Advanced Fmoc Solid-Phase Strategy for Commercial Telaprevir Manufacturing
Advanced Fmoc Solid-Phase Strategy for Commercial Telaprevir Manufacturing
The pharmaceutical industry continuously seeks robust, scalable, and cost-effective methodologies for synthesizing complex antiviral agents, particularly for the treatment of Hepatitis C Virus (HCV). Patent CN102875649B introduces a transformative approach to the preparation of Telaprevir, a potent reversible protease inhibitor, by leveraging Fmoc solid-phase synthesis techniques. This innovation addresses critical bottlenecks associated with traditional liquid-phase methods, offering a pathway to higher yields and simplified operational protocols. By utilizing a resin carrier to sequentially couple five specific substances, including a novel amino acid derivative, the process achieves a level of efficiency that is highly attractive for large-scale commercial production. The structural complexity of Telaprevir, characterized by its specific peptide backbone and cyclopropylamide moiety, demands precise stereochemical control, which this solid-phase strategy effectively delivers.
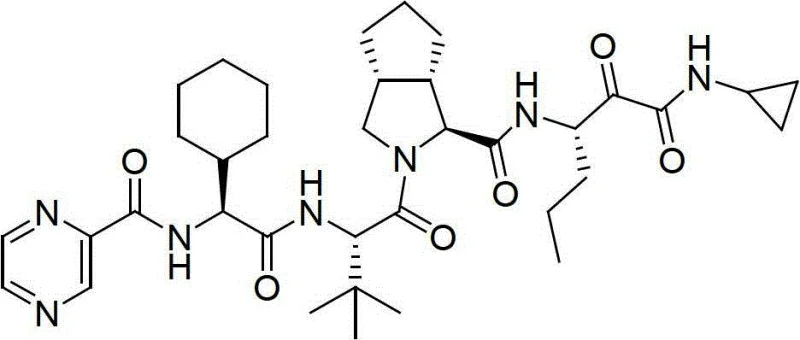
Telaprevir acts as a critical component in HCV therapy regimens, necessitating a supply chain capable of delivering high-purity intermediates and active pharmaceutical ingredients consistently. The conventional reliance on liquid-phase synthesis has historically imposed limitations regarding yield and equipment requirements, often necessitating high-pressure reactors and costly catalytic systems. In contrast, the methodology outlined in CN102875649B shifts the paradigm towards solid-phase peptide synthesis (SPPS), a technique renowned for its ability to drive reactions to completion through the use of excess reagents and facile washing steps. This transition not only enhances the purity profile of the resulting peptide resin but also streamlines the downstream processing required to isolate the final deprotected peptide. For R&D directors and process chemists, this represents a significant opportunity to optimize impurity profiles and reduce the burden of extensive purification steps typically associated with solution-phase peptide coupling.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Telaprevir has been dominated by liquid-phase methods, which, while effective on a small laboratory scale, present substantial challenges when translated to industrial manufacturing. These traditional routes often suffer from low overall yields due to the accumulation of byproducts at each coupling step, necessitating rigorous and costly purification interventions between stages. Furthermore, conventional liquid-phase protocols frequently require the use of expensive transition metal catalysts and specialized high-pressure equipment to facilitate difficult amide bond formations, thereby inflating capital expenditure and operational costs. The complexity of handling multiple protection and deprotection cycles in solution also increases the risk of racemization and epimerization, which can compromise the biological activity of the final drug substance. Additionally, the environmental footprint of liquid-phase synthesis is often larger due to the substantial volumes of organic solvents required for extraction and purification, posing challenges for waste management and regulatory compliance in an increasingly eco-conscious manufacturing landscape.
The Novel Approach
The novel approach detailed in patent CN102875649B circumvents these historical limitations by adopting an Fmoc solid-phase synthetic strategy anchored on a resin carrier. This method allows for the orderly coupling of five key substances: the novel amino acid Fmoc-P-OH, Fmoc-octahydrocyclopenta[c]pyrrole-1-carboxylic acid, Fmoc-L-tert-leucine, Fmoc-2-cyclohexylglycine, and 2-pyrazinecarboxylic acid. By immobilizing the growing peptide chain on a solid support, the reaction equilibrium is driven forward, significantly improving coupling efficiency and minimizing the formation of deletion sequences. The process operates under mild conditions, specifically at room temperature and atmospheric pressure, eliminating the need for energy-intensive high-pressure reactors. Moreover, the use of standard Fmoc chemistry ensures compatibility with a wide range of commercially available coupling reagents and resins, such as 2-CTC or King resin, facilitating easier technology transfer and scale-up. The final step involves a liquid-phase reaction with cyclopropylamine to install the critical warhead, combining the precision of solid-phase assembly with the flexibility of solution-phase functionalization.
Mechanistic Insights into Fmoc Solid-Phase Peptide Assembly
The core of this innovative synthesis lies in the preparation and utilization of the non-natural amino acid Fmoc-P-OH, which serves as the foundational building block attached to the resin. The synthesis of this key intermediate begins with a Henry reaction (nitroaldol condensation) between 1-nitrobutane and glyoxylic acid, catalyzed by triethylamine in methanol, to form a nitro-hydroxy intermediate. This intermediate is subsequently reduced, typically using hydrogen with a Pd/C catalyst or ammonium formate, to generate the corresponding amine. Protection of the amine with Fmoc-OSu yields a protected amino alcohol, which is then oxidized using IBX (2-iodoxybenzoic acid) to afford the target alpha-keto acid derivative, Fmoc-P-OH. This multi-step sequence is crucial for establishing the correct stereochemistry and functionality required for the subsequent peptide chain elongation.
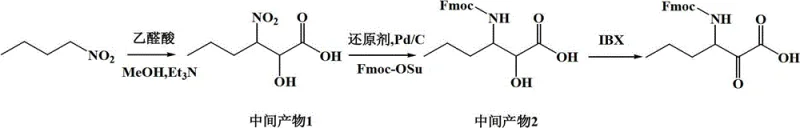
Once the Fmoc-P-OH is prepared, it is loaded onto the resin carrier, initiating the solid-phase assembly. The mechanism relies on the iterative cycle of Fmoc deprotection using piperidine, followed by activation of the incoming amino acid carboxyl group using coupling agents like DIPCDI, PyBOP, or HATU in the presence of additives like HOBt or HOAt. This activation generates a highly reactive species, such as an O-acylisourea or active ester, which rapidly reacts with the free amine on the resin-bound peptide. The use of 2-CTC resin allows for acid-labile cleavage under mild conditions, preserving sensitive side-chain protecting groups until the final global deprotection step. The sequential addition of the remaining four components—octahydrocyclopenta[c]pyrrole, tert-leucine, cyclohexylglycine, and pyrazine carboxylic acid—builds the peptidomimetic backbone with high fidelity. The final cleavage from the resin yields the deprotected peptide, which retains the integrity of the complex stereocenters established during the solid-phase synthesis.
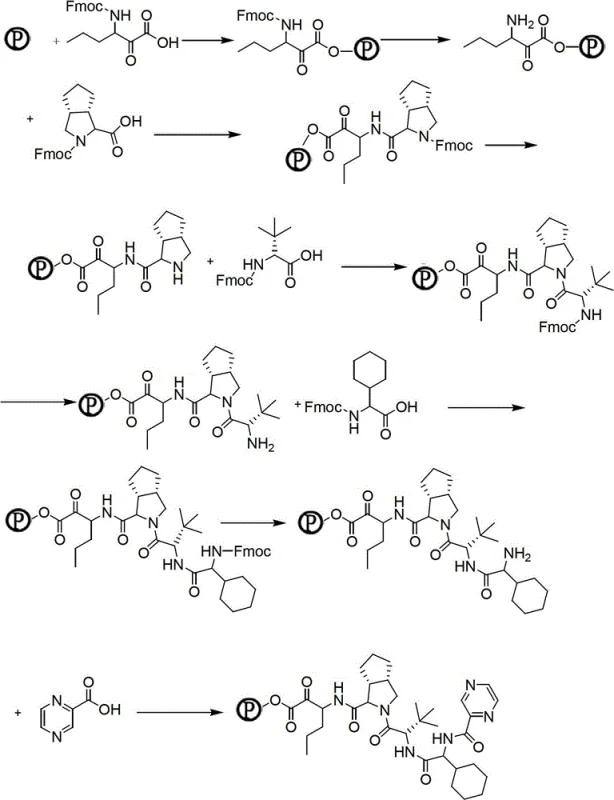
How to Synthesize Telaprevir Efficiently
The synthesis of Telaprevir via this patented route offers a streamlined workflow that integrates both solid-phase and liquid-phase techniques to maximize efficiency and purity. The process begins with the meticulous preparation of the resin-bound intermediate, followed by sequential coupling cycles that build the peptide chain with high precision. Each coupling step is monitored, often using the ninhydrin test, to ensure complete reaction before proceeding, thereby minimizing the propagation of impurities. After the full sequence is assembled on the solid support, the peptide is cleaved from the resin using specific reagent cocktails tailored to the resin type, such as TFE/DCM for 2-CTC resin or TFA-based mixtures for King resin. The resulting deprotected peptide is then isolated and subjected to the final amidation reaction with cyclopropylamine in a liquid-phase environment, utilizing EDC and NMM as coupling promoters to form the final ketoamide bond essential for protease inhibition.
- Preparation of the key non-natural amino acid Fmoc-P-OH via nitroaldol reaction, reduction, Fmoc protection, and oxidation.
- Sequential coupling of five specific substances onto a 2-CTC or King resin carrier using standard Fmoc solid-phase synthesis protocols.
- Cleavage of the peptide resin to obtain the deprotected intermediate, followed by reaction with cyclopropylamine to yield Telaprevir.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of the Fmoc solid-phase synthesis method described in CN102875649B presents compelling economic and logistical advantages over legacy manufacturing routes. The shift away from high-pressure equipment and expensive transition metal catalysts directly translates to a reduction in capital investment and operational overhead. By operating at ambient temperature and pressure, the process lowers energy consumption and reduces the safety risks associated with high-pressure hydrogenation or exothermic solution-phase reactions. Furthermore, the modular nature of solid-phase synthesis allows for greater flexibility in production scheduling and easier scaling from pilot batches to commercial tonnage without the need for extensive process re-engineering. The use of commercially available resins and standard coupling reagents ensures a stable supply of raw materials, mitigating the risk of shortages that can plague specialized catalyst markets.
- Cost Reduction in Manufacturing: The elimination of high-pressure reactors and precious metal catalysts significantly lowers the barrier to entry for manufacturing this complex API intermediate. The solid-phase approach minimizes solvent usage during intermediate workups, as impurities are simply washed away from the resin rather than requiring energy-intensive distillation or chromatography at every step. This reduction in solvent load and purification complexity leads to substantial cost savings in waste disposal and raw material procurement. Additionally, the higher yields associated with driving solid-phase reactions to completion reduce the cost of goods sold by maximizing the output from each batch of starting materials.
- Enhanced Supply Chain Reliability: Relying on standard Fmoc chemistry and widely available resin carriers like 2-CTC or King resin diversifies the supplier base for critical raw materials. Unlike proprietary catalysts that may be sourced from a single vendor, the reagents used in this process are commodity chemicals with robust global supply chains. This redundancy ensures continuity of supply even in the face of market fluctuations or geopolitical disruptions. The simplified operational protocol also reduces the dependency on highly specialized labor, allowing for more flexible staffing and training models within the manufacturing facility.
- Scalability and Environmental Compliance: The process is inherently scalable, as solid-phase peptide synthesis is a well-established technology in the pharmaceutical industry with proven track records for multi-kilogram production. The ability to perform reactions at room temperature reduces the thermal load on manufacturing plants, aligning with sustainability goals and reducing the carbon footprint of production. Moreover, the reduced generation of heavy metal waste from catalysts simplifies environmental compliance and wastewater treatment, making the facility more resilient to tightening environmental regulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel Telaprevir synthesis route. These insights are derived directly from the experimental data and process descriptions provided in patent CN102875649B, offering clarity on the practical aspects of adoption. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this method into their existing manufacturing portfolios.
Q: What are the primary advantages of using Fmoc solid-phase synthesis for Telaprevir over traditional liquid-phase methods?
A: The Fmoc solid-phase method described in patent CN102875649B eliminates the need for expensive catalysts and high-pressure equipment required in liquid-phase synthesis. It operates under mild conditions (room temperature and atmospheric pressure), significantly simplifying operations, improving yield, and reducing overall production costs while maintaining high purity standards.
Q: Which resin carriers are recommended for this specific Telaprevir synthesis route?
A: The patent specifically identifies 2-CTC resin (2-chlorotrityl chloride resin) and King resin as the preferred carriers. These resins offer active sites compatible with the carboxyl groups of the starting substances without interfering with other functional groups, ensuring efficient coupling and easy cleavage.
Q: How is the final purification of Telaprevir achieved in this process?
A: Purification is preferably achieved through recrystallization. This involves dissolving the crude Telaprevir in a benign solvent (such as methanol or ethyl acetate) and precipitating it by adding a poor solvent (such as n-hexane or water). This method effectively removes impurities to achieve high-purity specifications suitable for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Telaprevir Supplier
NINGBO INNO PHARMCHEM stands at the forefront of pharmaceutical intermediate manufacturing, possessing the technical expertise to translate complex patent methodologies like CN102875649B into robust commercial processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of Telaprevir intermediate meets the highest global regulatory standards. Our commitment to quality assurance ensures that our clients receive materials that are ready for immediate use in downstream API synthesis without the need for additional remediation.
We invite global pharmaceutical partners to engage with our technical procurement team to discuss how this advanced Fmoc solid-phase strategy can optimize your supply chain for Telaprevir. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential efficiencies of switching to this novel route. We encourage you to request specific COA data and route feasibility assessments to validate the performance of our materials against your internal benchmarks. Let us partner with you to secure a reliable, cost-effective, and high-quality supply of this critical antiviral intermediate.
