Revolutionizing Tribromoneopentyl Alcohol Production: A Technical Breakthrough for Industrial Scale-Up
The chemical manufacturing landscape for high-performance flame retardant intermediates is undergoing a significant transformation, driven by the urgent need for safer, more environmentally compliant, and cost-effective synthetic routes. Patent CN110903163A represents a pivotal advancement in this domain, introducing a novel preparation method for tribromoneopentyl alcohol that fundamentally reimagines the bromination mechanism. Unlike conventional methodologies that rely on hazardous pressurized gases or expensive phosphorus reagents, this innovation leverages elemental sulfur as a reducing agent to generate hydrobromic acid in situ. This strategic shift not only mitigates the severe safety risks associated with handling anhydrous hydrogen bromide gas but also streamlines the downstream purification process. For R&D directors and procurement specialists alike, this patent offers a compelling blueprint for achieving product purity exceeding 99.5% while simultaneously addressing the growing regulatory pressures surrounding industrial wastewater management. The implications of this technology extend beyond mere laboratory curiosity; it provides a robust framework for the reliable supply of critical intermediates used in elastomers, coatings, and even pharmaceutical applications such as the synthesis of montelukast sodium side chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of tribromoneopentyl alcohol has been plagued by significant engineering and environmental hurdles that hinder efficient commercial scale-up. Traditional protocols, such as those disclosed in earlier patent literature, often necessitate the direct introduction of hydrogen bromide gas into the reaction vessel under pressures ranging from 0.6 to 0.8 MPa. This requirement imposes stringent demands on equipment integrity, necessitating expensive pressure-rated reactors and specialized corrosion-resistant lining to prevent catastrophic failure. Furthermore, alternative methods utilizing phosphorus tribromide introduce substantial quantities of phosphorus into the waste stream, resulting in wastewater with high chemical oxygen demand (COD) and complex composition that is notoriously difficult and costly to treat. Other approaches employing urea or hydrazine hydrate as reducing agents suffer from uncontrolled gas evolution, specifically nitrogen, which can entrain valuable hydrogen bromide out of the system, leading to inconsistent stoichiometry and reduced atom economy. These legacy processes create a bottleneck for supply chain heads, as the complexity of waste treatment and the volatility of raw material costs directly impact the continuity and reliability of the final product supply.
The Novel Approach
In stark contrast to these cumbersome legacy systems, the methodology outlined in CN110903163A introduces a paradigm shift by utilizing sulfur powder to reduce bromine directly within the reaction medium. This elegant solution eliminates the need for external high-pressure gas feeds, allowing the bromination to proceed under much milder and safer conditions. By generating the reactive hydrobromic acid species in situ, the process ensures a controlled and steady release of the brominating agent, which significantly reduces the formation of unwanted side products and improves the overall selectivity of the reaction. The use of acetic acid as a solvent further enhances the solubility of the reactants and facilitates the recovery of valuable byproducts through simple distillation. From a commercial perspective, this approach drastically simplifies the operational workflow, removing the need for specialized high-pressure infrastructure and reducing the burden on environmental compliance teams. The result is a synthesis route that is not only chemically superior in terms of yield and purity but also economically advantageous due to the utilization of low-cost, widely available commodity chemicals like sulfur and acetic acid.
Mechanistic Insights into Sulfur-Mediated In-Situ Bromination
The core innovation of this patented process lies in the intricate interplay between elemental sulfur and molecular bromine within an acetic acid medium. When sulfur powder is introduced to the reaction mixture containing bromine, it acts as a potent reducing agent, effectively converting the molecular bromine into reactive hydrobromic acid species without the need for external water sources initially. This in-situ generation creates a highly reactive environment that facilitates the nucleophilic substitution of the hydroxyl groups on the pentaerythritol backbone. The reaction kinetics are carefully managed by controlling the addition rate of bromine and maintaining the temperature below 50°C during the initial exothermic phase, preventing thermal runaway and ensuring the formation of the desired tri-brominated intermediate rather than over-brominated byproducts. The subsequent heating to 110-130°C drives the substitution to completion, ensuring that the steric hindrance of the neopentyl structure is overcome efficiently. This mechanistic precision is crucial for R&D teams aiming to replicate high-purity results, as it minimizes the formation of dibromo- or tetrabromo- impurities that are difficult to separate in later stages.
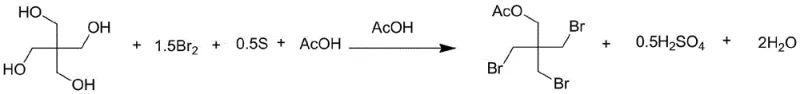
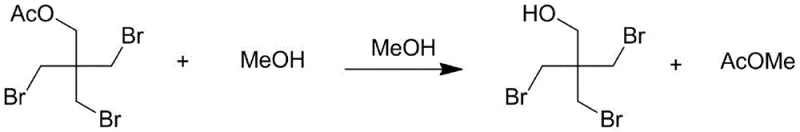
Following the initial bromination and substitution phase, the process transitions into a critical alcoholysis step designed to deprotect the acetate groups and finalize the molecular structure. The intermediate triacetate species formed during the heating phase undergoes transesterification when treated with methanol. This step is vital for restoring the primary hydroxyl group essential for the molecule's functionality as a reactive flame retardant or pharmaceutical intermediate. The use of methanol serves a dual purpose: it acts as both the reactant for alcoholysis and a solvent that aids in the precipitation of inorganic salts during the subsequent neutralization phase. The recovery of methyl acetate, a valuable solvent byproduct, during the distillation phase further underscores the economic efficiency of this route. By carefully controlling the pH during the neutralization with sodium carbonate, the process ensures that any residual acidic components are safely quenched without inducing hydrolysis of the sensitive carbon-bromine bonds. This delicate balance of acidity and nucleophilicity is what allows the process to achieve yields exceeding 90% while maintaining a purity profile that meets the rigorous standards of the fine chemical industry.
How to Synthesize Tribromoneopentyl Alcohol Efficiently
Implementing this synthesis route requires precise adherence to the stoichiometric ratios and thermal profiles defined in the patent to ensure optimal conversion and safety. The process begins with the careful charging of pentaerythritol, sulfur powder, and acetic acid, followed by the controlled dropwise addition of liquid bromine to manage the exotherm. Detailed standardized operating procedures regarding temperature ramping, vacuum levels for distillation, and crystallization cooling rates are essential for reproducibility. For technical teams looking to adopt this methodology, the following guide outlines the critical operational parameters derived from the patent examples.
- Charge pentaerythritol, sulfur powder, and acetic acid into a reactor, then add liquid bromine dropwise while maintaining temperature below 50°C to generate HBr in situ.
- Heat the mixture to 110-130°C for substitution, recover acetic acid and HBr under reduced pressure, and proceed to alcoholysis with methanol.
- Neutralize the residue with sodium carbonate, extract with alkane solvents, and crystallize the organic phase at 0-5°C to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this sulfur-mediated synthesis route offers profound strategic advantages that extend well beyond the laboratory bench. The primary value proposition lies in the drastic simplification of the raw material supply chain; by replacing specialized, high-pressure gases or expensive phosphorus reagents with commodity sulfur and acetic acid, manufacturers can insulate themselves from the volatility of niche chemical markets. This shift to widely available feedstocks ensures a more resilient supply chain, reducing the risk of production stoppages due to raw material shortages. Furthermore, the elimination of complex wastewater treatment requirements associated with phosphorus or nitrogen-heavy byproducts translates into significant operational expenditure savings. The ability to treat wastewater directly in standard sewage systems removes the need for costly, dedicated effluent treatment plants, thereby lowering the barrier to entry for scaling production capacity. These factors combined create a robust economic model that supports long-term price stability and reliable delivery schedules for downstream customers in the flame retardant and pharmaceutical sectors.
- Cost Reduction in Manufacturing: The transition to sulfur powder as a reducing agent eliminates the capital expenditure associated with high-pressure reactor vessels and the ongoing costs of handling hazardous compressed gases. Additionally, the recovery and reuse of acetic acid and methanol, along with the saleable byproduct methyl acetate, create a circular economy within the plant that significantly lowers the net cost per kilogram of the final product. This efficiency allows for competitive pricing strategies without compromising on margin, providing a distinct advantage in price-sensitive markets.
- Enhanced Supply Chain Reliability: By relying on commodity chemicals that are produced globally in massive quantities, the risk of supply disruption is minimized compared to processes dependent on specialized reagents like phosphorus tribromide. The simplified process flow also reduces the number of unit operations required, decreasing the likelihood of mechanical failures or bottlenecks that could delay shipments. This reliability is critical for just-in-time manufacturing environments where consistent feedstock availability is paramount for maintaining production schedules.
- Scalability and Environmental Compliance: The inherent safety of the atmospheric pressure operation and the benign nature of the waste stream make this process exceptionally easy to scale from pilot batches to multi-ton commercial production. Regulatory compliance is streamlined as the wastewater profile is less toxic and easier to manage, reducing the administrative burden and potential fines associated with environmental violations. This scalability ensures that suppliers can rapidly respond to surges in market demand for high-purity flame retardant intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this advanced synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity for stakeholders evaluating this route for potential adoption or sourcing.
Q: How does the sulfur-mediated method improve safety compared to traditional HBr gas processes?
A: Traditional methods require handling pressurized hydrogen bromide gas (0.6-0.8 MPa), posing significant corrosion and leakage risks. The patented sulfur-mediated approach generates HBr in situ within the liquid phase, eliminating the need for high-pressure gas cylinders and specialized pressure-rated reactors, thereby drastically enhancing operational safety.
Q: What are the wastewater treatment advantages of this new synthesis route?
A: Unlike prior art using phosphorus tribromide which generates high-phosphorus wastewater, or urea-based methods creating complex nitrogenous waste, this process yields alkaline wastewater after neutralization. The absence of heavy metals and complex organic byproducts allows for direct treatment in standard sewage systems, significantly lowering environmental compliance costs.
Q: Is this synthesis route suitable for large-scale commercial production of flame retardant intermediates?
A: Yes, the process utilizes commodity chemicals like sulfur powder and acetic acid, ensuring raw material availability. The reaction conditions are mild (atmospheric pressure after initial addition), and the work-up involves standard distillation and crystallization, making it highly scalable from pilot plants to multi-ton annual production capacities without complex engineering bottlenecks.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tribromoneopentyl Alcohol Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic routes like the one described in CN110903163A requires a partner with deep technical expertise and proven industrial capability. As a leading CDMO and manufacturer, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practice. Our facilities are equipped with state-of-the-art corrosion-resistant reactors and rigorous QC labs capable of verifying stringent purity specifications, including the >99.5% purity benchmark set by this new method. We understand the critical nature of supply continuity for your downstream applications, whether for high-performance polymers or pharmaceutical intermediates, and we are committed to delivering consistent quality that meets global regulatory standards.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the potential economic impact of switching to this sulfur-mediated process. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your volume needs, ensuring that your transition to this superior grade of tribromoneopentyl alcohol is seamless and commercially viable.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
