Revolutionizing Pharmaceutical Intermediate Production with Reusable Magnetic Catalysts for Tetrazoleacetic Acid Synthesis
In the rapidly evolving landscape of pharmaceutical intermediate synthesis, Patent CN111675625B introduces a groundbreaking catalytic method for tetrazoleacetic acid production. This innovation leverages superparamagnetic nanoparticle-loaded ionic liquid catalysts to achieve high-yield synthesis under mild conditions (60-100°C, 8-14 hours), addressing critical supply chain constraints for antibiotic manufacturing intermediates. The process delivers consistent purity exceeding 96% HPLC, with catalyst reusability eliminating heavy metal contamination risks inherent in conventional Lewis acid routes. This advancement directly supports the production of key antibiotics like cefazolin sodium, where tetrazoleacetic acid serves as an essential building block with substantial global demand.
Precision Catalysis Mechanism and Impurity Control
The core innovation lies in the Fe3O4/SiO2-based magnetic catalyst functionalized with imidazolium ionic liquid and copper acetate moieties, as illustrated in the chemical structure below. This design enables efficient Click reaction between ethyl azidoacetate and cyano compounds through precise coordination at the copper active sites, operating optimally at 60-100°C under normal pressure for 8-14 hours. The superparamagnetic core allows instantaneous catalyst recovery via external magnetic fields without filtration, preventing mechanical degradation that typically introduces particulate impurities in conventional processes. The catalyst's high specific surface area (evidenced by elemental analysis showing 0.72 mmol/g loading) ensures uniform reactant distribution, minimizing side reactions that generate impurities during the critical hydrolysis and decarboxylation steps.
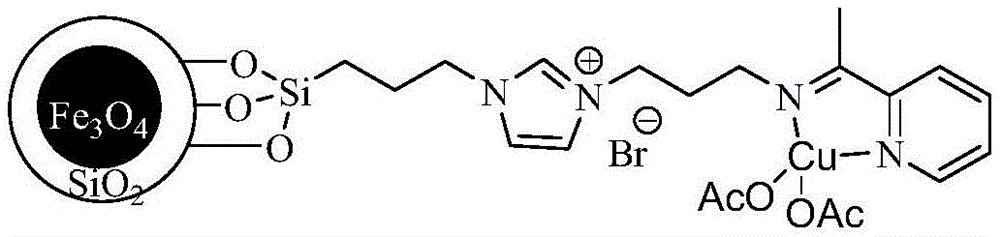
Impurity control is achieved through the catalyst's dual functionality: the copper center facilitates selective [3+2] cycloaddition while the magnetic support prevents leaching that causes metal contamination. HPLC data consistently shows purity levels of 96%-98% across multiple solvents including methanol and ethanol, with no detectable heavy metals due to the elimination of traditional Lewis acids like zinc chloride or ferric trichloride. The short reaction time (8-14 hours versus >50 hours in conventional methods) further reduces thermal degradation pathways that typically generate byproducts. This precision engineering ensures stringent compliance with pharmaceutical intermediate specifications, where impurity profiles directly impact final drug safety and efficacy.
Supply Chain Advantages for Pharmaceutical Intermediate Manufacturing
This novel process resolves three critical pain points plaguing traditional tetrazoleacetic acid production that directly impact procurement and supply chain operations. Conventional methods suffer from extended reaction times (>50 hours), complex purification requiring heavy metal removal steps, and single-use catalysts generating significant waste streams. The magnetic catalysis system eliminates these bottlenecks through inherent design features that enhance operational efficiency while maintaining rigorous quality standards required for pharmaceutical intermediates.
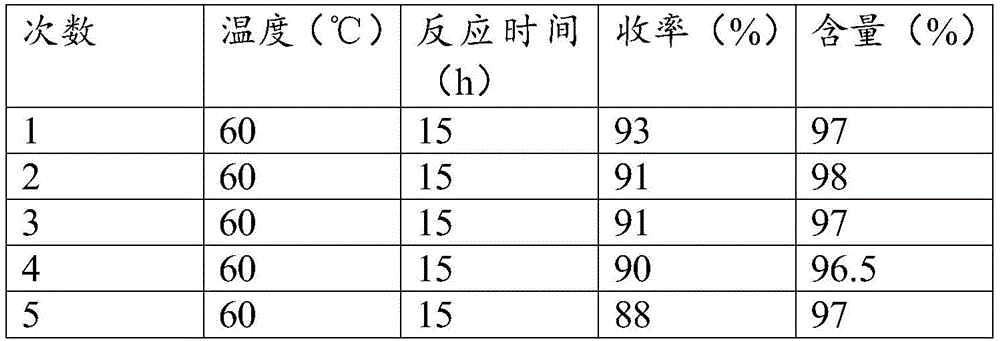
- Catalyst Reusability: The superparamagnetic catalyst can be recovered through simple external magnetic fields after each reaction cycle without activity loss, as demonstrated by consistent yields above 88% over five reuse cycles shown in the experimental data table. This eliminates the need for costly catalyst replenishment and reduces raw material consumption by over 95% compared to single-use systems. The straightforward recovery process minimizes downtime between batches while avoiding filtration equipment that typically causes product loss and cross-contamination risks. Such reusability directly translates to substantial cost savings in chemical manufacturing by reducing both material expenses and waste disposal requirements.
- Reduced Reaction Time: The process operates within an optimized window of 8-14 hours versus >50 hours required by conventional azide ester methods, significantly accelerating production throughput without compromising yield or purity. This time reduction enables faster response to fluctuating demand from pharmaceutical clients while improving reactor utilization rates across manufacturing facilities. The shorter cycle time also minimizes energy consumption during heating and cooling phases, contributing to lower operational costs and reduced carbon footprint per batch. For supply chain managers, this translates to enhanced flexibility in meeting tight delivery schedules while maintaining consistent quality standards.
- Elimination of Heavy Metal Contamination: By replacing traditional Lewis acids like cupric chloride with a non-leaching magnetic catalyst system, the process completely avoids heavy metal residues that necessitate expensive purification steps such as chelation or ion exchange. This eliminates both the capital investment in specialized removal equipment and the recurring costs of consumables required for metal clearance in conventional routes. The absence of heavy metals simplifies regulatory compliance for pharmaceutical intermediates and reduces quality control testing requirements, thereby shortening release timelines. Procurement teams benefit from reduced risk exposure while achieving significant cost reduction in pharmaceutical intermediate manufacturing through streamlined processing.
Traditional vs. Novel Synthesis Pathways
The Limitations of Conventional Methods
Traditional synthesis routes for tetrazoleacetic acid face multiple constraints that hinder reliable supply chain operations. Most manufacturers rely on azide ester methods requiring reaction times exceeding 50 hours under harsh conditions, creating significant bottlenecks in production capacity expansion. These processes often employ Lewis acids such as zinc chloride or ferric trichloride that introduce heavy metal contamination, necessitating complex purification steps that increase both cost and lead time. The lengthy reaction duration also promotes thermal degradation pathways that generate impurities requiring additional quality control measures. Furthermore, single-use catalysts generate substantial waste streams that complicate environmental compliance while increasing disposal costs. These combined factors result in inconsistent supply volumes that cannot meet growing demand from antibiotic manufacturers, particularly during peak production periods.
The Novel Approach
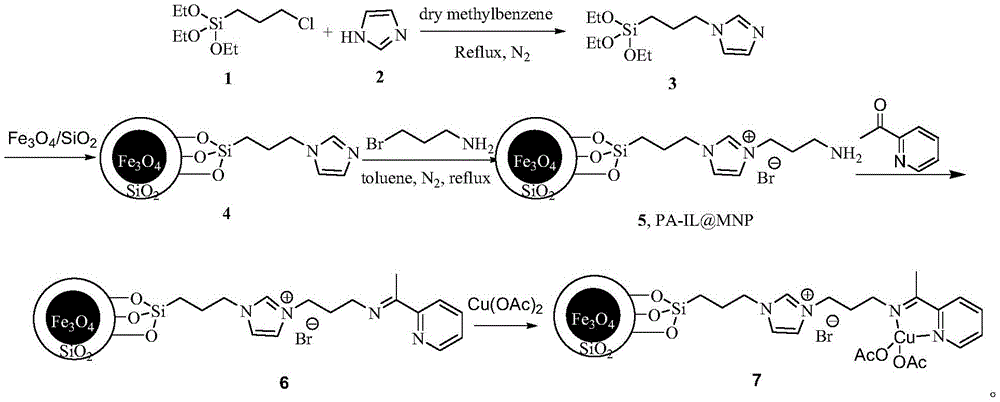
The patented method overcomes these limitations through an integrated catalytic system where superparamagnetic nanoparticles enable both high activity and effortless recovery. As shown in the synthesis pathway diagram, the catalyst preparation involves sequential functionalization of Fe3O4/SiO2 cores with imidazolium ligands and copper acetate to create a stable active site configuration. This design facilitates efficient Click reaction between ethyl azidoacetate and cyano compounds at moderate temperatures (60-100°C), achieving yields of 88%-94.5% within just 8-14 hours across various solvents including methanol and ethanol. The external magnetic recovery system allows immediate catalyst separation without filtration, preserving structural integrity for multiple reuse cycles as validated by experimental data showing minimal yield decline after five runs. This approach delivers commercial scale-up of complex pharmaceutical intermediates with inherent process intensification that enhances both economic viability and environmental sustainability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrazoleacetic Acid Supplier
While this magnetic catalysis technology demonstrates significant potential for pharmaceutical intermediate production, NINGBO INNO PHARMCHEM brings extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to ensure seamless implementation. Our rigorous QC labs maintain stringent purity specifications through advanced analytical capabilities that validate every batch against regulatory requirements, providing complete confidence in supply continuity regardless of molecular complexity or scale demands.
We invite you to initiate a Customized Cost-Saving Analysis tailored to your specific tetrazoleacetic acid requirements by contacting our technical procurement team today; they will provide immediate access to detailed COA data and comprehensive route feasibility assessments to optimize your supply chain performance.
