Advanced Stereoselective Alkylation for High-Purity Monatin Derivatives and Glutamic Acid Intermediates
Introduction to Novel Glutamic Acid Derivative Synthesis
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to produce high-value amino acid derivatives, particularly those with significant biological activity such as Monatin. Patent CN1756734A introduces a groundbreaking production method for glutamic acid derivatives represented by Monatin, addressing critical limitations in existing synthetic routes. This technology focuses on the utilization of 4-protected hydroxypyroglutamic acid derivatives as key intermediates, enabling a highly efficient and industrially viable manufacturing process. By leveraging a novel alkylation strategy, this method bypasses the cumbersome redox cycles typically required in traditional synthesis, offering a streamlined approach to achieving high optical purity. For R&D directors and procurement specialists, understanding this technological shift is vital for optimizing supply chains and reducing the environmental footprint of complex amino acid production.
Monatin, a naturally occurring amino acid derivative isolated from the root bark of Schlerochiton ilicifolius, is renowned for its intense sweetness, reported to be 800 to 1400 times that of sucrose. Despite its potential as a superior sweetener and pharmaceutical intermediate, industrial-scale synthesis has been hindered by complex multi-step procedures involving hazardous reagents. The disclosed invention provides a robust solution by establishing a direct route to 4-protected hydroxy-4-substituted pyroglutamic acid derivatives. These intermediates can be easily converted into the final glutamic acid products through simple hydrolysis and deprotection steps. This advancement represents a significant leap forward in the commercial scale-up of complex pharmaceutical intermediates, ensuring consistent quality and reduced operational risks for manufacturers.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those reported by D.J. Oliveira et al., rely on a convoluted sequence that begins with the reduction of the carboxyl group of a pyroglutamic acid derivative to a hydroxymethyl group. This intermediate must then be protected before alkylation can occur at the 4-position. Following alkylation, the protecting group is removed, and the hydroxymethyl group must be oxidized back to a carboxyl group to restore the glutamic acid skeleton. This redox cycle is not only inefficient but also introduces significant safety and environmental hazards. Specifically, the oxidation step often employs chromium trioxide (CrO3), a toxic and carcinogenic reagent that poses severe handling risks and creates difficult waste disposal challenges. Furthermore, the presence of sensitive functional groups, such as the indole ring in Monatin precursors, makes them susceptible to unwanted oxidation side reactions, leading to lower yields and complex impurity profiles that are costly to remove.
The Novel Approach
In stark contrast, the method described in patent CN1756734A eliminates the need for carboxyl reduction and subsequent re-oxidation entirely. The core innovation lies in the direct regio- and stereoselective alkylation of a 4-protected hydroxypyroglutamic acid derivative at the 4-position. This approach maintains the carboxyl functionality throughout the synthesis, drastically shortening the reaction sequence. By avoiding the use of toxic chromium oxidants, the process aligns better with modern green chemistry principles and regulatory standards for pharmaceutical manufacturing. The ability to introduce diverse alkyl groups, including N-protected-3-indolylmethyl groups, directly onto the lactam ring simplifies the synthesis of various Monatin stereoisomers. This streamlined workflow not only enhances overall yield but also significantly reduces the cost reduction in pharmaceutical intermediate manufacturing by minimizing raw material consumption and waste treatment expenses.
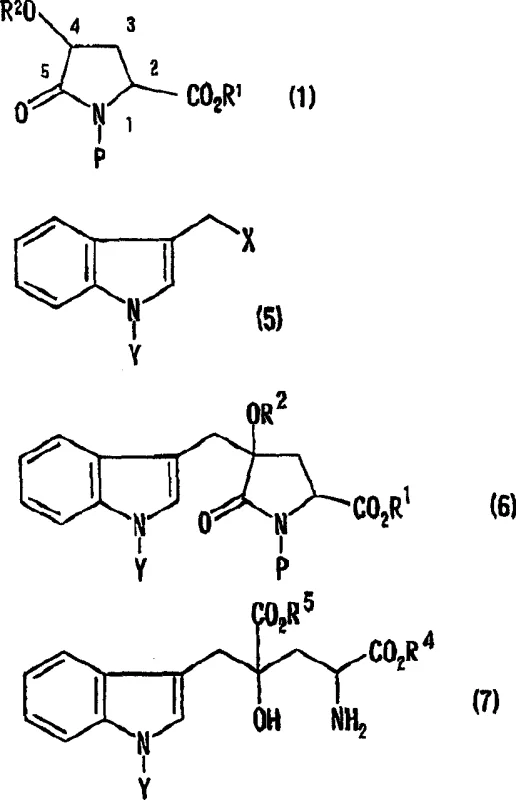
Mechanistic Insights into Stereoselective Alkylation
The success of this synthesis hinges on the precise control of regioselectivity and stereoselectivity during the alkylation of the 4-protected hydroxypyroglutamic acid derivative. Although the molecule possesses potential reaction sites at both the 2-position and the 4-position, the inventors discovered that under specific basic conditions, the 4-position is selectively activated. The reaction mechanism involves the formation of an enolate or similar nucleophilic species at the 4-position, facilitated by strong non-nucleophilic bases such as lithium hexamethyldisilazide (LiHMDS). Crucially, the alkylation proceeds stereoselectively from the face of the pyrrolidone ring opposite to the substituent at the 2-position. This steric directing effect ensures that the newly formed bond at the 4-position adopts a specific spatial orientation relative to the existing chiral center at the 2-position.
This stereochemical outcome is pivotal for producing biologically active isomers like (2S,4S)-Monatin or its enantiomer (2R,4R)-Monatin. The relative configuration established during the alkylation step is preserved throughout the subsequent hydrolysis and deprotection stages. By selecting the appropriate enantiomer of the starting 4-hydroxyproline (either cis-4-hydroxy-L-proline or cis-4-hydroxy-D-proline), manufacturers can dictate the absolute stereochemistry of the final product. This level of control is essential for meeting the stringent purity specifications required for food additives and active pharmaceutical ingredients. The robustness of this mechanistic pathway allows for the reliable production of high-purity optical isomers without the need for difficult chiral separations later in the process, thereby enhancing the overall economic viability of the synthesis.
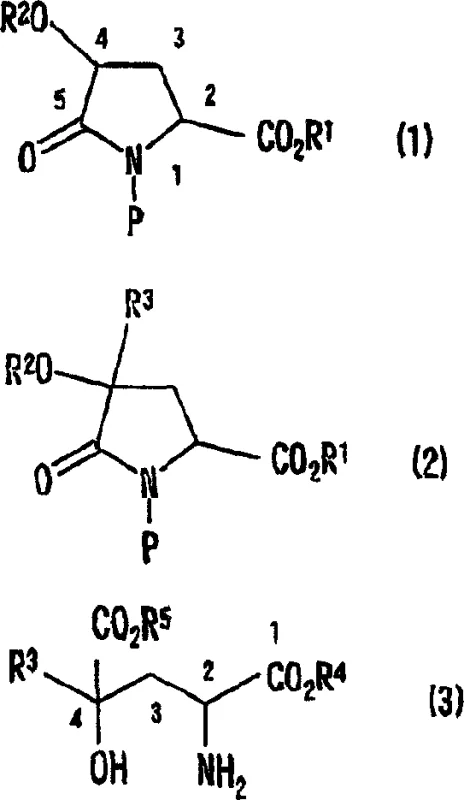
How to Synthesize Monatin Derivatives Efficiently
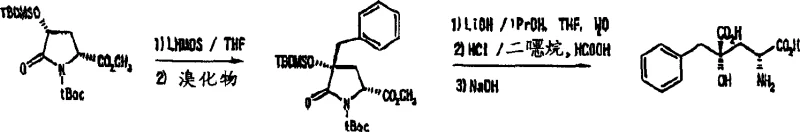
The practical implementation of this technology involves a straightforward sequence that begins with the preparation of the key 4-protected hydroxypyroglutamic acid intermediate from readily available 4-hydroxyproline. Following esterification and protection of the amino and hydroxyl groups, the substrate is subjected to alkylation conditions using an appropriate alkylating agent, such as N-protected-3-bromomethylindole. The reaction is typically conducted in anhydrous solvents like tetrahydrofuran (THF) at low temperatures to ensure selectivity. Once the alkylated intermediate is obtained, the final step involves opening the lactam ring and removing all protecting groups to yield the target glutamic acid derivative. The detailed standardized synthesis steps see the guide below for specific reaction conditions and workup procedures.
- Prepare the 4-protected hydroxypyroglutamic acid derivative starting from 4-hydroxyproline via esterification and protection group installation.
- Perform stereoselective alkylation at the 4-position using a strong base like LHMDS and an alkylating agent such as N-protected-3-halomethylindole.
- Execute hydrolysis of the lactam ring and removal of protecting groups using aqueous base and acid treatment to obtain the final glutamic acid derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis method offers tangible benefits beyond mere technical elegance. The elimination of the carboxyl reduction-oxidation cycle translates directly into substantial cost savings by reducing the number of unit operations and the quantity of reagents required. Traditional methods involving chromium-based oxidants incur high costs related to hazardous waste disposal and environmental compliance; removing these steps drastically simplifies the waste management profile of the facility. Furthermore, the use of stable, protected intermediates enhances supply chain reliability by allowing for the storage of key precursors, decoupling the production timeline from the immediate availability of fresh starting materials. This flexibility is crucial for maintaining continuous production schedules and mitigating risks associated with raw material shortages.
- Cost Reduction in Manufacturing: The streamlined process significantly lowers the cost of goods sold by removing expensive and toxic oxidation reagents like chromium trioxide. By avoiding the need to reduce and then re-oxidize the carboxyl group, the method reduces solvent usage, energy consumption, and labor hours associated with additional purification steps. This efficiency gain allows for a more competitive pricing structure for the final Monatin derivatives, making them more accessible for large-scale applications in the food and pharmaceutical sectors without compromising on quality or safety standards.
- Enhanced Supply Chain Reliability: The reliance on commercially available starting materials such as 4-hydroxyproline and standard alkylating agents ensures a stable supply base. The robustness of the alkylation reaction, which tolerates various protecting groups and conditions, minimizes the risk of batch failures due to sensitive reaction parameters. This stability supports reducing lead time for high-purity pharmaceutical intermediates, enabling manufacturers to respond more quickly to market demand fluctuations and secure long-term contracts with confidence in their ability to deliver consistent volumes.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing common organic solvents and bases that are manageable in large reactor systems. The absence of heavy metal contaminants simplifies the purification process and ensures that the final product meets strict regulatory limits for residual metals. This environmental advantage facilitates easier permitting and operation in regions with stringent ecological regulations, supporting the sustainable growth of production capacity from pilot scale to multi-ton annual commercial production while maintaining a minimal environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the method's versatility and performance. Understanding these details helps stakeholders evaluate the feasibility of integrating this technology into their existing manufacturing portfolios.
Q: What is the primary advantage of this synthesis method over conventional routes?
A: Unlike conventional methods that require reducing the carboxyl group to a hydroxymethyl group and then re-oxidizing it using toxic chromium reagents, this method allows for direct selective alkylation at the 4-position of the pyroglutamic acid derivative without altering the carboxyl functionality, significantly simplifying the process and improving safety.
Q: How is stereochemical control achieved in this alkylation reaction?
A: The reaction proceeds with high stereoselectivity because the alkylation occurs from the direction opposite to the substituent at the 2-position on the pyrrolidone ring. This ensures that the relative configuration of the resulting 2,4-disubstituted derivative matches the desired (2S,4S) or (2R,4R) configuration of Monatin.
Q: Can this method be adapted for large-scale pharmaceutical manufacturing?
A: Yes, the process utilizes standard organic solvents like THF and common bases such as lithium hexamethyldisilazide. The avoidance of hazardous oxidation steps and the use of stable intermediates make it highly suitable for commercial scale-up and regulatory compliance in API production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Monatin Derivative Supplier
As the global demand for high-intensity sweeteners and specialized amino acid intermediates continues to grow, partnering with an experienced CDMO is essential for successful commercialization. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex synthetic routes like the one described in CN1756734A can be translated into efficient industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest industry standards, guaranteeing that our clients receive materials suitable for sensitive pharmaceutical and food applications.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis method can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits specific to your production volume. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring a seamless transition from laboratory innovation to commercial reality.
