Advanced Synthetic Route for Bioactive 3,4,5-Substituted Benzodiazepine-2-One Derivatives
Introduction to Patent CN107879989B and Technological Breakthroughs
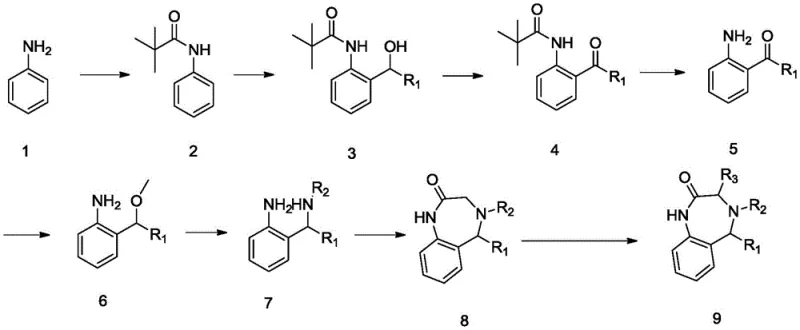
The pharmaceutical landscape is constantly evolving with the demand for novel scaffolds that can address resistant cancer strains, and patent CN107879989B presents a significant advancement in this domain by disclosing a series of 3,4,5-substituted benzodiazepine-2-one drug molecules. This intellectual property outlines a comprehensive preparation method that not only ensures the structural integrity of the complex heterocyclic core but also optimizes the substitution patterns at the 3, 4, and 5 positions to maximize biological efficacy. The invention specifically targets the synthesis of derivatives where R1 can be methyl, pyridyl, phenyl, or isopropyl, while R2 and R3 are independently selected from methyl, ethyl, or phenyl groups, creating a diverse library of potential therapeutic agents. By leveraging a strategic combination of protection group chemistry and directed metalation, the patent provides a robust pathway that addresses the historical challenges of regioselectivity often encountered in benzodiazepine synthesis. This technological breakthrough is particularly relevant for R&D teams focused on oncology, as preliminary data indicates strong inhibitory effects on K562 leukemia and MCF-7 breast cancer cell lines, positioning these intermediates as high-value assets for drug discovery pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for benzodiazepine derivatives often suffer from significant drawbacks related to poor regiocontrol and harsh reaction conditions that can compromise yield and purity. Conventional methods frequently rely on non-selective alkylation or acylation steps that result in complex mixtures of isomers, necessitating extensive and costly purification processes such as preparative HPLC or multiple recrystallizations. Furthermore, many existing protocols utilize sensitive reagents that require stringent anhydrous conditions or cryogenic temperatures throughout the entire sequence, which poses substantial challenges for scale-up in a commercial manufacturing environment. The lack of a robust protecting group strategy in older methods often leads to side reactions at the amine functionality during the critical ring-closing steps, resulting in lower overall throughput and increased waste generation. These inefficiencies translate directly into higher production costs and longer lead times, making it difficult for procurement managers to secure reliable supplies of high-purity intermediates for clinical trials.
The Novel Approach
In contrast, the methodology described in CN107879989B introduces a streamlined approach that utilizes pivaloyl chloride for amine protection, effectively masking the reactive nitrogen center during the initial carbon-carbon bond-forming events. This strategic protection allows for the precise installation of substituents via n-butyllithium mediated addition to aldehydes, ensuring that the desired substitution pattern is established early in the synthesis with high fidelity. The subsequent oxidation and deprotection steps are conducted under controlled conditions using accessible metal oxidants like manganese dioxide, which simplifies the operational complexity compared to more exotic catalytic systems. By decoupling the ring formation from the initial substitution steps, this novel route minimizes the formation of byproducts and allows for easier isolation of intermediates, thereby enhancing the overall efficiency of the process. This modular design not only improves the chemical yield but also offers greater flexibility for generating structural analogs, making it an ideal platform for medicinal chemists exploring structure-activity relationships.
Mechanistic Insights into Directed Ortho-Metalation and Cyclization
The core mechanistic innovation of this synthesis lies in the use of directed ortho-metalation (DoM) facilitated by the pivaloyl amide group, which acts as a powerful directing group for the n-butyllithium species. Upon treatment with n-BuLi at low temperatures, the lithiation occurs selectively at the ortho-position relative to the amide, generating a highly reactive aryl lithium intermediate that subsequently undergoes nucleophilic addition to the chosen aldehyde (R1-CHO). This step is critical as it establishes the benzylic alcohol functionality which is later oxidized to the ketone, setting the stage for the eventual cyclization. The precision of this metalation step is paramount, as it avoids the random substitution patterns that plague electrophilic aromatic substitution methods, ensuring that the R1 group is positioned exactly where it is needed for the final biological activity. Following the addition, the reaction is quenched and worked up to yield the protected amino-alcohol, which serves as the foundation for the subsequent oxidative transformations.
Following the establishment of the carbon skeleton, the synthesis proceeds through a carefully orchestrated sequence of oxidation and cyclization events to construct the seven-membered diazepine ring. The benzylic alcohol is oxidized to a ketone using metal oxides, and the pivaloyl group is removed under acidic conditions to reveal the free amine, which is then methylated to prevent unwanted side reactions. The key ring-closing step involves the reaction of the amino-ketone intermediate with chloroacetyl chloride in the presence of a base like sodium hydride, which promotes the intramolecular nucleophilic attack of the amine onto the acid chloride. This cyclization forms the characteristic benzodiazepine-2-one core, and the final alkylation at the 3-position using LDA and alkyl halides completes the molecular architecture. This mechanistic pathway ensures that the stereochemistry and substitution patterns are maintained throughout the synthesis, resulting in a final product with the specific structural features required for potent antitumor activity.
How to Synthesize 3,4,5-Substituted Benzodiazepine-2-One Efficiently
The synthesis of these bioactive molecules requires strict adherence to the optimized reaction conditions outlined in the patent to ensure maximum yield and purity. The process begins with the protection of aniline, followed by the critical lithiation and addition steps which must be monitored closely by TLC to prevent over-reaction or decomposition of the sensitive organolithium species. Detailed standardized operating procedures for each transformation, including specific solvent choices like anhydrous THF and precise temperature controls during the addition of reagents, are essential for reproducibility. For a complete breakdown of the experimental parameters, stoichiometry, and workup procedures required to execute this synthesis successfully, please refer to the technical guide below.
- Protect aniline with pivaloyl chloride, followed by n-butyllithium mediated addition to an aldehyde to form the alcohol intermediate.
- Oxidize the alcohol to a ketone using metal oxides like MnO2, then remove the protecting group under acidic conditions.
- Perform methylation and amine substitution, followed by cyclization with chloroacetyl chloride to close the seven-membered ring.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the synthetic route disclosed in CN107879989B offers substantial benefits for supply chain stability and cost management in the production of pharmaceutical intermediates. The reliance on commodity chemicals such as aniline, pivaloyl chloride, and chloroacetyl chloride means that raw material sourcing is straightforward and less susceptible to the volatility associated with specialized catalysts or rare earth metals. This accessibility translates into a more resilient supply chain, reducing the risk of production delays caused by material shortages and allowing for better long-term planning for large-scale campaigns. Furthermore, the high yields reported in the examples, such as the 83% yield achieved in Example 4 for the oxidation step, indicate a material-efficient process that minimizes waste and maximizes the output per batch, directly contributing to lower unit costs.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of standard oxidants like manganese dioxide significantly lower the direct material costs associated with the synthesis. By avoiding complex catalytic cycles that require rigorous removal of metal residues to meet pharmaceutical standards, the downstream purification burden is drastically reduced, leading to further savings in processing time and solvent consumption. The modular nature of the synthesis also allows for the optimization of individual steps without compromising the overall sequence, enabling continuous process improvements that drive down manufacturing expenses over time.
- Enhanced Supply Chain Reliability: The use of robust and well-understood chemical transformations ensures that the process can be reliably transferred between different manufacturing sites or scaled up from pilot plant to commercial production without significant re-engineering. The stability of the intermediates and the tolerance of the reaction conditions to minor variations in temperature or mixing rates provide a safety margin that enhances operational reliability. This consistency is crucial for maintaining a steady supply of high-purity intermediates to downstream drug manufacturers, ensuring that clinical trial timelines and commercial launch schedules are met without interruption.
- Scalability and Environmental Compliance: The synthetic pathway is designed with scalability in mind, utilizing solvents and reagents that are compatible with standard industrial equipment and waste treatment protocols. The avoidance of highly toxic or environmentally hazardous reagents simplifies the regulatory compliance landscape, reducing the costs and administrative burden associated with environmental health and safety permits. Additionally, the high atom economy of the key coupling and cyclization steps minimizes the generation of chemical waste, aligning the manufacturing process with modern green chemistry principles and sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these benzodiazepine derivatives, based on the detailed disclosures within the patent documentation. Understanding these aspects is vital for stakeholders evaluating the feasibility of integrating this technology into their existing development pipelines. The answers provided reflect the specific experimental data and structural claims made in the original filing.
Q: What is the primary biological activity of these benzodiazepine derivatives?
A: According to patent CN107879989B, these compounds exhibit significant antitumor activity, specifically showing inhibition against human leukemia cell lines (K562) and breast cancer cells (MCF-7).
Q: What are the key advantages of this synthetic route over conventional methods?
A: The process utilizes a pivaloyl protection strategy combined with directed ortho-lithiation, which enhances regioselectivity and simplifies the construction of the substituted benzene ring prior to cyclization, leading to higher purity intermediates.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the synthesis employs common reagents such as chloroacetyl chloride, dimethyl sulfate, and standard metal oxidants, avoiding exotic catalysts, which facilitates scalability and cost-effective production for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzodiazepine-2-One Supplier
As a leader in the fine chemical industry, NINGBO INNO PHARMCHEM is uniquely positioned to support the development and commercialization of these advanced 3,4,5-substituted benzodiazepine-2-one intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical clients. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the highest quality standards required for oncology drug development. Our commitment to technical excellence allows us to navigate the complexities of heterocyclic chemistry with precision, delivering materials that accelerate your research and production timelines.
We invite you to collaborate with us to leverage this innovative synthetic route for your next-generation anticancer therapies. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain and reduce overall project costs.
