Advanced One-Step Synthesis of Chiral Oxazolines for High-Performance Asymmetric Catalysis
Advanced One-Step Synthesis of Chiral Oxazolines for High-Performance Asymmetric Catalysis
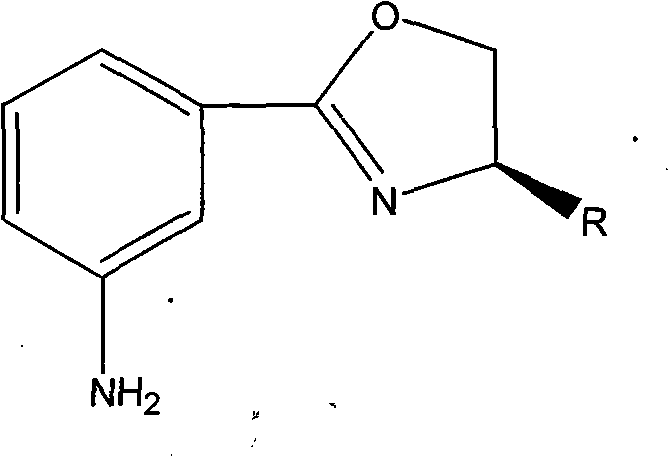
In the rapidly evolving landscape of asymmetric synthesis, the demand for robust, high-enantiopurity ligands has never been more critical for the development of next-generation active pharmaceutical ingredients (APIs). Patent CN102225915B introduces a significant technological advancement in this domain by detailing a novel, efficient synthesis method for a specific class of chiral oxazolines, chemically defined as 2-[(4R)-4,5-dihydro-4-R-3-oxazolinyl]phenylamine. These compounds serve as pivotal chiral ligands that, when complexed with metals, exhibit exceptional catalytic activity and enantioselectivity in fundamental organic transformations such as Diels-Alder cycloadditions, Michael condensations, and Henry reactions. The innovation lies not merely in the molecular structure but in the streamlined manufacturing process that bypasses traditional multi-step complexities, offering a direct route from readily available starting materials to high-value chiral intermediates.
The structural versatility of these oxazolines is defined by the variable 'R' group at the chiral center, which can be tailored to include isobutyl, sec-propyl, phenyl, or benzyl moieties depending on the specific stereochemical requirements of the downstream catalytic application. This adaptability makes them indispensable tools for medicinal chemists aiming to construct complex chiral architectures with precision. By leveraging a zinc chloride-catalyzed cyclization strategy, this technology addresses the persistent industry challenge of balancing high optical purity with economic feasibility, positioning these intermediates as a cornerstone for reliable chiral oxazoline supplier networks seeking to optimize their catalog offerings for the global pharmaceutical market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of chiral oxazoline ligands has often been plagued by synthetic inefficiencies that hinder large-scale adoption. Traditional routes frequently necessitate the use of expensive and moisture-sensitive reagents, such as acid chlorides or activated esters, which require stringent anhydrous conditions and cryogenic temperatures to prevent racemization or hydrolysis. Furthermore, conventional methodologies often involve multi-step sequences where the formation of the oxazoline ring is preceded by protection and deprotection strategies, leading to cumulative yield losses and increased waste generation. The reliance on precious metal catalysts or rare-earth elements in older protocols also introduces significant cost volatility and supply chain fragility, as the availability of these metals can be subject to geopolitical fluctuations. Additionally, purification of the final ligand from metal residues often requires specialized scavenging resins or extensive chromatography, adding substantial time and operational expenditure to the manufacturing timeline.
The Novel Approach
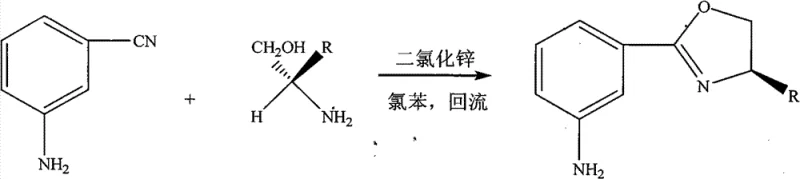
The methodology disclosed in the patent data revolutionizes this landscape by employing a direct condensation reaction between m-cyanoaniline and chiral amino alcohols, effectively collapsing the synthesis into a single, atom-economical step. This approach utilizes zinc chloride (ZnCl2) as a robust Lewis acid catalyst, which activates the nitrile group of the aniline derivative towards nucleophilic attack by the hydroxyl group of the amino alcohol. By operating at elevated temperatures between 110°C and 145°C in high-boiling solvents like chlorobenzene, the reaction drives the equilibrium towards the cyclic oxazoline product with impressive conversion rates. This thermal activation eliminates the need for cryogenic cooling or ultra-dry environments typically associated with sensitive organometallic chemistry, thereby simplifying the engineering controls required for production. The result is a process that not only achieves yields ranging from 65% to 75% but also drastically reduces the environmental footprint by minimizing solvent usage and eliminating hazardous by-products associated with activation reagents.
Mechanistic Insights into ZnCl2-Catalyzed Cyclization
The core of this synthetic breakthrough relies on the precise coordination chemistry facilitated by the zinc cation. In the proposed mechanism, the zinc chloride acts as a potent Lewis acid by coordinating with the nitrogen lone pair of the nitrile group on the m-cyanoaniline substrate. This coordination significantly increases the electrophilicity of the nitrile carbon, rendering it susceptible to nucleophilic attack. Simultaneously, the chiral amino alcohol, possessing both a nucleophilic hydroxyl group and an amine functionality, approaches the activated nitrile. The intramolecular cyclization proceeds through a transition state where the oxygen atom attacks the activated carbon, followed by proton transfer and subsequent ring closure to form the dihydro-oxazole ring system. The stereocenter present in the starting amino alcohol is preserved throughout this process, ensuring that the resulting oxazoline retains the high enantiomeric excess required for effective asymmetric induction in downstream catalytic applications.
From an impurity control perspective, the choice of zinc chloride is particularly strategic. Unlike stronger Lewis acids that might promote polymerization or decomposition of the sensitive amino alcohol side chains, zinc offers a "Goldilocks" level of acidity—strong enough to drive the reaction but mild enough to maintain chemoselectivity. The reaction conditions, specifically the use of chlorobenzene under reflux, ensure that any water generated during the condensation is effectively managed, either through azeotropic removal or by the hygroscopic nature of the catalyst system itself, preventing the hydrolysis of the newly formed oxazoline ring back to the starting materials. This mechanistic robustness translates directly to a cleaner crude reaction profile, reducing the burden on downstream purification units and ensuring that the final product meets the stringent purity specifications demanded by the fine chemical industry for use in GMP-regulated environments.
How to Synthesize 2-[(4R)-4,5-dihydro-4-R-3-oxazolinyl]phenylamine Efficiently
The practical implementation of this synthesis protocol is designed for seamless integration into existing pilot and production facilities. The process begins with the charging of anhydrous zinc chloride and the selected chiral amino alcohol into a reactor containing an inert organic solvent such as chlorobenzene or dichlorobenzene. The m-cyanoaniline is then introduced, and the mixture is heated to maintain a steady reflux, typically sustaining temperatures between 120°C and 140°C for a duration of 20 to 30 hours to ensure complete conversion. Following the reaction period, the mixture is cooled, and the solvent is removed under reduced pressure. The residue is subjected to a standard aqueous workup involving dissolution in water and extraction with organic solvents like chloroform, followed by drying over anhydrous sodium sulfate. Final purification is achieved through column chromatography using petroleum ether and methylene chloride mixtures, yielding the target chiral oxazoline as a colorless oil or solid with verified optical rotation values. For detailed standardized operating procedures and safety data, please refer to the technical guide below.
- Combine m-cyanoaniline and the selected chiral amino alcohol (such as D-leucinol or D-phenylalaninol) in an inert organic solvent like chlorobenzene.
- Add a catalytic amount of anhydrous zinc chloride (ZnCl2), typically 1-3 wt% relative to the reactants, to initiate the Lewis acid catalysis.
- Heat the mixture to reflux temperatures between 110°C and 145°C for 20 to 30 hours, followed by aqueous workup and column chromatography purification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this zinc-catalyzed synthesis route presents a compelling value proposition centered on cost stability and operational resilience. By shifting away from multi-step syntheses that rely on volatile precious metal catalysts or exotic reagents, manufacturers can significantly decouple their production costs from fluctuating commodity markets. The use of zinc chloride, a ubiquitous and inexpensive industrial chemical, replaces costly alternatives, leading to a substantial reduction in the raw material cost of goods sold (COGS). Furthermore, the simplification of the process flow—from multiple reaction vessels to a single reflux pot—drastically reduces utility consumption, including energy for heating and cooling, as well as labor hours required for intermediate monitoring and handling. This streamlining of the manufacturing workflow directly contributes to enhanced margin potential for downstream API producers who rely on these chiral building blocks.
- Cost Reduction in Manufacturing: The elimination of expensive activation reagents and the replacement of rare-earth catalysts with zinc chloride fundamentally alters the cost structure of chiral ligand production. This transition removes the need for specialized metal scavenging steps, which are often capital-intensive and time-consuming, thereby lowering the overall operational expenditure. The high atom economy of the direct condensation reaction ensures that a greater proportion of the input mass is converted into valuable product rather than waste, further optimizing the cost-per-kilogram metric. Additionally, the ability to use technical-grade solvents like chlorobenzene without the need for ultra-high purity grades reduces procurement overheads, allowing for significant cost reduction in pharmaceutical intermediates manufacturing without compromising on the quality of the final chiral catalyst.
- Enhanced Supply Chain Reliability: Sourcing reliability is markedly improved as the key starting materials, m-cyanoaniline and various chiral amino alcohols, are commodity chemicals produced by multiple global suppliers. This diversification of the supply base mitigates the risk of single-source bottlenecks that often plague the supply of specialized chiral reagents. The robustness of the reaction conditions, which tolerate minor variations in temperature and moisture better than cryogenic organometallic processes, ensures consistent batch-to-batch output even in facilities with varying levels of infrastructure maturity. Consequently, lead times for high-purity chiral oxazolines can be stabilized, providing downstream partners with the predictability necessary for just-in-time inventory management and long-term production planning.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing unit operations such as reflux and distillation that are standard in chemical processing plants worldwide, facilitating the commercial scale-up of complex pharmaceutical intermediates from kilogram to tonnage quantities. From an environmental standpoint, the reduction in synthetic steps correlates with a lower E-factor (mass of waste per mass of product), aligning with green chemistry principles and increasingly strict regulatory frameworks regarding solvent discharge and heavy metal contamination. The absence of toxic heavy metals in the catalyst system simplifies wastewater treatment protocols and reduces the regulatory burden associated with the disposal of metal-laden waste streams, making this technology a sustainable choice for environmentally conscious manufacturing operations.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this chiral oxazoline synthesis technology. These insights are derived directly from the experimental data and embodiments provided in the patent literature, offering clarity on catalyst selection, reaction parameters, and product characterization. Understanding these nuances is essential for process chemists aiming to replicate the high yields and enantioselectivity reported in the intellectual property documentation.
Q: What is the primary advantage of using ZnCl2 over rare-earth catalysts for this synthesis?
A: While the patent allows for rare-earth chlorides, zinc chloride (ZnCl2) is highlighted as a preferred transition metal catalyst due to its lower cost, widespread availability, and proven efficacy in driving the cyclization to high yields (up to 75%) without requiring exotic handling conditions.
Q: Can this synthesis method be scaled for industrial production?
A: Yes, the process utilizes standard reflux conditions in common solvents like chlorobenzene and avoids sensitive reagents, making it highly amenable to scale-up from laboratory grams to multi-ton commercial production batches.
Q: What represents the key structural variable in these chiral oxazolines?
A: The chirality and steric bulk are determined by the 'R' group at the 4-position of the oxazoline ring, which is derived directly from the side chain of the chiral amino alcohol starting material, such as isobutyl, sec-propyl, phenyl, or benzyl groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Oxazoline Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful deployment of advanced asymmetric catalysis relies on the consistent availability of high-quality chiral ligands. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to verify the optical rotation and chemical integrity of every batch of chiral oxazoline we produce. Our commitment to quality assurance means that our clients receive materials that are ready for immediate use in sensitive catalytic cycles, minimizing the risk of reaction failure due to impurity interference.
We invite global partners to collaborate with us to leverage this efficient synthesis technology for their specific drug discovery programs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our optimized process can lower your overall project costs. We encourage potential partners to contact us directly to obtain specific COA data for our chiral oxazoline inventory and to discuss route feasibility assessments for custom derivatives, ensuring that your supply chain is built on a foundation of technical excellence and commercial reliability.
