Advanced Synthesis of Edoxaban Key Intermediates for Commercial Scale-up
Advanced Synthesis of Edoxaban Key Intermediates for Commercial Scale-up
The pharmaceutical landscape for anticoagulants continues to evolve with the demand for more efficient manufacturing processes for Factor Xa inhibitors like Edoxaban. Patent CN115724792A introduces a groundbreaking approach to synthesizing a critical Edoxaban intermediate, specifically targeting the optimization of the amide bond formation and stereochemical integrity. This technology addresses the longstanding challenges associated with the complex multi-step sequences traditionally required to assemble the chiral cyclohexyl core. By leveraging a novel ammonolysis strategy, the patent outlines a pathway that not only enhances chemical efficiency but also aligns with the rigorous purity standards demanded by global regulatory bodies. For procurement and supply chain leaders, this represents a pivotal shift towards more robust and cost-effective sourcing strategies for high-value pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Edoxaban and its precursors has been plagued by intricate reaction sequences that involve multiple protection and deprotection steps, often utilizing expensive chiral resolving agents. Prior art, such as the routes disclosed by Daiichi Sankyo in US2005119486A1 and WO2008156159A1, typically relies on the resolution of racemic mixtures or the use of heavily functionalized starting materials that require careful handling of sensitive groups like Boc and mesylates. These conventional pathways often suffer from cumulative yield losses, where each additional step introduces potential impurities and reduces the overall mass balance. Furthermore, the reliance on specific chiral auxiliaries or enzymatic resolutions can create bottlenecks in the supply chain, leading to increased lead times and volatility in raw material pricing. The complexity of these legacy routes also poses significant challenges for waste management and environmental compliance, as the generation of stoichiometric byproducts increases the burden on downstream purification processes.
The Novel Approach
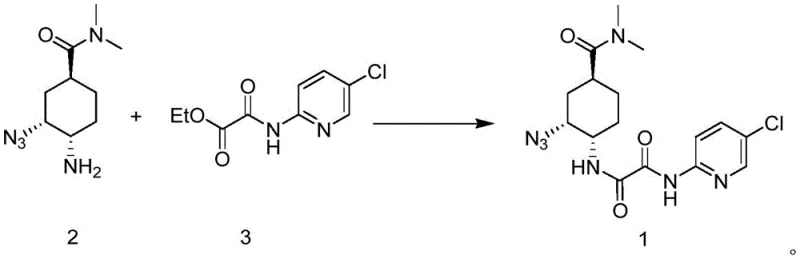
In stark contrast, the methodology presented in CN115724792A streamlines the construction of the key intermediate through a direct ammonolysis reaction between a chiral amine and an ethyl oxamate derivative. This innovative route eliminates the need for several intermediate isolation and protection steps, thereby collapsing the synthetic timeline and reducing the consumption of auxiliary reagents. The core transformation involves the nucleophilic attack of a chiral cyclohexyl amine on an activated ester, facilitated by mild basic conditions and optimized solvent systems. This direct coupling approach not only simplifies the operational workflow but also demonstrates superior yield profiles, with experimental data indicating yields exceeding 88% under optimized conditions. By minimizing the number of unit operations, this novel approach significantly lowers the risk of cross-contamination and simplifies the purification protocol, making it an ideal candidate for continuous manufacturing or large-batch production.
Mechanistic Insights into Ammonolysis and Stereochemical Control
The success of this synthetic route hinges on the precise control of the ammonolysis mechanism, where the nucleophilicity of the chiral amine must be balanced against the electrophilicity of the oxamate ester without compromising the stereocenters on the cyclohexyl ring. The reaction proceeds through a tetrahedral intermediate, where the leaving group (ethoxide) is expelled to form the stable amide bond. Critical to this process is the maintenance of reaction temperatures between 50°C and 100°C, which provides sufficient thermal energy to drive the reaction to completion while avoiding thermal degradation or epimerization of the chiral centers. The choice of solvent plays a pivotal role in stabilizing the transition state; polar aprotic solvents like acetonitrile or protic solvents like ethanol are employed to solubilize the reactants and facilitate proton transfer. Additionally, the stoichiometric ratio of the amine to the ester is carefully tuned, typically ranging from 1:1.1 to 1:1.5, to ensure complete consumption of the limiting reagent while minimizing the formation of bis-amide byproducts or unreacted starting materials.
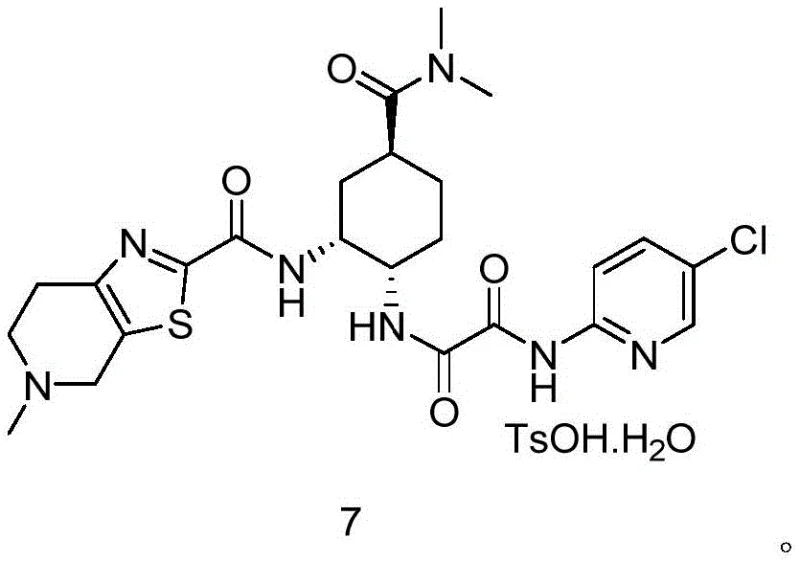
Impurity control is another cornerstone of this mechanistic design, particularly concerning the preservation of the (1S, 2R, 4S) configuration which is essential for biological activity. The mild reaction conditions prevent the inversion of configuration that might occur under harsher acidic or basic environments found in older synthetic routes. Furthermore, the downstream processing involves a straightforward aqueous workup where the product precipitates or can be extracted with high selectivity, leaving behind water-soluble impurities and salts. This inherent selectivity reduces the reliance on extensive chromatographic purification, which is often a cost-prohibitive step in large-scale manufacturing. The subsequent reduction of the azide group to an amine and the final coupling with the thiazole fragment are designed to be orthogonal to the existing functionality, ensuring that the integrity of the newly formed amide bond is maintained throughout the synthesis of the final API salt.
How to Synthesize Edoxaban Intermediate Efficiently
The practical implementation of this synthesis requires adherence to specific operational parameters to maximize yield and purity. The process begins with the preparation of the reaction mixture under controlled temperature conditions, followed by the gradual addition of reagents to manage exotherms. Detailed standard operating procedures emphasize the importance of monitoring reaction progress via HPLC to determine the optimal endpoint before quenching. The subsequent isolation steps utilize crystallization techniques that leverage the solubility differences between the product and impurities in mixed solvent systems. For a comprehensive guide on the exact reagent quantities, stirring rates, and drying protocols, please refer to the standardized synthesis steps outlined below.
- Perform ammonolysis between a chiral cyclohexyl amine and an ethyl oxamate derivative to form the key azido-intermediate with high stereochemical retention.
- Execute catalytic hydrogenation to reduce the azide group to an amine, followed by amide coupling with the thiazole-carboxylic acid fragment.
- Complete the synthesis by reacting the free base with p-toluenesulfonic acid in a controlled solvent system to crystallize the final monohydrate salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthetic route offers tangible strategic benefits that extend beyond simple chemical efficiency. The simplification of the process flow directly translates to a reduction in the number of raw materials required, thereby decreasing the complexity of vendor management and inventory control. By eliminating the need for specialized chiral resolving agents and complex protecting group reagents, manufacturers can source starting materials from a broader base of commodity chemical suppliers, enhancing supply security and reducing exposure to single-source risks. The robustness of the reaction conditions also implies a lower rate of batch failures, which is critical for maintaining consistent delivery schedules to downstream API manufacturers. This reliability is paramount in the pharmaceutical sector, where supply disruptions can have cascading effects on drug availability and regulatory compliance.
- Cost Reduction in Manufacturing: The streamlined nature of the ammonolysis route fundamentally alters the cost structure of Edoxaban intermediate production. By removing multiple synthetic steps, the process significantly reduces the consumption of solvents, energy, and labor hours associated with reactor occupancy. The elimination of expensive chromatography columns and specialized reagents further drives down the variable costs per kilogram. Moreover, the high yield achieved in the key coupling step minimizes the loss of valuable chiral starting materials, ensuring that the maximum amount of input mass is converted into saleable product. This efficiency gain allows for a more competitive pricing model without compromising on the stringent quality specifications required for pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The reliance on readily available and stable starting materials, such as ethyl oxamate derivatives and chiral amines, mitigates the risk of supply chain bottlenecks. Unlike routes that depend on bespoke catalysts or unstable intermediates, this method utilizes commodity chemicals that are produced at scale by multiple global suppliers. This diversification of the supply base ensures continuity of supply even in the face of regional disruptions or market fluctuations. Additionally, the simplified workflow reduces the lead time from raw material intake to finished goods, enabling manufacturers to respond more agilely to changes in market demand and inventory levels.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this process is designed for seamless transition from pilot scale to commercial production. The use of common solvents like ethanol and acetonitrile facilitates solvent recovery and recycling, aligning with green chemistry principles and reducing the environmental footprint of the manufacturing site. The reduction in waste generation, particularly the avoidance of heavy metal catalysts or toxic byproducts, simplifies waste treatment protocols and lowers disposal costs. This environmental compatibility not only supports corporate sustainability goals but also ensures compliance with increasingly stringent global regulations regarding pharmaceutical manufacturing emissions and effluent discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational feasibility and strategic value of the new route for industry stakeholders. Understanding these nuances is essential for making informed decisions about technology transfer and long-term sourcing partnerships.
Q: How does the new ammonolysis route improve upon conventional Edoxaban synthesis?
A: The novel route described in CN115724792A bypasses complex resolution steps and multiple protection-deprotection cycles found in prior art, utilizing a direct ammonolysis reaction that significantly simplifies the process flow and improves overall yield.
Q: What are the critical parameters for maintaining chirality during the intermediate synthesis?
A: Maintaining strict temperature control between 50°C and 100°C and using specific solvent systems like acetonitrile or ethanol are crucial to preventing racemization while ensuring high conversion rates of the chiral amine starting material.
Q: Is this synthetic method suitable for large-scale industrial production?
A: Yes, the method employs readily available reagents, mild reaction conditions, and standard workup procedures such as filtration and crystallization, making it highly adaptable for commercial scale-up from pilot plants to multi-ton manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Edoxaban Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic methodologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has extensively evaluated the route described in CN115724792A and possesses the technical capability to execute this process with precision and consistency. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with unwavering reliability. Our facilities are equipped with state-of-the-art rigorous QC labs and analytical instruments to guarantee stringent purity specifications for every batch of Edoxaban intermediate we produce, adhering to the highest international standards.
We invite you to collaborate with us to leverage this innovative technology for your supply chain. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to reach out to us to request specific COA data and route feasibility assessments that demonstrate how our optimized manufacturing capabilities can enhance your project's bottom line. Let us be your partner in delivering high-quality pharmaceutical intermediates that drive the success of your final drug products.
