Advanced Synthesis of Edoxaban Free Base Intermediates for Commercial Scale-up
Advanced Synthesis of Edoxaban Free Base Intermediates for Commercial Scale-up
The pharmaceutical industry continuously seeks robust, scalable, and safe pathways for the production of critical anticoagulant agents like edoxaban. Patent CN111763222B introduces a significant technological breakthrough in the synthesis of edoxaban free base, specifically focusing on a novel intermediate strategy that addresses long-standing challenges in impurity control and process safety. This innovation centers on the preparation of a specific activated ester intermediate, denoted as compound (II), which serves as a pivotal building block for the final API. By shifting away from traditional coupling reagents and heavy metal catalysis, this method offers a streamlined route that enhances both the chemical purity and the environmental profile of the manufacturing process. The structural integrity of the target molecule, edoxaban free base (I), and its pharmaceutically active salt form (I-a), is maintained with high fidelity throughout this optimized synthetic sequence.
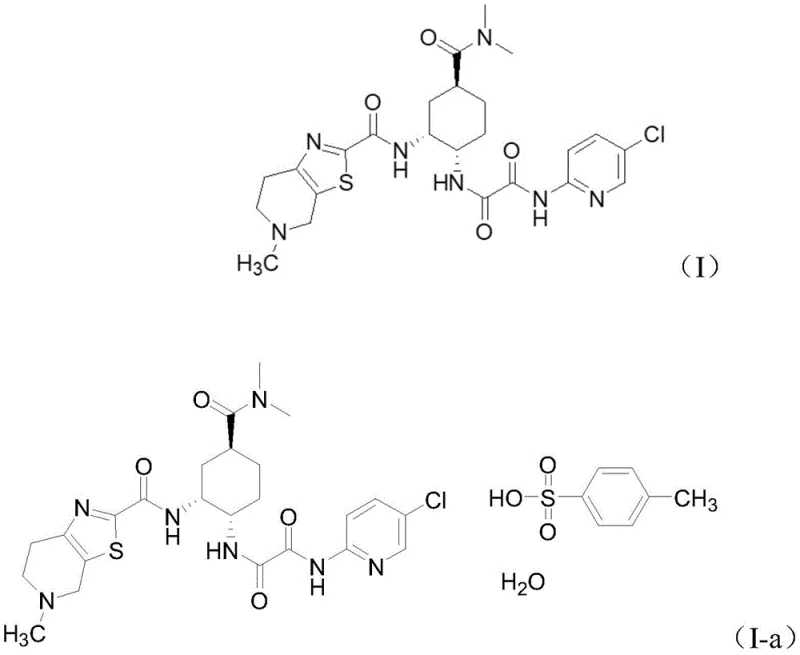
For procurement specialists and supply chain managers evaluating reliable edoxaban intermediate supplier options, understanding the underlying chemistry is crucial for assessing long-term viability. The patent details a method where 5-methyl-4,5,6,7-tetrahydro[1,3]thiazolo[5,4-c]pyridine-2-carboxylic acid is converted into a reactive intermediate using acid chlorides. This strategic modification allows for a subsequent coupling reaction with the complex cyclohexyl amine fragment under mild conditions. The result is a process that not only improves yield consistency but also drastically simplifies the downstream processing requirements, making it a highly attractive candidate for cost reduction in pharmaceutical intermediates manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of edoxaban and its precursors has relied on methodologies that present significant operational and safety hurdles. One common approach involves the use of carbodiimide coupling reagents such as EDC (1-(3-dimethylaminopropyl)-3-ethylcarbodiimide) alongside additives like HOBt (1-hydroxybenzotriazole). While chemically effective on a small scale, this route generates substantial quantities of difficult-to-remove urea byproducts and poses serious toxicological concerns due to the potential genotoxicity of residual EDC. Furthermore, the wastewater generated from washing away these reagents places a heavy burden on environmental treatment facilities, as HOBt is notoriously resistant to biochemical degradation.
Another prior art method, disclosed in patents such as CN201580020396.9, utilizes palladium-catalyzed carbonylation. This technique requires the handling of carbon monoxide, a colorless and highly toxic gas that demands rigorous safety protocols and specialized equipment to prevent leaks and explosions. Additionally, the use of heavy metal palladium catalysts introduces the risk of metal residues in the final API, necessitating expensive and time-consuming purification steps to meet stringent regulatory limits. The combination of hazardous gases, expensive catalysts, and phosphorus-containing waste streams makes these conventional routes less desirable for modern, green chemical manufacturing.
The Novel Approach
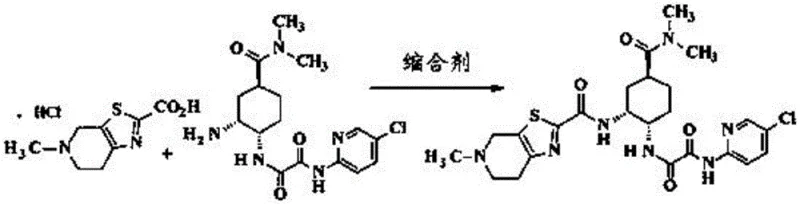
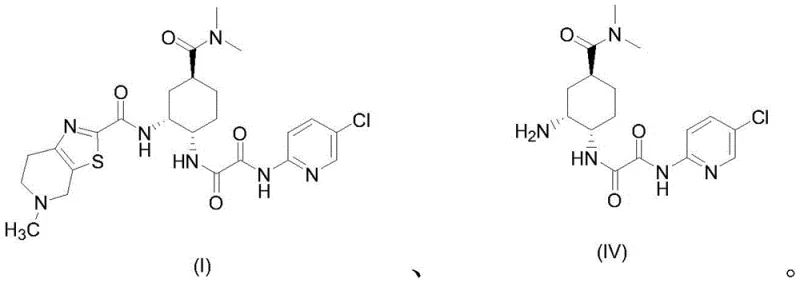
In stark contrast, the methodology described in CN111763222B employs an acid chloride activation strategy that circumvents these issues entirely. By reacting the thiazolopyridine carboxylic acid with reagents like pivaloyl chloride or tosyl chloride in the presence of a base, a highly reactive mixed anhydride or activated ester intermediate (II) is formed in situ. This activated species then couples efficiently with the amine component (IV) to form the target amide bond. This approach eliminates the need for genotoxic carbodiimides and avoids the use of palladium and carbon monoxide altogether. The reaction proceeds smoothly at low temperatures, typically between -40°C and -10°C, ensuring high selectivity and minimizing side reactions.
The operational simplicity of this novel route is a key differentiator for commercial scale-up of complex pharmaceutical intermediates. Unlike previous methods that might require column chromatography for purification, this process allows for the isolation of high-purity edoxaban free base through simple aqueous workups and concentration. Experimental data from the patent indicates that this method can achieve HPLC purities exceeding 99% with molar yields ranging from approximately 80% to over 90%, depending on the specific acid chloride and conditions used. This level of efficiency directly translates to reduced material costs and shorter production cycles, providing a compelling value proposition for high-purity pharmaceutical intermediates.
Mechanistic Insights into Acid Chloride Activation and Coupling
The core of this synthetic innovation lies in the controlled activation of the carboxylic acid moiety on the thiazolopyridine ring. In the first stage, the carboxylic acid (III) reacts with an acid chloride (such as pivaloyl chloride) in the presence of a tertiary amine base like triethylamine or pyridine. This reaction generates an activated acylating agent, likely a mixed anhydride or an acyl pyridinium species, which is significantly more electrophilic than the parent acid. This activation step is critical and is performed at low temperatures (-40 to -10°C) to prevent the decomposition of the reactive intermediate and to suppress potential racemization or side reactions on the sensitive heterocyclic system.
Once the activated intermediate (II) is generated, it is introduced to the solution containing the amine partner (IV). The nucleophilic attack of the amine nitrogen on the activated carbonyl carbon proceeds rapidly under these mild conditions to form the desired amide bond. The choice of base and solvent plays a vital role in facilitating this transformation while neutralizing the hydrochloric acid byproduct. Common solvents include dichloromethane, ethyl acetate, or acetonitrile, which provide excellent solubility for both reactants. The mechanistic elegance of this route ensures that the stereochemistry of the cyclohexyl ring and the thiazolopyridine system is preserved, resulting in a product with a clean impurity profile that is essential for regulatory approval.
How to Synthesize Edoxaban Intermediate Efficiently
The practical execution of this synthesis involves a straightforward two-vessel protocol that is well-suited for industrial implementation. The process begins with the preparation of the activated acid solution (Solution A) under inert atmosphere, followed by the preparation of the amine solution (Solution B). These solutions are then combined under strict temperature control to drive the coupling reaction to completion. The detailed procedural steps below outline the specific conditions and stoichiometry required to replicate the high yields reported in the patent examples.
- Activate the carboxylic acid precursor with pivaloyl chloride or tosyl chloride in an organic solvent at low temperatures (-40 to -10°C) using a base like triethylamine.
- Prepare the amine component (Compound IV) in a separate vessel, optionally as a salt, and dissolve in an organic solvent with base.
- Combine the activated acid solution with the amine solution at controlled low temperatures, stir for 1-2 hours, and isolate the product via aqueous workup and concentration without column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For decision-makers focused on the bottom line and supply continuity, the shift to this acid chloride-mediated pathway offers substantial strategic benefits. The elimination of expensive and hazardous reagents fundamentally alters the cost structure of the manufacturing process. By removing the dependency on palladium catalysts and specialized high-pressure equipment for carbon monoxide handling, capital expenditure and raw material costs are significantly reduced. Furthermore, the simplified purification workflow means that production throughput can be increased without the bottleneck of complex chromatographic separations.
- Cost Reduction in Manufacturing: The replacement of costly coupling reagents like EDC and HOBt with commodity acid chlorides results in direct material savings. Additionally, the avoidance of heavy metal catalysts removes the need for expensive scavenging resins and extensive metal testing, further lowering the cost of goods sold. The high crude purity achieved allows for simpler crystallization or washing steps, reducing solvent consumption and energy usage associated with distillation and drying.
- Enhanced Supply Chain Reliability: The reagents required for this process, such as pivaloyl chloride, tosyl chloride, and triethylamine, are widely available bulk chemicals with stable supply chains. This contrasts sharply with specialized ligands or catalysts that may have limited suppliers. The robustness of the reaction conditions also means that the process is less susceptible to batch failures due to minor variations in raw material quality, ensuring consistent delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this route is superior. It eliminates the generation of genotoxic waste and phosphorus-containing effluents, simplifying wastewater treatment compliance. The absence of carbon monoxide gas removes a major safety hazard from the plant floor. These factors make the technology easier to scale from pilot plants to multi-ton commercial production without requiring massive upgrades to safety infrastructure or waste treatment capabilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and technical specifications provided in the patent documentation, offering clarity on the feasibility and advantages of the technology.
Q: How does this new method improve drug safety compared to conventional EDC/HOBt coupling?
A: Conventional methods using EDC and HOBt introduce potential genotoxic impurities that are difficult to remove and create hazardous wastewater. This novel acid chloride activation method eliminates these reagents entirely, significantly reducing toxicological risks and simplifying purification.
Q: Does this process avoid the use of expensive heavy metal catalysts?
A: Yes, unlike palladium-catalyzed carbonylation routes which require expensive Pd catalysts and dangerous carbon monoxide gas, this method relies on standard organic acylation chemistry, removing heavy metal residue concerns and improving production safety.
Q: Is the final purification step suitable for industrial scale-up?
A: Absolutely. The process avoids complex column chromatography. The final product is isolated through simple aqueous washing and concentration, yielding high-purity material (over 99% HPLC purity in examples), which is ideal for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Edoxaban Intermediate Supplier
As the global demand for anticoagulants continues to rise, securing a supply of high-quality intermediates is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced synthetic technologies like the one described in CN111763222B to deliver superior products. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of major pharmaceutical companies. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of edoxaban intermediate meets the highest international standards for safety and efficacy.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in green chemistry can drive value for your organization.
