Revolutionizing Tetrazoleacetic Acid Production: A Safe, Scalable Hydrazine Hydrate Route for Global Pharma Supply Chains
Revolutionizing Tetrazoleacetic Acid Production: A Safe, Scalable Hydrazine Hydrate Route for Global Pharma Supply Chains
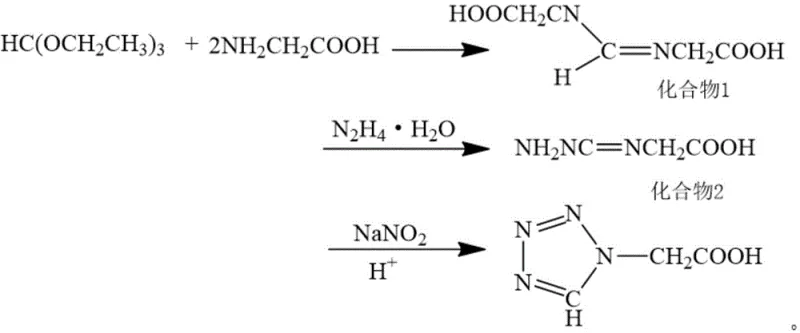
The global demand for high-purity heterocyclic intermediates continues to surge, driven by the expanding antibiotic and agrochemical sectors. A pivotal breakthrough in this domain is documented in patent CN110305070B, which discloses a novel method for synthesizing tetrazoleacetic acid using a hydrazine hydrate approach. This technology represents a paradigm shift away from the hazardous traditional azido ester methods, offering a robust solution for manufacturers seeking to enhance safety and efficiency. By utilizing readily available raw materials such as triethyl orthoformate, glycine, and hydrazine hydrate, this process addresses critical bottlenecks in the supply chain of essential pharmaceutical building blocks. For R&D directors and procurement specialists, understanding this synthetic pathway is crucial for securing a reliable pharmaceutical intermediates supplier capable of delivering consistent quality without the volatility associated with explosive precursors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of tetrazoleacetic acid has been plagued by significant safety and environmental challenges inherent to the azido ester method. Traditional routes typically rely on sodium azide as a key raw material, a substance notorious for its high toxicity and explosive potential, which necessitates extreme caution and specialized infrastructure. Furthermore, conventional processes often suffer from low yields, prolonged production cycles, and harsh operating conditions that increase energy consumption and operational risk. The generation of substantial waste liquid and the difficulty in obtaining stable precursors like ethyl isocyanoacetate further exacerbate the economic and ecological burden. These factors have led to frequent production stoppages among domestic manufacturers due to stringent safety and environmental regulations, creating a tight supply market heavily reliant on imports and restricting the growth of downstream antibiotic industries.
The Novel Approach
In stark contrast, the hydrazine hydrate method outlined in the patent offers a streamlined and inherently safer alternative that bypasses the use of sodium azide entirely. This innovative route leverages the condensation of glycine and triethyl orthoformate followed by cyclization with hydrazine hydrate and sodium nitrite under acidic conditions. The process is characterized by its simplicity, strong operability, and excellent repeatability, making it highly suitable for large-scale industrial application. By shifting the stoichiometry to use excess glycine rather than excess triethyl orthoformate, the method effectively lowers raw material costs while maintaining high reaction efficiency. Moreover, the absence of wastewater and waste gas emissions aligns perfectly with modern green chemistry standards, ensuring that production facilities can operate continuously without the threat of regulatory shutdowns.
Mechanistic Insights into Hydrazine-Mediated Tetrazole Cyclization
The core of this synthetic strategy lies in a multi-step cascade reaction that efficiently constructs the tetrazole ring without the need for hazardous azide ions. Initially, glycine reacts with triethyl orthoformate in a solvent such as glacial acetic acid to form an imine intermediate, often existing as a transition state that does not require isolation. This step is critical for setting up the carbon-nitrogen framework necessary for subsequent cyclization. Following this, the introduction of hydrazine hydrate facilitates a nucleophilic attack, leading to the formation of a hydrazono-acetic acid derivative. The final and most crucial stage involves the diazotization and cyclization induced by sodium nitrite in the presence of concentrated sulfuric acid. This acidic environment promotes the intramolecular closure of the ring, transforming the linear precursor into the stable tetrazole structure with high fidelity.
Controlling impurities in this mechanism is achieved through precise regulation of reaction temperatures and molar ratios, as detailed in the patent examples. For instance, maintaining the initial condensation temperature between 25°C and 45°C prevents the hydrolysis loss of triethyl orthoformate, while the subsequent heating to 55-75°C ensures complete conversion of the hydrazine adduct. The careful addition of 98% concentrated sulfuric acid acts not only as a catalyst but also as a dehydrating agent that drives the equilibrium towards the final tetrazole product. This meticulous control over reaction parameters minimizes side reactions, resulting in a crude product that already possesses exceptional purity levels, thereby reducing the burden on downstream purification processes and ensuring the final API intermediate meets stringent quality specifications.
How to Synthesize Tetrazoleacetic Acid Efficiently
Implementing this synthesis route requires strict adherence to the optimized conditions regarding temperature, timing, and reagent ratios to maximize yield and safety. The process begins with the preparation of the reaction mixture in a standard vessel, followed by sequential additions of reagents under controlled thermal conditions. Operators must monitor the exothermic nature of the acid addition carefully to prevent runaway reactions, although the overall process is designed to be thermally manageable compared to azide-based routes. The detailed standardized synthesis steps, including specific gram-scale quantities and precise timing for each stage, are provided in the technical guide below to ensure reproducibility across different manufacturing scales.
- Condense triethyl orthoformate with glycine in glacial acetic acid at 25-45°C for 1-3 hours to form the initial imine intermediate.
- Add 85% hydrazine hydrate to the reaction system and heat to 55-75°C for 3-5 hours to facilitate the formation of the hydrazono intermediate.
- Introduce sodium nitrite and catalytic concentrated sulfuric acid at 60°C to induce cyclization, followed by filtration and drying to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this hydrazine hydrate method translates into tangible strategic advantages that go beyond mere chemical curiosity. The elimination of sodium azide removes a major logistical and safety hurdle, simplifying the storage and handling requirements for raw materials and significantly lowering insurance and compliance costs associated with hazardous substances. Additionally, the ability to sell co-produced sodium sulfate as a valuable byproduct creates a new revenue stream that offsets production expenses, contributing to a more favorable cost structure. This economic efficiency, combined with the robustness of the process, ensures a stable supply of high-purity intermediates, mitigating the risks of market shortages that have historically plagued the antibiotic sector.
- Cost Reduction in Manufacturing: The shift to using excess glycine, a commodity chemical, instead of excess triethyl orthoformate optimizes the raw material bill of materials. Furthermore, the process generates no waste water or waste gas, which drastically reduces the operational expenditure related to environmental treatment and waste disposal. The direct saleability of the sodium sulfate byproduct further enhances the economic viability, allowing for substantial cost savings in the overall manufacturing budget without compromising on product quality or yield.
- Enhanced Supply Chain Reliability: By removing the dependency on unstable or difficult-to-source precursors like ethyl isocyanoacetate and hazardous sodium azide, the supply chain becomes significantly more resilient. The raw materials used in this method, such as glycine and hydrazine hydrate, are widely available in the global chemical market, ensuring consistent availability even during periods of market volatility. This reliability is critical for maintaining continuous production schedules for downstream antibiotic manufacturers who depend on a steady flow of tetrazoleacetic acid.
- Scalability and Environmental Compliance: The simple synthetic route and mild reaction conditions make this process highly scalable from pilot plant to commercial tonnage production. The absence of toxic emissions and wastewater aligns with increasingly strict global environmental regulations, future-proofing the manufacturing facility against potential regulatory crackdowns. This environmental compliance not only safeguards the company's license to operate but also enhances its reputation as a sustainable partner in the global pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of tetrazoleacetic acid via this novel method. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing a clear picture of the technology's capabilities. Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their own synthesis pipelines or for procurement officers assessing supplier qualifications.
Q: Why is the hydrazine hydrate method safer than the traditional azido ester method?
A: The traditional method relies on sodium azide, which is highly toxic and explosive, posing severe safety risks. The new hydrazine hydrate route eliminates sodium azide entirely, significantly improving the safety coefficient of the production process.
Q: What is the expected purity of tetrazoleacetic acid produced via this method?
A: According to patent data, this method consistently yields tetrazoleacetic acid with a content of over 99%, specifically demonstrated at 99.6% via HPLC analysis in multiple examples.
Q: How does this process address environmental concerns in chemical manufacturing?
A: The process generates no waste water or waste gas during the reaction. Furthermore, the co-produced sodium sulfate can be directly sold as a byproduct, aligning with green chemical production requirements and reducing waste disposal costs.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrazoleacetic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications. Our technical team has extensively analyzed the hydrazine hydrate synthesis route and possesses the expertise to scale this diverse pathway from 100 kgs to 100 MT/annual commercial production with precision. We are committed to delivering products that meet stringent purity specifications, supported by our rigorous QC labs that ensure every batch complies with international standards. Our capability to manage complex heterocyclic syntheses allows us to offer a stable and secure supply of tetrazoleacetic acid, empowering our partners to focus on innovation without supply chain interruptions.
We invite global pharmaceutical and agrochemical companies to collaborate with us to leverage this advanced manufacturing technology. By contacting our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your project moves forward with the most efficient and safe chemical foundation available in the market.
