Advanced In-Situ Halogenation Strategy for Commercial Scale-up of Complex Pharmaceutical Intermediates
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes that balance high purity with operational safety and cost efficiency. A significant advancement in this domain is detailed in patent CN110903163B, which discloses a novel preparation method for tribromoneopentyl alcohol, a critical intermediate for flame retardants and the synthesis of Montelukast sodium side chains. This technology addresses long-standing challenges in halogenation chemistry by replacing hazardous gaseous reagents with a safer, in-situ generation strategy using elemental sulfur. For R&D directors and procurement specialists, this patent represents a pivotal shift towards more sustainable and scalable manufacturing protocols, offering a pathway to high-purity products (>99.5%) with yields exceeding 90% while mitigating the severe environmental liabilities associated with traditional phosphorus-based or high-pressure gas methods.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of tribromoneopentyl alcohol has been plagued by significant technical and economic hurdles that hinder efficient commercial scale-up. Traditional routes often rely on the direct introduction of hydrogen bromide gas, which necessitates operation under elevated pressures (0.6-0.8 MPa), imposing stringent requirements on reactor integrity and safety systems that drive up capital expenditure. Alternatively, methods utilizing phosphorus tribromide, while operationally simpler, introduce severe downstream complications; they generate wastewater with high phosphorus content and elevated Chemical Oxygen Demand (COD), creating a complex waste stream that is both costly and difficult to treat to meet modern environmental standards. Furthermore, processes employing urea or hydrazine hydrate as reducing agents can lead to uncontrolled gas evolution during scale-up, resulting in the loss of valuable bromine resources and inconsistent reaction control, ultimately compromising the overall yield and economic viability of the production line.
The Novel Approach
The methodology outlined in patent CN110903163B fundamentally reengineers the bromination step by utilizing elemental sulfur powder as a reducing agent to generate hydrogen bromide directly within the reaction mixture. This in-situ generation eliminates the need for external high-pressure gas feeds and avoids the introduction of phosphorus contaminants entirely. By conducting the reaction in acetic acid at moderate temperatures (110-130°C), the process ensures a controlled release of the brominating species, minimizing side reactions and thermal runaways. This approach not only simplifies the equipment requirements—allowing for the use of standard atmospheric pressure reactors—but also transforms the waste profile; the resulting wastewater is alkaline and readily treatable, while valuable solvents like acetic acid and methanol are efficiently recovered and recycled, creating a circular economy within the manufacturing process that drastically reduces raw material overheads.
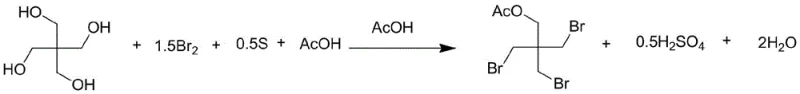
Mechanistic Insights into Sulfur-Mediated In-Situ Halogenation
The core innovation of this synthesis lies in the elegant redox chemistry between elemental sulfur and molecular bromine within an acidic medium. In the initial substitution phase, sulfur acts as a sacrificial reducing agent, reacting with bromine to generate hydrogen bromide (HBr) in situ, which then serves as the active nucleophile for the substitution of hydroxyl groups on the pentaerythritol backbone. This mechanism ensures a steady, controlled concentration of HBr throughout the reaction, preventing the localized excesses that often lead to over-bromination or degradation in gas-phase processes. The stoichiometry is carefully balanced, typically employing a molar ratio of bromine to pentaerythritol between 1.5:1 and 2.0:1, with sulfur present in slight excess relative to the bromine to ensure complete conversion. The byproduct of this redox step is sulfuric acid, which remains dissolved in the acetic acid medium and is subsequently neutralized, avoiding the formation of solid precipitates that could complicate filtration or heat transfer during the exothermic reaction phase.
Following the bromination, the process transitions to an alcoholysis step where the acetylated intermediate undergoes transesterification with methanol. This step is crucial for revealing the final hydroxyl group required for the tribromoneopentyl alcohol structure while generating methyl acetate as a valuable, separable byproduct. The reaction conditions are optimized to drive this equilibrium forward, typically heating the mixture to 80°C in the presence of excess methanol. The mechanistic pathway ensures that the three bromomethyl groups remain intact and stable under these mild alcoholic conditions, preserving the structural integrity of the molecule. This two-stage cascade—substitution followed by alcoholysis—allows for the sequential removal of volatile components (acetic acid, HBr, methanol, methyl acetate) via reduced pressure distillation, effectively purifying the crude product before the final crystallization, which is key to achieving the reported purity levels of greater than 99.5%.
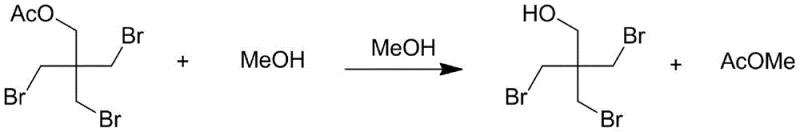
How to Synthesize Tribromoneopentyl Alcohol Efficiently
The practical execution of this synthesis requires precise control over addition rates and temperature profiles to maximize safety and yield. The process begins with the charging of pentaerythritol, sulfur powder, and acetic acid into a reactor equipped with efficient agitation and cooling capabilities. Liquid bromine is then added dropwise, with strict temperature control maintained below 50°C to manage the initial exotherm of the HBr generation. Once the addition is complete, the mixture is heated to reflux conditions (110-130°C) to drive the substitution to completion. After recovering the volatile acids, methanol is introduced for the alcoholysis step, followed by a workup procedure involving neutralization with sodium carbonate and extraction with an alkane solvent such as n-heptane or n-hexane. The detailed standardized synthesis steps, including specific molar ratios and crystallization parameters, are provided in the guide below.
- Charge pentaerythritol, sulfur powder, and acetic acid into a reactor, then add liquid bromine dropwise while controlling temperature below 50°C to generate hydrogen bromide in situ.
- Heat the reaction mixture to 110-130°C to complete the substitution, recover acetic acid and HBr under reduced pressure, and add methanol for alcoholysis at 80°C.
- Neutralize the residue with sodium carbonate solution, extract with alkane solvent, cool the organic phase to 0-5°C for crystallization, and filter to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this sulfur-mediated technology offers profound strategic advantages that extend beyond simple yield improvements. The shift away from specialized, high-pressure equipment and hazardous gas logistics significantly lowers the barrier to entry for manufacturing this intermediate, allowing for more flexible production scheduling and reduced dependency on specialized infrastructure. The ability to recover and reuse major solvents like acetic acid and methanol creates a substantial reduction in variable operating costs, insulating the production budget from volatility in solvent markets. Furthermore, the generation of methyl acetate as a saleable byproduct introduces a new revenue stream that can offset production costs, enhancing the overall margin profile of the manufacturing campaign.
- Cost Reduction in Manufacturing: The elimination of expensive phosphorus tribromide and the replacement of high-pressure hydrogen bromide gas with low-cost elemental sulfur and liquid bromine results in a drastic reduction in raw material expenditures. Additionally, the simplified workup procedure, which avoids complex degreasing steps and extensive water washing, reduces utility consumption and labor hours, leading to a leaner and more cost-effective production cycle that maximizes return on investment.
- Enhanced Supply Chain Reliability: By utilizing commodity chemicals such as sulfur powder, acetic acid, and methanol, the supply chain becomes far more resilient to disruptions compared to processes relying on specialized reagents like tetrabutyl titanate or anhydrous HBr gas. The robustness of the reaction conditions also means that production can be scaled up with greater confidence, ensuring consistent delivery schedules and reducing the risk of batch failures that could interrupt the supply of critical downstream pharmaceutical intermediates.
- Scalability and Environmental Compliance: The process generates alkaline wastewater that is compatible with standard treatment facilities, avoiding the high costs and regulatory hurdles associated with treating high-phosphorus or high-COD effluents. This environmental compatibility facilitates easier permitting for capacity expansion and ensures long-term operational continuity in regions with strict environmental regulations, making it an ideal candidate for sustainable, large-scale commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms legacy technologies in terms of safety, purity, and operational simplicity. Understanding these nuances is essential for technical teams evaluating the feasibility of integrating this process into existing manufacturing portfolios.
Q: How does the sulfur-mediated method improve safety compared to traditional HBr gas processes?
A: Traditional methods often require handling pressurized hydrogen bromide gas (0.6-0.8 MPa), which poses significant equipment and safety risks. The patented sulfur-mediated route generates HBr in situ at atmospheric pressure, eliminating the need for high-pressure gas cylinders and specialized pressure-rated reactors, thereby drastically simplifying operational safety protocols.
Q: What are the environmental advantages regarding wastewater treatment in this new process?
A: Unlike processes using phosphorus tribromide which generate high-phosphorus, high-COD wastewater that is difficult to treat, this method produces alkaline wastewater after neutralization. This effluent is compatible with standard sewage treatment systems, significantly reducing the complexity and cost of environmental compliance and waste disposal.
Q: Can the solvents used in this synthesis be recovered and reused?
A: Yes, the process is designed for high atom economy and solvent efficiency. Acetic acid and hydrobromic acid are recovered via reduced pressure distillation after the substitution step, and methanol along with the byproduct methyl acetate are recovered after alcoholysis. This closed-loop solvent management substantially lowers raw material consumption.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tribromoneopentyl Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your final pharmaceutical and specialty chemical products. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of tribromoneopentyl alcohol meets the exacting standards required for sensitive applications such as Montelukast synthesis and advanced flame retardant formulations.
We invite you to collaborate with us to leverage this advanced synthetic technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your product quality while driving down your total cost of ownership.
