Advanced Neutral Alumina Chromatography for High-Purity Ornidazole Manufacturing
Introduction to Novel Ornidazole Purification Technology
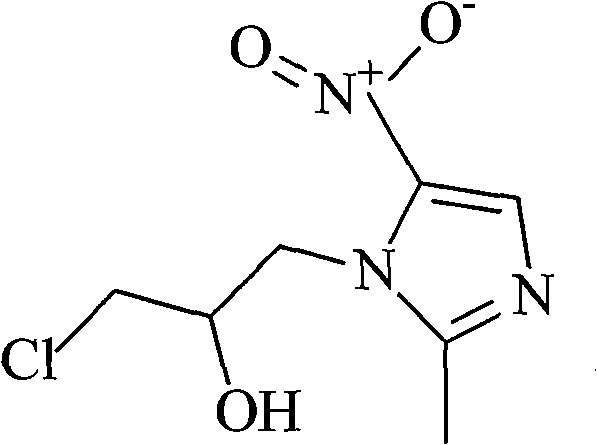
The pharmaceutical industry continuously demands higher purity standards for active pharmaceutical ingredients (APIs) to ensure patient safety and therapeutic efficacy. Patent CN102321029A introduces a groundbreaking purification methodology specifically designed for Ornidazole, a critical nitroimidazole derivative used extensively as an antiprotozoal and antibacterial agent. This technology addresses the longstanding challenges associated with residual impurities and toxic solvent contamination found in conventional manufacturing routes. By implementing a sophisticated three-stage purification protocol involving activated carbon adsorption, specialized neutral alumina chromatography, and controlled crystallization, manufacturers can achieve purity levels exceeding 99.6%. This represents a significant leap forward in quality control, directly impacting the safety profile of the final drug product by minimizing the presence of genotoxic impurities and related substances that often plague older synthesis routes.
For R&D directors and technical decision-makers, the significance of this patent lies in its ability to transform crude Ornidazole, which typically contains complex impurity profiles including unreacted starting materials and degradation products, into a highly refined intermediate suitable for sensitive formulation processes. The method avoids the use of harsh acidic conditions often associated with silica gel chromatography, thereby preserving the structural integrity of the nitroimidazole ring. This technological advancement not only enhances the chemical stability of the compound during storage but also ensures that the final API meets the rigorous specifications required by global regulatory bodies. As we delve deeper into the technical specifics, it becomes evident that this process offers a robust solution for producing high-quality Ornidazole that aligns with modern Good Manufacturing Practice (GMP) standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the purification of Ornidazole has relied heavily on recrystallization techniques using organic solvents such as toluene or ethyl acetate, as disclosed in earlier patents like US3435049A. These traditional methods suffer from inherent inefficiencies, primarily due to the inability of simple recrystallization to effectively separate structurally similar impurities such as 2-methyl-5-nitroimidazole. Furthermore, the reliance on toxic solvents like toluene poses significant environmental and safety hazards, necessitating complex solvent recovery systems and stringent residue testing to ensure patient safety. Alternative approaches involving silica gel chromatography have also been explored, but the acidic nature of silica often leads to product degradation or insufficient resolution of polar impurities, resulting in final purities that frequently fall below the 99% threshold required for high-grade pharmaceutical applications. Additionally, some prior art methods involve multi-step syntheses with low overall yields, sometimes as low as 51%, which drastically increases production costs and generates substantial chemical waste.
The Novel Approach
In stark contrast to these outdated techniques, the novel approach detailed in CN102321029A leverages the unique adsorption properties of neutral alumina to achieve unprecedented separation efficiency. By replacing acidic silica gel with neutral alumina having a specific pore size distribution (approximately 6nm) and pH range (7.0-7.5), the process creates a chemically inert environment that prevents the hydrolysis or decomposition of the sensitive nitroimidazole moiety. This method integrates a preliminary activated carbon treatment step to remove bulk colored impurities and high molecular weight organics, followed by the high-resolution chromatographic separation. The final crystallization from pure water eliminates the need for hazardous organic anti-solvents, significantly simplifying the downstream processing. This holistic strategy not only boosts the isolated yield to over 95% but also ensures that the final product is free from toxic solvent residues, offering a cleaner, greener, and more economically viable pathway for commercial manufacturing.
Mechanistic Insights into Neutral Alumina Chromatographic Separation
The core of this technological breakthrough resides in the mechanistic interaction between the Ornidazole molecule and the neutral alumina stationary phase. Unlike silica gel, which possesses surface silanol groups that can act as Bronsted acids and catalyze unwanted side reactions or cause tailing of basic compounds, neutral alumina offers a balanced surface chemistry. The specific pore structure of the alumina (with apertures around 6nm) allows for optimal diffusion of the Ornidazole molecule while effectively retaining polar impurities and degradation products through Lewis acid-base interactions. The mobile phase, typically a mixture of acetonitrile and water in a 3:7 volume ratio, is carefully tuned to modulate the polarity and elution strength, ensuring that the target compound is eluted sharply while impurities remain retained on the column. This precise control over the chromatographic parameters results in a highly resolved peak profile, allowing for the collection of fractions with exceptional chemical homogeneity.
Furthermore, the purification mechanism extends to the effective removal of genotoxic alerts and related substances that are critical quality attributes for nitroimidazole drugs. The activated carbon pretreatment step acts as a coarse filter, adsorbing large conjugated systems responsible for coloration, which reduces the load on the chromatographic column and extends its operational life. The subsequent water-induced crystallization exploits the differential solubility of Ornidazole versus its impurities in aqueous media. As the concentrated eluate is diluted with pure water, the solubility of Ornidazole decreases rapidly, prompting the formation of high-purity crystals while soluble impurities remain in the mother liquor. This synergistic combination of adsorption, partition chromatography, and solubility-based fractionation creates a multi-barrier defense against contamination, ensuring that the final API possesses the requisite purity for safe clinical use.
How to Synthesize High-Purity Ornidazole Efficiently
Implementing this purification protocol requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and maximum yield. The process begins with the dissolution of crude Ornidazole in a water-miscible organic solvent, preferably ethanol, followed by a controlled heating and stirring phase with activated carbon. After filtration and concentration, the residue is subjected to the critical neutral alumina column chromatography step, where flow rates and mobile phase composition must be meticulously maintained. The final stage involves the careful addition of pure water to induce crystallization, followed by centrifugal washing and drying. For a comprehensive understanding of the operational parameters and equipment specifications required to replicate this high-yield process, please refer to the detailed technical guide below.
- Dissolve crude Ornidazole in a water-miscible organic solvent like ethanol, treat with 0.1-0.5% activated carbon at 40-50°C, and filter to remove colored impurities.
- Perform preparative chromatography using neutral alumina (pH 7.0-7.5, 6nm pore size) as the stationary phase and an acetonitrile-water mixture as the mobile phase.
- Concentrate the eluate, induce crystallization by adding pure water, and perform centrifugal washing followed by drying with anhydrous calcium chloride.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel purification technology offers substantial strategic advantages that extend beyond mere technical specifications. The elimination of toxic solvents like toluene and the reduction of complex multi-step synthetic routes translate directly into simplified regulatory compliance and reduced environmental liability. By utilizing a purification method that relies on widely available and cost-effective stationary phases like neutral alumina, manufacturers can mitigate the risks associated with supply chain disruptions for specialized reagents. The high yield achieved through this process means that less raw material is required to produce the same amount of finished API, effectively lowering the cost of goods sold (COGS) and improving margin potential. Moreover, the robustness of the crystallization step ensures consistent batch-to-batch quality, reducing the incidence of out-of-specification results that can lead to costly production delays.
- Cost Reduction in Manufacturing: The transition to this purification method eliminates the need for expensive and hazardous solvent systems, thereby reducing waste disposal costs and solvent procurement expenses. The high recovery rate of the product during the chromatography and crystallization steps minimizes material loss, ensuring that the maximum amount of valuable API is harvested from each batch. By avoiding the use of transition metal catalysts or complex chiral resolving agents often required in alternative synthetic routes, the process further streamlines the bill of materials. This cumulative effect results in a significantly leaner manufacturing operation that is better positioned to compete on price in the global generic pharmaceutical market.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as ethanol, acetonitrile, and neutral alumina ensures that the supply chain is resilient against shortages of exotic reagents. The simplicity of the unit operations involved allows for easy scaling from pilot plant to full commercial production without the need for specialized equipment modifications. This scalability guarantees a steady and continuous supply of high-purity Ornidazole, enabling pharmaceutical companies to meet market demand without interruption. Furthermore, the reduced complexity of the process shortens the overall production cycle time, allowing for faster turnaround on orders and improved responsiveness to fluctuating market needs.
- Scalability and Environmental Compliance: The process is inherently designed with green chemistry principles in mind, utilizing water as the primary anti-solvent for crystallization and minimizing the generation of hazardous waste streams. This alignment with environmental regulations facilitates smoother permitting processes and reduces the carbon footprint of the manufacturing facility. The ability to scale the chromatographic separation using standard preparative columns ensures that production capacity can be expanded seamlessly as demand grows. Consequently, this technology supports sustainable growth strategies while maintaining strict adherence to international environmental and safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced Ornidazole purification technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent literature, providing a clear understanding of the method's capabilities and limitations. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this process into their existing manufacturing workflows.
Q: Why is neutral alumina preferred over silica gel for Ornidazole purification?
A: Neutral alumina (pH 7.0-7.5) provides superior separation efficiency for Ornidazole compared to acidic silica gel, effectively removing related substances like 2-methyl-5-nitroimidazole without degrading the product, resulting in purity levels exceeding 99.6%.
Q: How does this purification method impact the safety profile of the final API?
A: By eliminating toxic organic solvents like toluene used in traditional recrystallization and reducing residual impurities through multi-stage purification, this method significantly lowers the potential for toxic side effects and improves the overall quality of the pharmaceutical preparation.
Q: Is this purification process scalable for industrial production?
A: Yes, the process utilizes standard unit operations such as filtration, column chromatography, and crystallization which are easily adaptable from laboratory to commercial scale, ensuring consistent supply continuity and high yield suitable for mass production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ornidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of purity and consistency in the production of anti-infective agents like Ornidazole. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated neutral alumina chromatography method described in CN102321029A can be seamlessly transferred to industrial scale. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art HPLC systems to verify that every batch meets the >99.6% purity benchmark. Our commitment to quality assurance ensures that the Ornidazole we supply is free from harmful impurities and ready for immediate formulation into final dosage forms.
We invite pharmaceutical partners to collaborate with us to leverage this advanced purification technology for their supply chains. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this high-yield process. We encourage you to reach out for specific COA data and route feasibility assessments tailored to your project requirements. Let us help you optimize your API sourcing strategy with a reliable, high-quality, and cost-effective Ornidazole supply solution.
