Advanced Catalytic Synthesis of Ornidazole: A Strategic Upgrade for Commercial Manufacturing
The pharmaceutical industry constantly seeks robust synthetic pathways that balance high purity with economic efficiency, particularly for established anti-infective agents like Ornidazole. Patent CN101633643A introduces a transformative approach to the synthesis of this critical nitroimidazole derivative, addressing long-standing inefficiencies in traditional manufacturing protocols. By leveraging a specific Lewis acid catalytic system involving Boron trifluoride diethyl etherate, this innovation achieves reaction yields exceeding 85%, a substantial improvement over the sub-60% yields typical of prior art methods. This technical breakthrough is not merely a laboratory curiosity but represents a viable strategy for cost reduction in pharmaceutical intermediates manufacturing, offering a streamlined route that minimizes waste and maximizes throughput. For R&D directors and procurement specialists alike, understanding the mechanistic nuances of this pathway is essential for securing a reliable ornidazole intermediate supplier capable of meeting rigorous global demand.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Ornidazole has been plagued by inefficient coupling reactions between the imidazole core and the chlorohydrin side chain. Traditional methods, such as those disclosed in earlier patents like CN1923817A, often relied on less effective catalytic systems or harsh conditions that resulted in yields generally lower than 60%. These low-yield processes necessitate the consumption of excessive amounts of starting materials, specifically 2-methyl-5-nitroimidazole and epichlorohydrin, leading to inflated raw material costs and significant environmental burdens due to waste generation. Furthermore, many conventional routes involve complex multi-step sequences or require expensive chiral resolution techniques when optical purity is desired, adding layers of operational complexity that hinder commercial scale-up of complex nitroimidazoles. The accumulation of by-products in these older methods also complicates downstream purification, often requiring extensive chromatography or multiple recrystallization steps that erode overall process economics.
The Novel Approach
In stark contrast, the methodology outlined in CN101633643A simplifies the synthetic landscape by employing a direct alkylation strategy facilitated by Boron trifluoride diethyl etherate. This novel approach allows for the direct reaction of 2-methyl-5-nitroimidazole with epichlorohydrin in a solvent system such as ethyl acetate or ether, under mild thermal conditions ranging from 0°C to 25°C. The use of this specific Lewis acid catalyst activates the epoxide ring of epichlorohydrin, promoting a highly selective nucleophilic attack by the imidazole nitrogen without the need for protecting groups or excessive heating. As illustrated in the reaction scheme below, this single-step coupling followed by a straightforward workup procedure dramatically enhances process efficiency.
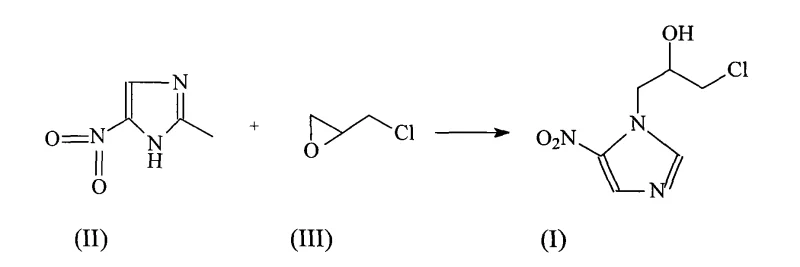
The result is a streamlined workflow that eliminates the need for cumbersome intermediate isolations, directly contributing to reducing lead time for high-purity API intermediates. By achieving yields of 85% to 88% as demonstrated in the patent examples, this route significantly lowers the cost of goods sold (COGS) and improves the sustainability profile of the manufacturing process, making it an attractive option for large-scale industrial adoption.
Mechanistic Insights into BF3-Catalyzed N-Alkylation
The core of this synthetic advancement lies in the precise activation of the electrophile by the Boron trifluoride catalyst. In the absence of a strong Lewis acid, the nucleophilic attack of the imidazole nitrogen on the epichlorohydrin epoxide ring can be sluggish or non-selective, leading to polymerization of the epoxide or N-alkylation at the wrong position. The Boron trifluoride diethyl etherate coordinates with the oxygen atom of the epoxide ring in intermediate (III), increasing the electrophilicity of the adjacent carbon atoms. This coordination lowers the activation energy for the ring-opening step, allowing the nitrogen of the 2-methyl-5-nitroimidazole (II) to attack the less hindered carbon of the epoxide with high regioselectivity. This mechanistic pathway ensures that the formation of the desired 1-(3-chloro-2-hydroxypropyl) side chain occurs predominantly, minimizing the formation of regioisomers or bis-alkylated by-products that are common in uncatalyzed thermal reactions.
Furthermore, the protocol emphasizes strict temperature control, maintaining the reaction mixture between 0°C and 5°C during the addition of reagents. This thermal management is critical for controlling the exothermic nature of the epoxide ring-opening reaction and preventing thermal degradation of the sensitive nitroimidazole moiety. From an impurity control perspective, the subsequent workup procedure involving pH adjustment is equally vital. By initially adjusting the pH to 1 with concentrated hydrochloric acid, the process ensures the protonation of unreacted basic impurities, allowing them to remain in the aqueous phase during separation. The subsequent neutralization of the aqueous layer to pH 7.0-7.5 facilitates the extraction of the neutral Ornidazole product into the organic phase, effectively scrubbing away acidic and basic contaminants. This rigorous control over the reaction environment and workup parameters is what enables the production of high-purity ornidazole suitable for pharmaceutical applications without the need for excessive purification steps.
How to Synthesize Ornidazole Efficiently
The implementation of this synthetic route requires careful attention to reagent addition rates and thermal regulation to maximize the benefits of the catalytic system. The process begins with the dissolution of the imidazole precursor in a suitable solvent like ethyl acetate, followed by the introduction of the catalyst under cooling. The controlled addition of epichlorohydrin is the rate-determining step where heat management is paramount to prevent runaway reactions. Following the reaction period, the quenching and pH manipulation steps are designed to isolate the product efficiently while maintaining its structural integrity. For detailed operational parameters and specific stoichiometric ratios required to replicate the high yields reported in the patent, please refer to the standardized synthesis guide below.
- Dissolve 2-methyl-5-nitroimidazole in ethyl acetate and cool to 0-5°C, then add Boron trifluoride diethyl etherate solution.
- Slowly drip epichlorohydrin into the reaction mixture while maintaining temperature below 5°C, followed by stirring for 5 hours.
- Quench with ice-water, adjust pH to 1 with HCl, separate layers, neutralize aqueous phase to pH 7.0-7.5, extract, and recrystallize from toluene.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this optimized synthetic route offers tangible strategic benefits beyond mere chemical yield. The shift from low-efficiency legacy processes to this high-yield catalytic method fundamentally alters the cost structure of Ornidazole production. By significantly increasing the conversion of raw materials into the final product, manufacturers can drastically reduce the volume of waste solvent and unreacted starting materials that require disposal, leading to substantial cost savings in waste treatment and raw material procurement. This efficiency gain translates directly into a more competitive pricing structure for the final API intermediate, allowing buyers to secure better margins in their own downstream formulations.
- Cost Reduction in Manufacturing: The elimination of complex multi-step sequences and the use of readily available, cost-effective solvents like ethyl acetate contribute to a leaner manufacturing process. Unlike methods requiring expensive chiral catalysts or exotic reagents, this route utilizes commodity chemicals that are easily sourced from the global supply chain, reducing the risk of supply bottlenecks. The high yield inherently lowers the unit cost of production, as fewer batches are needed to meet the same output targets, optimizing facility utilization rates and labor costs associated with batch processing.
- Enhanced Supply Chain Reliability: The robustness of this synthetic method ensures consistent output quality and quantity, which is critical for maintaining uninterrupted supply chains for essential anti-infective medications. The simplicity of the workup procedure, involving standard liquid-liquid extraction and recrystallization, reduces the likelihood of batch failures due to operational errors. This reliability allows supply chain planners to forecast inventory levels with greater confidence, mitigating the risks associated with stockouts or emergency sourcing scenarios that can disrupt pharmaceutical production schedules.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing reaction conditions that are easily transferable from pilot plant to full commercial scale. The use of ethyl acetate, a solvent with a favorable environmental profile compared to chlorinated alternatives often used in older methods, aligns with increasingly stringent global environmental regulations. This compliance reduces the regulatory burden on manufacturing sites and minimizes the potential for environmental fines or shutdowns, ensuring long-term operational continuity for suppliers adopting this green chemistry approach.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Ornidazole synthesis route. These insights are derived directly from the experimental data and beneficial effects described in the patent literature, providing a clear picture of what stakeholders can expect when transitioning to this methodology. Understanding these details is crucial for evaluating the feasibility of integrating this process into existing manufacturing portfolios.
Q: What is the primary advantage of the BF3 catalyzed route for Ornidazole?
A: The primary advantage is a significant increase in yield, reaching over 85% compared to traditional methods which often struggle below 60%, thereby reducing raw material waste and production costs.
Q: Why is temperature control critical in this synthesis?
A: Maintaining the reaction temperature between 0°C and 5°C is essential to control the exothermic nature of the epoxide ring opening and to minimize side reactions, ensuring high purity of the final nitroimidazole derivative.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes common solvents like ethyl acetate and avoids complex multi-step sequences, making it highly scalable and economically viable for commercial manufacturing of API intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ornidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthetic routes in maintaining a competitive edge in the global pharmaceutical market. Our technical team has extensively analyzed the BF3-catalyzed pathway for Ornidazole and possesses the expertise to implement this high-yield process at an industrial scale. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory optimization to full-scale manufacturing is seamless and compliant with international standards. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Ornidazole meets the exacting requirements of modern drug development.
We invite pharmaceutical partners to collaborate with us to leverage this advanced technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to determine how this optimized synthesis can enhance your product portfolio. Let us help you secure a stable, high-quality supply of this essential intermediate while driving down your overall manufacturing costs.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
