Advanced Synthesis of 2-Amino-4,6-Dimethoxypyrimidine: A Commercial Scale-Up Breakthrough for Herbicide Intermediates
Introduction to Patent CN114230525B
The global demand for high-efficiency sulfonylurea herbicides continues to drive the need for robust and environmentally compliant manufacturing processes for their key intermediates. Patent CN114230525B, published in May 2022, introduces a transformative preparation method for 2-amino-4,6-dimethoxypyrimidine, a critical building block in agrochemical synthesis. This innovation addresses longstanding industrial pain points associated with traditional routes, specifically the reliance on hazardous chlorinating agents and the operational complexity of multi-step sequences. By leveraging a streamlined methanolysis strategy starting from malononitrile, the disclosed technology achieves a total yield exceeding 82% and product purity greater than 99%. For R&D directors and supply chain leaders, this patent represents a significant opportunity to optimize production lines, reduce wastewater treatment burdens, and secure a more reliable source of high-purity agrochemical intermediates. The following analysis dissects the technical merits and commercial implications of this novel synthetic pathway.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of 2-amino-4,6-dimethoxypyrimidine has been dominated by two primary methodologies, both of which suffer from significant drawbacks that impact cost and sustainability. The first conventional route, often depicted in legacy literature, utilizes guanidine nitrate and diethyl malonate, necessitating the use of phosphorus oxychloride (POCl3) as a chlorinating agent followed by sodium methoxide for methoxylation. As illustrated in the reaction scheme below, this process generates substantial amounts of colored phosphorus-containing wastewater, posing severe environmental risks and incurring high disposal costs. Furthermore, the handling of POCl3 introduces potential safety hazards related to corrosion and toxicity, leading to production halts in regions with stringent environmental regulations.
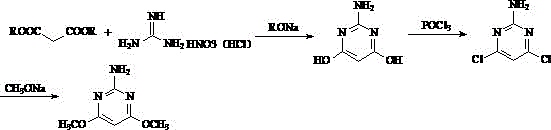
The second prevalent method involves a three-step sequence starting from malononitrile, proceeding through an imine salt intermediate, cyanamide substitution, and finally cyclization. While this route avoids phosphorus reagents, it imposes rigorous operational constraints, particularly the requirement for absolutely anhydrous conditions during the formation of the 1,3-dimethoxypropanediimine dihydrochloride intermediate. This intermediate is notoriously unstable in humid air and prone to hydrolysis into monoamide impurities, malonamide, and malonic acid. Consequently, the process demands complex filtration, separation, and drying steps, often resulting in a suboptimal total yield of approximately 65%. The sensitivity of the intermediate to moisture and temperature fluctuations creates bottlenecks in large-scale manufacturing, limiting throughput and increasing the risk of batch failures.
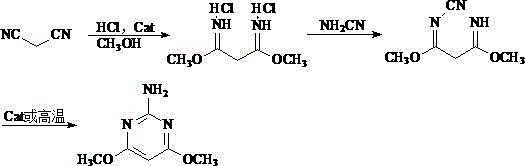
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN114230525B offers a streamlined, two-step reaction sequence that fundamentally simplifies the manufacturing landscape. By reacting malononitrile directly with methanol in the presence of hydrogen chloride gas, the process generates a hexamethoxypropane intermediate in situ, effectively bypassing the isolation of the unstable imine salt. This strategic modification eliminates the need for strict anhydrous environments and complex intermediate purification steps. The subsequent addition of a guanidine salt facilitates rapid cyclization to form the acid salt of the target pyrimidine, which is then neutralized to yield the final product. This approach not only shortens the synthetic route but also significantly reduces the volume of wastewater generated, as the solvent (methanol) is easily recoverable and the reaction byproducts are minimal. The result is a safer, more efficient, and economically superior process suitable for modern green chemistry standards.
Mechanistic Insights into Malononitrile Methanolysis and Cyclization
The core innovation of this patent lies in the precise control of reaction conditions during the initial methanolysis of malononitrile. The process employs a staged temperature protocol to manage the formation of intermediates and prevent side reactions. Initially, dry hydrogen chloride gas is introduced into anhydrous methanol at low temperatures (-5 to 10°C) to ensure complete dissolution and minimize volatilization losses. The malononitrile-methanol mixture is then added dropwise at 5-25°C, promoting the formation of an amidine salt intermediate. Following this, the temperature is carefully raised to 25-45°C to drive the conversion of the amidine salt into the stable 1,1,1,3,3,3-hexamethoxypropane species. This two-stage heating strategy is critical; maintaining lower temperatures initially prevents the premature degradation of malononitrile or the formation of polymeric byproducts, while the subsequent warming ensures complete conversion to the hexamethoxy derivative required for efficient cyclization.
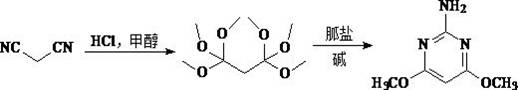
Following the formation of the hexamethoxypropane intermediate, the reaction mixture is filtered to remove ammonium chloride salts, a simple physical separation that avoids the need for solvent swaps or extractive workups. The filtrate is then treated with a guanidine salt, such as guanidine hydrochloride, nitrate, or carbonate, at moderate temperatures (5-25°C). The nucleophilic attack of the guanidine on the electrophilic centers of the hexamethoxypropane triggers the cyclization, closing the pyrimidine ring and forming the acid salt of 2-amino-4,6-dimethoxypyrimidine. The final step involves dissolving this acid salt in water and adjusting the pH to neutrality (7.0-10.0) using an inorganic base like aqueous ammonia or sodium hydroxide. This pH adjustment induces crystallization of the pure product, leveraging the solubility differences between the target molecule and residual impurities. The mechanism ensures high selectivity, minimizing the formation of open-chain urea byproducts common in less controlled environments.

How to Synthesize 2-Amino-4,6-Dimethoxypyrimidine Efficiently
The implementation of this synthesis route requires careful attention to the stoichiometry of reagents and the thermal profile of the reaction vessel to maximize yield and purity. The process begins with the preparation of a hydrogen chloride-methanol solution, followed by the controlled addition of malononitrile. Operators must monitor the temperature closely during the exothermic addition phase to prevent runaway reactions. Once the hexamethoxypropane intermediate is formed and ammonium chloride is removed, the addition of the guanidine component must be performed under stirring to ensure homogeneity. The detailed standardized operating procedures, including specific molar ratios, drop rates, and crystallization parameters, are essential for reproducing the >82% yield reported in the patent examples. For a comprehensive guide on executing this synthesis at scale, please refer to the structured protocol below.
- Prepare hydrogen chloride methanol solution and react with malononitrile at controlled temperatures (5-45°C) to form hexamethoxypropane intermediate.
- Filter ammonium chloride salts and add guanidine salt solution to the filtrate for cyclization reaction.
- Neutralize the acid salt with inorganic base, crystallize, and dry to obtain high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN114230525B offers tangible strategic advantages beyond mere technical elegance. The elimination of phosphorus oxychloride removes a major regulatory and safety liability, simplifying compliance with environmental protection laws and reducing the overhead associated with hazardous waste management. Furthermore, the use of malononitrile as a starting material leverages a widely available and cost-effective commodity chemical, insulating the supply chain from the volatility of specialized reagent markets. The simplification of the process flow, characterized by the removal of intermediate isolation and drying steps, translates directly into reduced cycle times and lower energy consumption. These factors collectively contribute to a more resilient and cost-efficient supply chain for agrochemical intermediates.
- Cost Reduction in Manufacturing: The new process achieves significant cost optimization by streamlining the synthetic sequence from three or more steps down to essentially two main reaction phases. By avoiding the use of expensive and hazardous chlorinating agents like phosphorus oxychloride, the method eliminates the need for specialized corrosion-resistant equipment and costly neutralization protocols for phosphorus waste. Additionally, the high total yield exceeding 82% means that less raw material is wasted per unit of product, directly improving the cost of goods sold. The ability to recover and reuse methanol solvent further enhances the economic viability of the process, making it highly competitive for large-scale commercial production.
- Enhanced Supply Chain Reliability: Traditional methods relying on unstable intermediates often suffer from batch-to-batch variability and yield fluctuations due to sensitivity to ambient humidity. The robust nature of the hexamethoxypropane intermediate in this new route ensures consistent product quality and reliable delivery schedules. Since the process does not require strictly anhydrous conditions for intermediate storage or transfer, it reduces the risk of production delays caused by equipment failure or environmental excursions. This stability allows manufacturers to maintain higher inventory levels of intermediates if necessary, providing a buffer against supply disruptions and ensuring continuous availability of the final active ingredient for downstream herbicide formulation.
- Scalability and Environmental Compliance: The environmental footprint of this synthesis is markedly smaller than that of conventional routes, primarily due to the drastic reduction in wastewater volume and the absence of toxic phosphorus contaminants. This aligns perfectly with global trends towards greener manufacturing and stricter discharge limits. The simplicity of the workup procedure, involving basic filtration and pH-adjusted crystallization, facilitates easy scale-up from pilot plants to multi-ton reactors without complex engineering modifications. The use of common inorganic bases for neutralization and the generation of benign salt byproducts simplify waste treatment, allowing facilities to operate with greater flexibility and reduced permitting hurdles in regulated jurisdictions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthesis method. Understanding these details is crucial for R&D teams evaluating the feasibility of technology transfer and for procurement specialists assessing supplier capabilities. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance to industrial applications.
Q: How does the new process improve upon the traditional phosphorus oxychloride route?
A: The new process eliminates the use of phosphorus oxychloride (POCl3), thereby removing significant safety hazards and the generation of colored phosphorus-containing wastewater, which drastically reduces environmental treatment costs.
Q: What are the yield and purity specifications achievable with this method?
A: According to patent data, the total yield can exceed 82% with product purity greater than 99%, surpassing the typical 65% yield of conventional cyanamide-based routes.
Q: Does this synthesis require strictly anhydrous conditions?
A: No, unlike previous methods requiring absolute anhydrous conditions for unstable imine salts, this method operates in methanol with controlled HCl gas, simplifying equipment requirements and operational complexity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Amino-4,6-Dimethoxypyrimidine Supplier
As the agrochemical industry evolves towards more sustainable and efficient manufacturing practices, the ability to adopt advanced synthetic routes becomes a key differentiator for market leaders. NINGBO INNO PHARMCHEM stands at the forefront of this transition, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is well-versed in the nuances of pyrimidine chemistry and is fully equipped to implement the high-yield, eco-friendly process described in CN114230525B. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of 2-amino-4,6-dimethoxypyrimidine meets the exacting standards required for sulfonylurea herbicide synthesis. Our commitment to quality and consistency makes us an ideal partner for long-term supply agreements.
We invite global partners to engage with our technical procurement team to discuss how this optimized manufacturing route can benefit your specific supply chain requirements. By collaborating with us, you can access a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating the economic impact of switching to this superior process. We encourage you to request specific COA data and route feasibility assessments to validate the performance metrics of our production capabilities. Let us help you secure a stable, high-quality supply of this critical intermediate while driving down costs and enhancing your environmental stewardship goals.
