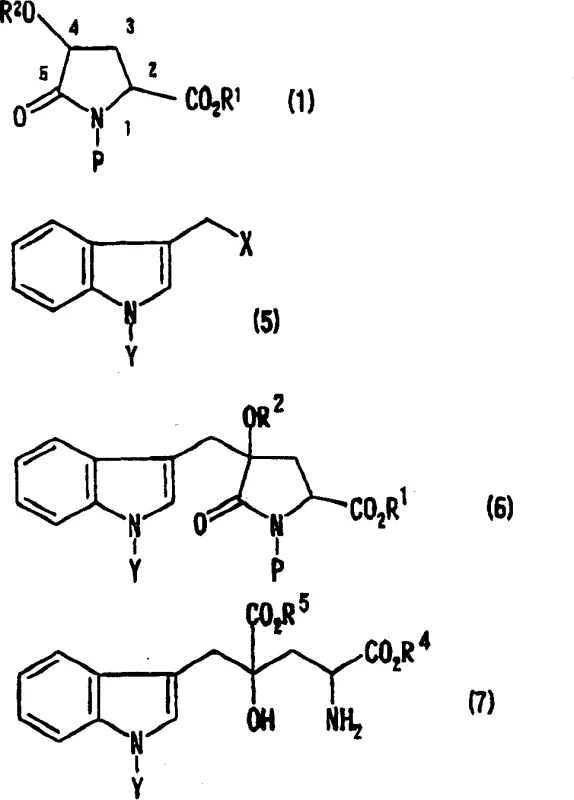
Advanced Stereoselective Alkylation for Industrial Monatin and Glutamic Acid Derivative Production
Advanced Stereoselective Alkylation for Industrial Monatin and Glutamic Acid Derivative Production
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to produce high-value amino acid derivatives, particularly those with potent biological activities such as sweetening properties. Patent CN100408552C introduces a groundbreaking methodology for the production of glutamic acid derivatives, specifically focusing on the synthesis of Monatin and its precursors. This technology addresses long-standing challenges in stereocontrol and process safety by utilizing a novel alkylation strategy on 4-protected hydroxypyroglutamic acid derivatives. Unlike traditional routes that involve cumbersome redox manipulations of the carboxyl group, this invention enables direct, regioselective, and stereoselective alkylation at the 4-position of the pyrrolidone ring. For R&D directors and procurement specialists, this represents a significant opportunity to optimize the supply chain for high-purity sweetener intermediates and pharmaceutical building blocks. The ability to access specific stereoisomers like (2S,4S)-Monatin with high optical purity directly impacts the efficacy and regulatory approval potential of the final product.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of Monatin and related glutamic acid derivatives was plagued by inefficient multi-step sequences that posed significant safety and environmental hazards. Conventional methods typically required the reduction of the carboxyl group of a pyroglutamic acid derivative to a hydroxymethyl group to facilitate alkylation, followed by a subsequent oxidation step to restore the carboxyl functionality. This redox cycle not only added unnecessary synthetic steps, thereby increasing cost and reducing overall yield, but also relied heavily on toxic oxidizing agents such as chromium trioxide (CrO3). The use of hexavalent chromium creates severe waste disposal issues and occupational health risks, making such processes increasingly untenable under modern environmental regulations. Furthermore, the presence of sensitive functional groups like the indole ring in Monatin precursors makes them susceptible to unwanted side reactions during harsh oxidation conditions, leading to complex impurity profiles that are difficult to purge.
The Novel Approach
The methodology disclosed in Patent CN100408552C circumvents these drawbacks by enabling the direct alkylation of 4-protected hydroxypyroglutamic acid derivatives without the need to modify the carboxyl group. By employing specific protecting groups on the nitrogen and the 4-hydroxyl moiety, the inventors achieved remarkable regioselectivity, ensuring that the alkylating agent attacks exclusively at the 4-position rather than the 2-position. This streamlined approach eliminates the reduction-oxidation cycle entirely, drastically shortening the synthetic route. As illustrated in the reaction schemes below, the process converts a protected pyroglutamic acid intermediate directly into the substituted derivative, which can then be hydrolyzed to the final glutamic acid structure. This reduction in step count translates directly to lower manufacturing costs and higher throughput, making it an ideal candidate for a reliable monatin supplier looking to scale production efficiently.
Mechanistic Insights into Stereoselective Alkylation
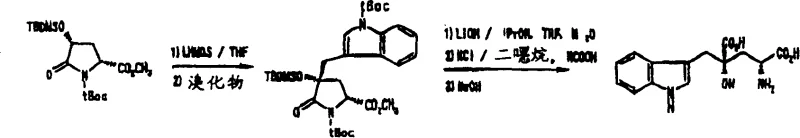
The core scientific breakthrough of this patent lies in the precise control of stereochemistry during the alkylation step. The reaction mechanism relies on the steric hindrance provided by the substituents on the pyrrolidone ring. When a strong base, such as lithium hexamethyldisilazide (LHMDS), deprotonates the 4-position of the 4-protected hydroxypyroglutamic acid derivative, it generates an enolate intermediate. The spatial arrangement of the existing substituents, particularly the bulky protecting groups like tert-butyldimethylsilyl (TBDMS) and tert-butoxycarbonyl (tBoc), directs the incoming electrophile to attack from the less hindered face. This results in a trans-relationship between the newly introduced alkyl group at the 4-position and the ester group at the 2-position. Consequently, starting from a chirally pure 4-hydroxyproline precursor allows for the predictable synthesis of either the (2S,4S) or (2R,4R) diastereomer. This level of stereocontrol is critical for pharmaceutical applications where biological activity is often confined to a single enantiomer.
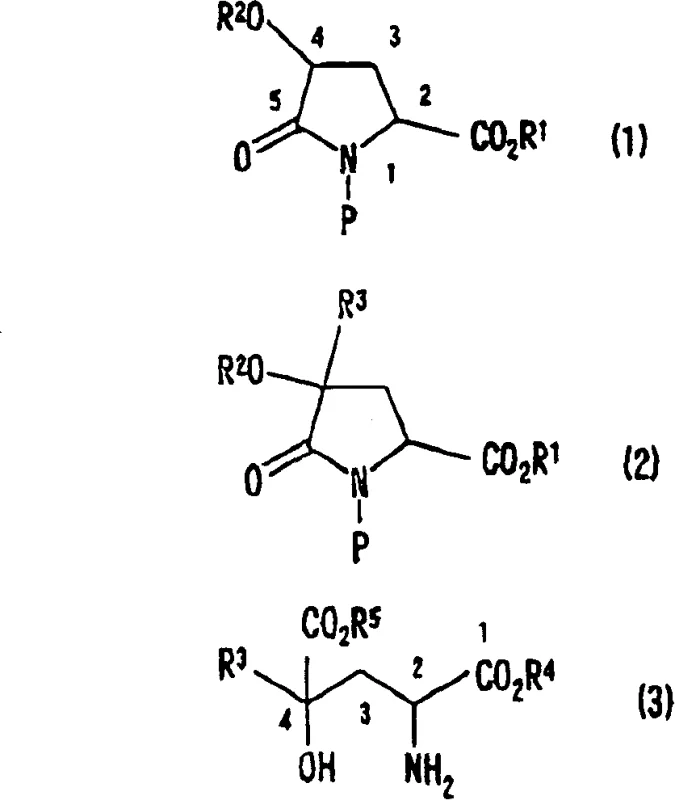
Furthermore, the stability of the protecting groups plays a pivotal role in maintaining the integrity of the molecule throughout the synthesis. The use of silyl ethers for hydroxyl protection and carbamates for nitrogen protection ensures that the reactive sites remain masked until the final deprotection stage. This orthogonal protection strategy prevents self-condensation or polymerization side reactions that could otherwise degrade the yield. The subsequent hydrolysis and deprotection steps are designed to be mild yet effective, utilizing aqueous bases or acids to cleave the lactam ring and remove the protecting groups simultaneously. This careful balancing of reactivity ensures that the sensitive indole moiety, if present, remains intact, preserving the structural features necessary for the compound's sweetening potency. For quality control teams, this robust mechanism means a cleaner reaction profile and simplified purification protocols.
How to Synthesize Monatin Derivatives Efficiently
Implementing this synthesis route requires careful attention to reaction conditions, particularly temperature and solvent choice, to maximize stereoselectivity. The process begins with the preparation of the key starting material, the 4-protected hydroxypyroglutamic acid derivative, which can be readily obtained from commercially available 4-hydroxyproline. The alkylation is typically conducted in anhydrous tetrahydrofuran (THF) at low temperatures, often around -78°C, to control the exotherm and prevent racemization. Following the addition of the alkylating agent, such as N-protected-3-bromomethylindole, the reaction is allowed to warm to room temperature to ensure completion. The detailed standardized synthesis steps, including specific molar ratios and workup procedures, are outlined in the guide below for technical reference.
- Preparation of 4-protected hydroxypyroglutamic acid derivative from 4-hydroxyproline via esterification and oxidation.
- Regio- and stereoselective alkylation at the 4-position using strong bases like LHMDS and N-protected halomethylindole.
- Hydrolysis of the lactam ring and deprotection of silyl/carbamate groups to yield the final glutamic acid derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented technology offers substantial strategic benefits beyond mere technical elegance. The primary advantage is the significant reduction in manufacturing complexity, which directly correlates to cost efficiency. By removing the need for hazardous chromium-based oxidants and the associated waste treatment infrastructure, facilities can operate with lower overhead and reduced environmental liability. This aligns perfectly with global trends towards greener chemistry and sustainable manufacturing practices. Additionally, the shortened synthetic route reduces the consumption of raw materials and solvents per kilogram of final product, leading to tangible savings in variable costs. These efficiencies make the production of high-purity glutamic acid derivatives more economically viable, allowing suppliers to offer competitive pricing without compromising on quality standards.
- Cost Reduction in Manufacturing: The elimination of the carboxyl reduction-oxidation cycle removes two entire synthetic steps, each requiring reagents, solvents, and purification resources. This streamlining drastically lowers the cost of goods sold (COGS). Furthermore, avoiding toxic heavy metals reduces the expense associated with specialized waste disposal and regulatory compliance monitoring. The use of common, scalable reagents like LHMDS and standard organic solvents ensures that the process remains cost-effective even at large volumes, providing a clear economic advantage over legacy methods.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials like 4-hydroxyproline and stable protecting group reagents minimizes the risk of supply disruptions. Unlike processes dependent on exotic catalysts or unstable intermediates, this method utilizes robust chemistry that is less sensitive to minor fluctuations in raw material quality. This stability ensures consistent batch-to-batch reproducibility, which is crucial for maintaining long-term contracts with pharmaceutical and food industry clients. A reliable monatin supplier can thus guarantee continuous delivery schedules, mitigating the risk of production stoppages.
- Scalability and Environmental Compliance: The reaction conditions described are highly amenable to scale-up, having been demonstrated effectively in multi-gram to kilogram ranges within the patent examples. The absence of highly exothermic or dangerous oxidation steps enhances operational safety, a key factor for scaling chemical processes. Moreover, the reduced generation of hazardous waste simplifies environmental permitting and ongoing compliance audits. This facilitates faster technology transfer from pilot plants to commercial-scale reactors, accelerating time-to-market for new products derived from this platform.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. Understanding these details helps stakeholders evaluate the feasibility of integrating this process into their existing manufacturing portfolios. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: How does this new alkylation method improve upon conventional Monatin synthesis?
A: Conventional methods often require reducing the carboxyl group to a hydroxymethyl group, performing alkylation, and then re-oxidizing it back to a carboxyl group using toxic chromium reagents. This new method allows for direct alkylation at the 4-position of the pyroglutamic acid derivative without altering the carboxyl group, significantly simplifying the workflow and eliminating hazardous oxidation steps.
Q: What is the stereochemical outcome of the alkylation reaction?
A: The alkylation proceeds with high stereoselectivity. When starting with a specific enantiomer of the 4-protected hydroxypyroglutamic acid, the alkyl group adds to the 4-position from the face opposite to the 2-position substituent. This ensures the formation of the desired (2S,4S) or (2R,4R) relative configuration essential for biological activity.
Q: Is this process suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes standard organic solvents like THF and common reagents such as lithium hexamethyldisilazide (LHMDS). By avoiding complex redox cycles and toxic heavy metal oxidants, the process is safer, more environmentally compliant, and easier to scale up for industrial production compared to prior art methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Monatin Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced synthetic methodologies like those described in Patent CN100408552C for the production of high-value amino acid derivatives. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are successfully translated into robust industrial operations. Our commitment to quality is underscored by our stringent purity specifications and rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify the stereochemical integrity and chemical purity of every batch. We understand that for applications in the food and pharmaceutical sectors, consistency and safety are paramount, and our infrastructure is designed to meet these exacting demands.
We invite you to collaborate with us to leverage this cutting-edge alkylation technology for your specific project needs. Whether you require custom synthesis of Monatin derivatives or optimization of existing glutamic acid production lines, our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with us, you gain access to a supply chain that is not only cost-effective and reliable but also aligned with the highest standards of chemical innovation and regulatory compliance.
