation procedures, thereby enhancing the overall throughput. The result is a process that is not only faster but also generates a cleaner crude product, simplifying the final purification and ensuring consistent quality for the end user.
Mechanistic Insights into Ruthenium-Catalyzed Oxidative Cleavage
The heart of this synthetic innovation is the oxidative cleavage step, which converts the allyl side chain into the requisite acetic acid moiety found in the final jasmonate structure. This transformation is achieved using a catalytic amount of ruthenium trichloride hydrate in the presence of sodium periodate as the terminal oxidant. Mechanistically, the ruthenium species cycles between different oxidation states, facilitating the formation of a cyclic ruthenate ester with the alkene double bond. This intermediate subsequently undergoes fragmentation to generate the aldehyde, which is further oxidized in situ to the carboxylic acid under the reaction conditions. The beauty of this system lies in its chemoselectivity; unlike ozonolysis, which requires cryogenic temperatures and specialized equipment, or potassium permanganate, which can over-oxidize the alpha-position of the ketone, the Ru/NaIO4 system operates under mild ambient conditions. This selectivity is crucial for maintaining the stereochemical integrity and purity of the cyclopentanone ring, preventing the formation of ring-opened byproducts or over-oxidized dicarboxylic acids that would be difficult to separate.
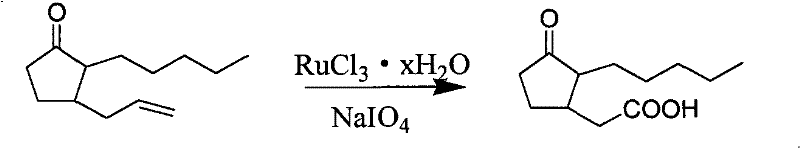
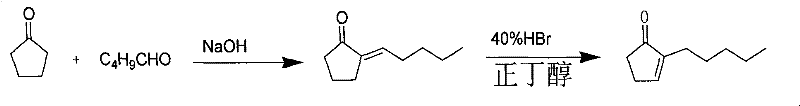
Complementing the oxidation step is the precise control exerted during the initial Michael addition. The use of organozinc reagents, generated in situ from allyl bromide and activated zinc powder, provides a nucleophile that is softer and more tolerant of functional groups than its Grignard or organolithium counterparts. In the presence of Lewis acids like trimethylchlorosilane or titanium tetrachloride, the reaction proceeds with high regioselectivity to favor the 1,4-addition product over the 1,2-addition product. This specificity is vital because 1,2-addition would result in an alcohol byproduct that does not lead to the desired jasmonate framework. The patent highlights the importance of temperature control during this stage, typically maintaining the reaction between 0°C and -15°C to suppress side reactions such as homocoupling of the organozinc species. By optimizing the ratio of catalyst to substrate and carefully managing the addition rate of the reagents, the process achieves a high degree of conversion, ensuring that the valuable starting materials are utilized efficiently with minimal waste generation.
How to Synthesize Methyl Dihydrojasmonate Efficiently
The execution of this synthesis requires careful attention to the preparation of the organometallic reagent and the sequential addition of reactants to maintain thermal control. The process begins with the activation of zinc powder, often using a small amount of 1,2-dibromoethane to remove surface oxides, followed by the formation of the allyl zinc bromide species in anhydrous tetrahydrofuran. Once the reagent is prepared, it is added to a solution containing the enone substrate and the transition metal catalyst system under an inert nitrogen atmosphere. Following the coupling reaction, the mixture is quenched, and the intermediate ketone is isolated before proceeding to the oxidation stage. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and workup procedures, are outlined in the guide below.
- Synthesize 2-pentylcyclopentenone via aldol condensation of cyclopentanone and valeraldehyde followed by dehydration and hydrobromination.
- Prepare allyl zinc bromide reagent in THF using activated zinc powder and allyl bromide under inert atmosphere.
- Perform 1,4-Michael addition of the organozinc reagent to 2-pentylcyclopentenone using a nickel or palladium catalyst system.
- Oxidatively cleave the terminal alkene of the intermediate using RuCl3 and NaIO4 to form the acid, followed by esterification with thionyl chloride and methanol.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented route offers compelling strategic benefits that extend beyond simple chemical yield. The primary advantage lies in the drastic simplification of the supply chain, as the process relies on commodity chemicals like cyclopentanone, valeraldehyde, and allyl bromide, which are produced on a massive global scale. This reliance on bulk petrochemical derivatives insulates the manufacturing process from the volatility often associated with specialty fine chemical suppliers, ensuring a steady and predictable flow of raw materials. Furthermore, the reduction in the number of synthetic steps directly correlates to a reduction in operational costs, as fewer unit operations mean lower energy consumption, reduced labor hours, and decreased equipment occupancy time. The elimination of harsh decarboxylation steps also lowers the burden on waste treatment facilities, contributing to a more sustainable and cost-effective production model that aligns with corporate sustainability goals.
- Cost Reduction in Manufacturing: The economic viability of this process is driven by the high atom economy of the Michael addition and the catalytic nature of the oxidation step. By avoiding stoichiometric amounts of expensive oxidants and eliminating the need for protecting group chemistry, the overall material cost is significantly lowered. The use of recyclable solvents like tetrahydrofuran and ethyl acetate further enhances the cost efficiency, while the high selectivity of the reactions minimizes the loss of valuable intermediates to byproduct formation. Consequently, manufacturers can achieve a substantial reduction in the cost of goods sold, allowing for more competitive pricing in the global fragrance market without compromising on margin.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route ensures consistent production output, which is critical for meeting the rigorous delivery schedules of multinational personal care companies. Because the reaction conditions are mild and the reagents are stable, the process is less susceptible to batch-to-batch variations caused by minor fluctuations in temperature or reagent quality. This reliability reduces the risk of production delays and stockouts, providing supply chain heads with greater confidence in their inventory planning. Additionally, the scalability of the organozinc chemistry has been well-documented in industrial settings, meaning that the transition from pilot plant to full-scale commercial production can be executed smoothly with minimal technical risk.
- Scalability and Environmental Compliance: From an environmental perspective, this methodology represents a significant improvement over legacy processes. The replacement of heavy metal oxidants with a catalytic ruthenium system reduces the toxicity of the effluent, simplifying wastewater treatment and lowering disposal costs. The process generates fewer solid wastes, such as salt cakes from neutralization steps, which are often difficult and expensive to handle. Moreover, the shorter reaction times and lower energy requirements contribute to a smaller carbon footprint for the manufacturing facility. These factors collectively enhance the environmental compliance profile of the production site, reducing regulatory risks and aligning with the increasingly stringent environmental standards imposed by global authorities and customers alike.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific experimental data and advantageous effects described in the patent documentation, providing clarity on the practical aspects of the technology. Understanding these details is essential for technical teams evaluating the feasibility of adopting this process for their own manufacturing lines or for sourcing partners who utilize this methodology.
Q: What are the primary advantages of using organozinc reagents over Grignard reagents in this synthesis?
A: Organozinc reagents offer superior chemoselectivity and milder reaction conditions compared to Grignard reagents. In the synthesis of methyl dihydrojasmonate, the use of allyl zinc bromide allows for a clean 1,4-Michael addition to the enone system without attacking the carbonyl group directly or causing polymerization, which significantly improves the purity profile and reduces the formation of complex byproducts often seen with harder nucleophiles.
Q: How does the ruthenium-catalyzed oxidation step impact the overall environmental footprint?
A: The use of catalytic ruthenium trichloride with sodium periodate as a co-oxidant represents a greener alternative to traditional stoichiometric oxidants like potassium permanganate or ozone. This system operates under mild aqueous conditions, generates less hazardous waste, and provides high atom economy by selectively cleaving the terminal alkene to a carboxylic acid while preserving the sensitive cyclopentanone ring structure, thereby simplifying downstream purification.
Q: Is the starting material 2-pentylcyclopentenone readily available for large-scale production?
A: Yes, the patent outlines a robust method for preparing 2-pentylcyclopentenone from commodity chemicals such as cyclopentanone and valeraldehyde. This ensures that the supply chain is not dependent on exotic or scarce intermediates. The ability to synthesize this key building block in-house from bulk petrochemical derivatives enhances supply security and allows for better cost control in the manufacturing of high-purity flavor and fragrance intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methyl Dihydrojasmonate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthesis routes in the competitive landscape of the flavor and fragrance industry. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volumetric demands of global clients with precision and consistency. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest international standards. By leveraging advanced technologies such as the organozinc-mediated synthesis described in CN101519355B, we are able to offer a superior product profile that balances performance with cost-effectiveness, making us a preferred partner for leading fragrance houses worldwide.
We invite you to engage with our technical procurement team to discuss how our capabilities can support your specific project requirements. Whether you are looking for a Customized Cost-Saving Analysis for your existing supply chain or require specific COA data and route feasibility assessments for new product development, we are ready to provide the detailed insights you need. Contact us today to explore how our innovative manufacturing solutions can drive value for your organization and secure your position in the market with reliable, high-quality chemical intermediates.
