Optimizing Zolpidem Production Through Direct Hydrogenation And Catalytic Innovation
Optimizing Zolpidem Production Through Direct Hydrogenation And Catalytic Innovation
The pharmaceutical industry continuously seeks more efficient pathways for synthesizing critical active pharmaceutical ingredients and their precursors. Patent CN1729188A introduces a transformative approach to the synthesis of heteroaryl acetamides, specifically addressing the production of Zolpidem, a widely recognized sedative-hypnotic agent. This technology shifts the paradigm from multi-step conversion processes to a direct hydrogenation method, leveraging a specific combination of strong acids, halides, and noble metal catalysts. For R&D directors and technical procurement leaders, this represents a significant opportunity to streamline manufacturing workflows while maintaining stringent purity standards. The core innovation lies in the direct reduction of heteroaryl alpha-hydroxyacetamides, bypassing the need for intermediate activation steps that traditionally complicate the supply chain and increase production costs. By understanding the mechanistic advantages detailed in this patent, stakeholders can better evaluate the feasibility of integrating this route into their existing production lines for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of heteroaryl acetamides like Zolpidem has relied on complex multi-step sequences that introduce significant inefficiencies into the manufacturing process. Conventional methods, such as those disclosed in earlier literature, typically involve the reaction of imidazopyridine with dialkoxy alkylamides to generate an alpha-hydroxy acetamide intermediate. This intermediate must then be converted into an alpha-leaving group, often a halide or sulfonate, before a final reduction step can yield the desired acetamide. Each additional step in this sequence introduces potential points of failure, including yield loss, accumulation of impurities, and increased consumption of solvents and reagents. Furthermore, the handling of activated leaving groups often requires stringent safety protocols and specialized waste treatment, adding to the operational burden. For procurement managers, these complexities translate into higher raw material costs and longer lead times, as the supply chain must support a broader range of specialized chemicals. The cumulative effect of these limitations is a manufacturing process that is less robust and more susceptible to variability, posing risks to supply continuity for critical pharmaceutical products.
The Novel Approach
In stark contrast to traditional pathways, the novel approach described in the patent utilizes a direct hydrogenation strategy that significantly simplifies the synthetic route. By treating the heteroaryl alpha-hydroxyacetamide directly with hydrogen in the presence of a strong acid, a halide, and a hydrogenation catalyst, the process achieves the desired reduction in a single operational step. This method eliminates the need for isolating and activating intermediate leaving groups, thereby reducing the overall number of unit operations required. The use of a strong acid environment, combined with specific halide additives, creates a reaction milieu that facilitates the cleavage of the hydroxyl group while simultaneously preventing unwanted side reactions. For supply chain heads, this simplification means fewer material transfers, reduced equipment occupancy time, and a smaller footprint for chemical storage. The robustness of this direct conversion allows for more predictable batch cycles, enhancing the reliability of production schedules. Additionally, the ability to achieve high conversion rates without complex activation steps suggests a pathway that is inherently more cost-effective and environmentally sustainable, aligning with modern green chemistry principles.
Mechanistic Insights into Pd-Catalyzed Direct Hydrogenation
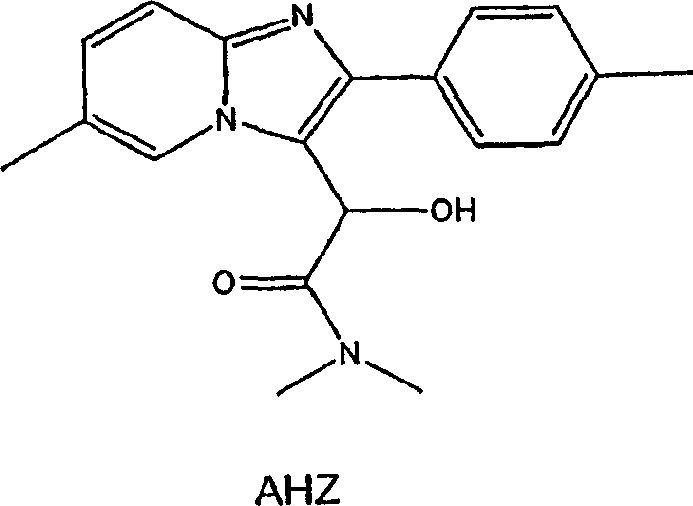
The success of this direct hydrogenation process relies heavily on the synergistic interaction between the noble metal catalyst, the strong acid medium, and the halide promoter. The catalyst, typically palladium supported on barium sulfate or carbon, serves as the active site for hydrogen activation. However, the presence of the strong acid, such as sulfuric acid, is crucial for protonating the hydroxyl group of the alpha-hydroxyacetamide, making it a better leaving group. Simultaneously, the halide ion, preferably bromide from sources like lithium bromide or sodium bromide, plays a pivotal role in stabilizing the reaction intermediate and preventing the reduction of sensitive carbon-nitrogen double bonds within the heteroaryl ring system. This selective reduction is vital for maintaining the structural integrity of the pharmacophore. Without the halide promoter, there is a risk of over-reduction or degradation of the heterocyclic core, which would compromise the purity of the final product. The careful balance of these components ensures that the reaction proceeds with high chemoselectivity, targeting only the benzylic hydroxyl group for reduction.
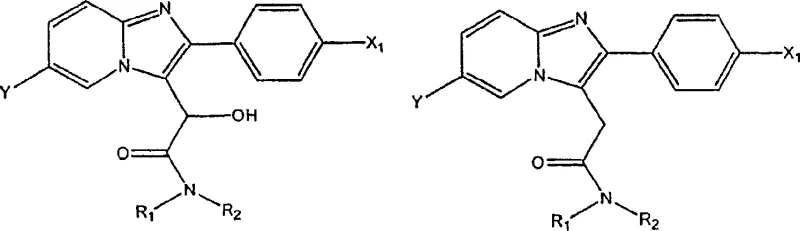
Furthermore, the choice of solvent and reaction conditions significantly influences the mechanistic pathway and the resulting impurity profile. The process is typically conducted in carboxylic acid solvents like glacial acetic acid, which not only solubilizes the reactants but also participates in the acid-base equilibrium of the system. The temperature range of 40 to 100 degrees Celsius, preferably around 70 to 75 degrees Celsius, provides sufficient thermal energy to drive the reaction without promoting thermal degradation. Pressure control, maintained between 1 to 4 atmospheres of hydrogen, ensures a steady supply of reducing equivalents to the catalyst surface. This precise control over reaction parameters allows for the minimization of by-products such as O-acetyl esters, which can form in acidic alcoholic media. By understanding these mechanistic nuances, R&D teams can optimize the process parameters to achieve consistent quality, ensuring that the final API intermediate meets the rigorous specifications required for downstream pharmaceutical formulation.
How to Synthesize Zolpidem Efficiently
Implementing this synthesis route requires a clear understanding of the operational parameters to ensure reproducibility and safety on a commercial scale. The process begins with the preparation of the alpha-hydroxy precursor, which is then subjected to the catalytic hydrogenation conditions described. The key to success lies in the precise addition of the halide promoter and the maintenance of the acidic environment throughout the reaction duration. Operators must monitor hydrogen uptake and temperature closely to prevent exothermic runaways, although the described conditions are generally mild enough to be managed with standard reactor control systems. The detailed standardized synthesis steps see the guide below for specific stoichiometric ratios and workup procedures.
- Prepare the reaction mixture by dissolving the heteroaryl alpha-hydroxyacetamide in a carboxylic acid solvent such as glacial acetic acid.
- Add a strong acid source like sulfuric acid and a halide source such as lithium bromide or sodium bromide to the solution.
- Introduce a noble metal catalyst like palladium on barium sulfate and conduct hydrogenation under controlled pressure and temperature.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this direct hydrogenation technology offers substantial advantages for procurement and supply chain management teams looking to optimize their sourcing strategies. The simplification of the synthetic route directly correlates with a reduction in manufacturing costs, as fewer processing steps mean lower labor, energy, and equipment utilization expenses. By eliminating the need for specialized reagents required to create leaving groups, the bill of materials becomes less complex and potentially less expensive. This efficiency gain allows suppliers to offer more competitive pricing structures without compromising on quality margins. For procurement managers, this translates into a more stable cost base for critical intermediates, shielding the organization from volatility in the prices of specialized synthetic reagents. The ability to source high-quality intermediates through a more efficient process enhances the overall value proposition of the supply partnership.
- Cost Reduction in Manufacturing: The elimination of intermediate activation steps significantly reduces the consumption of reagents and solvents, leading to lower variable costs per kilogram of product. By avoiding the use of expensive activating agents and the associated waste disposal costs, the overall cost of goods sold is drastically improved. This efficiency allows for better resource allocation and investment in quality control measures. The streamlined process also reduces the time required for batch completion, increasing the throughput capacity of existing manufacturing facilities without the need for capital expansion. Consequently, the economic efficiency of producing high-purity pharmaceutical intermediates is substantially enhanced, providing a competitive edge in the market.
- Enhanced Supply Chain Reliability: The use of common and readily available reagents such as acetic acid, sulfuric acid, and palladium catalysts minimizes the risk of supply disruptions associated with specialized chemicals. This robustness ensures that production schedules can be maintained even during periods of raw material scarcity. The simplified workflow reduces the number of potential bottlenecks in the manufacturing line, leading to more predictable lead times for order fulfillment. For supply chain heads, this reliability is crucial for maintaining inventory levels and meeting the just-in-time delivery requirements of downstream pharmaceutical manufacturers. The stability of the supply chain is further reinforced by the scalability of the process, which can be easily adapted to different production volumes.
- Scalability and Environmental Compliance: The process is designed to be scalable from laboratory to commercial production, utilizing standard hydrogenation equipment that is widely available in the fine chemical industry. The reduction in the number of chemical transformations also leads to a decrease in waste generation, aligning with increasingly stringent environmental regulations. By minimizing the use of halogenated solvents and toxic activating agents, the environmental footprint of the manufacturing process is significantly reduced. This compliance with green chemistry principles not only mitigates regulatory risks but also enhances the corporate social responsibility profile of the manufacturing partner. The ability to scale up complex pharmaceutical intermediates while maintaining environmental standards is a key differentiator in the global market.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived from the specific technical disclosures and experimental data provided in the patent documentation, ensuring accuracy and relevance for decision-makers. Understanding these details helps clarify the feasibility of adopting this route for commercial production.
Q: What is the primary advantage of the direct hydrogenation method for Zolpidem synthesis?
A: The primary advantage is the elimination of multiple intermediate steps required in conventional methods, such as the conversion to alpha-leaving groups, which significantly simplifies the process and improves overall yield.
Q: How does the addition of halides impact the reaction purity?
A: The addition of halides, specifically bromides, in the presence of strong acid helps prevent side reactions such as carbon-nitrogen double bond reduction, thereby maintaining high chemical purity of the final acetamide product.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes standard hydrogenation equipment and common reagents like acetic acid and palladium catalysts, making it highly adaptable for commercial scale-up from kilogram to metric ton quantities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Zolpidem Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and reliable synthesis routes for high-value pharmaceutical intermediates like Zolpidem. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize advanced analytical techniques to verify every batch. Our capability to implement complex catalytic hydrogenation processes allows us to offer clients a supply partner that truly understands the nuances of fine chemical manufacturing. By leveraging technologies such as the direct hydrogenation method, we can provide cost-effective solutions without compromising on the quality required for pharmaceutical applications.
We invite global partners to engage with us for a Customized Cost-Saving Analysis tailored to your specific production needs. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain. Whether you require pilot-scale quantities for clinical trials or metric tons for commercial launch, our infrastructure is designed to support your growth. Contact us today to discuss how we can collaborate to enhance the efficiency and reliability of your pharmaceutical intermediate supply.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
