Advanced Catalytic Oxidation for High-Purity 3-Mercaptoindole Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust and environmentally sustainable pathways for constructing complex heterocyclic scaffolds, particularly those serving as critical building blocks for bioactive molecules. Patent CN108586310B introduces a transformative catalytic oxidation synthesis method for 3-mercaptoindole compounds, addressing long-standing challenges in efficiency and ecological impact. This technology leverages a sophisticated catalytic system utilizing potassium iodide and sodium nitrite to facilitate the coupling of indole derivatives with thiosulfates under an oxygen atmosphere. By shifting away from stoichiometric oxidants and harsh conditions, this method offers a compelling value proposition for manufacturers aiming to optimize their supply chains for high-purity pharmaceutical intermediates. The strategic implementation of molecular oxygen as the terminal oxidant not only aligns with green chemistry principles but also drastically simplifies the post-reaction workup, making it an ideal candidate for commercial scale-up of complex heterocyclic intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-mercaptoindole derivatives has relied heavily on protocols that utilize dimethyl sulfoxide (DMSO) as both the solvent and the oxidant, often in conjunction with iodine catalysts. While effective in laboratory settings, these conventional methods present significant drawbacks when translated to industrial manufacturing environments. The primary concern lies in the generation of dimethyl sulfide as a reaction byproduct, a compound notorious for its potent, unpleasant odor and environmental toxicity, which necessitates expensive scrubbing systems and rigorous waste management protocols. Furthermore, DMSO possesses a high boiling point and strong hydrophilicity, making its removal from the final product energetically intensive and technically challenging. This often leads to prolonged processing times and increased utility costs, creating a bottleneck in the production of reliable pharmaceutical intermediate supplier pipelines. Additionally, the use of stoichiometric amounts of oxidants can lead to atom inefficiency, generating substantial chemical waste that contradicts modern sustainability mandates.
The Novel Approach
In stark contrast, the methodology disclosed in CN108586310B represents a paradigm shift by employing molecular oxygen as a clean, renewable, and cost-effective terminal oxidant. This novel approach utilizes a catalytic amount of potassium iodide and sodium nitrite in the presence of acetic acid to drive the reaction forward with exceptional selectivity. The substitution of DMSO with acetonitrile or similar organic solvents eliminates the formation of malodorous sulfur byproducts, replacing them with water as the sole oxidation waste product. This fundamental change in reaction design significantly lowers the environmental footprint and reduces the complexity of the purification process. The mild reaction conditions, operating at atmospheric pressure and temperatures between 50°C and 80°C, further enhance the safety profile and operational feasibility of the process. This innovation directly supports cost reduction in API manufacturing by minimizing raw material consumption and waste disposal fees while maintaining high product integrity.
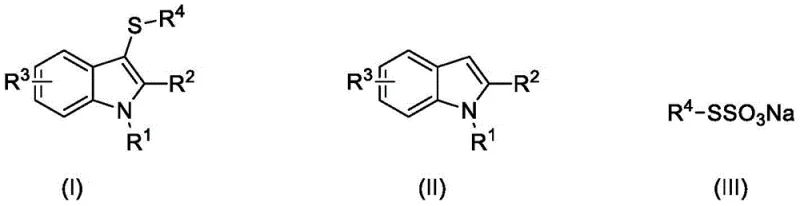
Mechanistic Insights into KI/NaNO2-Catalyzed Oxidative Coupling
The core of this synthetic breakthrough lies in the intricate interplay between the iodide catalyst and the nitrite co-catalyst under an oxygen atmosphere. Mechanistically, the sodium nitrite serves to oxidize the iodide ions into active electrophilic iodine species in situ, which then activate the indole ring at the C3 position. This activation allows for the nucleophilic attack by the thiosulfate anion, forming the critical carbon-sulfur bond. The continuous regeneration of the active iodine species by molecular oxygen ensures that the catalytic cycle proceeds efficiently without the need for excessive catalyst loading. This mechanism avoids the formation of radical intermediates that often lead to polymerization or non-selective side reactions, thereby ensuring a clean impurity profile. The presence of acetic acid acts as a crucial promoter, likely stabilizing the transition states and facilitating proton transfer steps essential for the turnover of the catalytic cycle. Understanding this mechanistic pathway is vital for R&D teams aiming to adapt this chemistry for diverse substrate libraries.
From an impurity control perspective, the mildness of this oxidative system is paramount. Traditional strong oxidants can often over-oxidize the sulfur moiety to sulfoxides or sulfones, or degrade the sensitive indole nucleus. However, the controlled potential of the in situ generated iodine species prevents such over-oxidation, preserving the integrity of the thioether linkage. Furthermore, the use of thiosulfates as sulfur sources offers distinct advantages over thiols or disulfides; thiosulfates are odorless, stable solids that are easier to handle and store, reducing occupational health hazards. The regioselectivity for the C3 position of the indole is inherently driven by the electronic properties of the heterocycle, which are amplified by the specific catalytic environment. This high degree of selectivity minimizes the formation of regioisomers, simplifying the downstream purification and ensuring that the final high-purity 3-mercaptoindole derivatives meet stringent quality specifications required for drug substance manufacturing.
How to Synthesize 3-Mercaptoindole Derivatives Efficiently
The practical execution of this synthesis involves a straightforward procedure that balances reaction kinetics with operational simplicity. The process begins by dissolving the indole substrate and the appropriate sodium thiosulfate derivative in acetonitrile, followed by the addition of the catalytic system comprising potassium iodide and sodium nitrite. Acetic acid is then introduced to the mixture, which is subsequently stirred under an oxygen balloon or air flow at temperatures ranging from 50°C to 80°C for a period of 10 to 24 hours. The detailed standardized synthesis steps see the guide below, which outlines the precise molar ratios and workup procedures optimized for maximum yield. Following the reaction, the mixture is treated with aqueous sodium thiosulfate to quench residual oxidants, extracted with dichloromethane, and purified via column chromatography to isolate the target compound.
- Combine indole substrate, sodium thiosulfate derivative, potassium iodide (KI), and sodium nitrite (NaNO2) in an organic solvent such as acetonitrile.
- Introduce acetic acid as a promoter and maintain the reaction mixture under an oxygen atmosphere at a controlled temperature between 50°C and 80°C.
- Upon completion, quench the reaction with sodium thiosulfate solution, extract the organic layer with dichloromethane, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic oxidation technology translates into tangible strategic benefits that extend beyond mere chemical efficacy. The elimination of expensive and hazardous oxidants like DMSO, coupled with the use of commodity chemicals such as potassium iodide and sodium nitrite, drives a significant reduction in raw material costs. The simplified workup procedure, which avoids difficult solvent removal steps associated with high-boiling polar solvents, reduces energy consumption and shortens the overall production cycle time. This efficiency gain enhances supply chain reliability by allowing for faster turnaround times and greater flexibility in meeting fluctuating market demands. Moreover, the use of oxygen as a reagent removes the logistical burden of sourcing and storing specialized oxidizing agents, streamlining the inventory management process for manufacturing facilities.
- Cost Reduction in Manufacturing: The economic viability of this process is underpinned by the replacement of stoichiometric oxidants with catalytic amounts of inexpensive salts and free oxygen from the air. This shift eliminates the cost associated with purchasing, handling, and disposing of large quantities of chemical oxidants. Additionally, the avoidance of DMSO removes the need for energy-intensive distillation processes required to remove high-boiling solvents, leading to substantial utility savings. The high atom economy of the reaction ensures that a greater proportion of the starting materials are converted into the desired product, minimizing waste disposal costs and maximizing the return on investment for every kilogram of raw material purchased.
- Enhanced Supply Chain Reliability: The reliance on widely available and stable raw materials, such as indoles and sodium thiosulfates, mitigates the risk of supply disruptions often associated with specialty reagents. Thiosulfates are commercially abundant and do not suffer from the stability issues or odor problems characteristic of thiols and disulfides, ensuring consistent quality and availability. The robustness of the reaction conditions, which tolerate a wide range of functional groups and operate at atmospheric pressure, means that the process can be easily transferred between different manufacturing sites without requiring specialized high-pressure equipment. This flexibility strengthens the resilience of the supply chain against geopolitical or logistical shocks.
- Scalability and Environmental Compliance: Scaling this reaction from laboratory to commercial production is facilitated by the use of common solvents like acetonitrile and the absence of exothermic hazards associated with strong oxidants. The generation of water as the primary byproduct aligns perfectly with increasingly stringent environmental regulations, reducing the regulatory burden and permitting costs for manufacturing plants. The process inherently supports green chemistry initiatives, allowing companies to market their products as sustainably produced, which is a growing requirement for partnerships with major multinational pharmaceutical corporations. This environmental compliance future-proofs the manufacturing asset against evolving global standards.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this catalytic oxidation method. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing clarity on reaction parameters and scope. Understanding these details is essential for process chemists evaluating the feasibility of this route for specific target molecules.
Q: What are the primary advantages of using molecular oxygen in this synthesis?
A: Using molecular oxygen as the terminal oxidant significantly reduces environmental costs compared to traditional oxidants like DMSO. The only byproduct is water, eliminating toxic waste streams and simplifying downstream purification processes.
Q: Which catalysts are employed in this catalytic oxidation system?
A: The process utilizes a dual-catalyst system comprising potassium iodide (KI) and sodium nitrite (NaNO2). This combination effectively generates the active iodine species required for the oxidative coupling under mild conditions.
Q: What is the typical reaction temperature range for this protocol?
A: The reaction proceeds efficiently under mild thermal conditions, typically requiring temperatures between 50°C and 80°C. This moderate range ensures energy efficiency and minimizes thermal degradation of sensitive functional groups.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Mercaptoindole Supplier
As the demand for high-quality heterocyclic building blocks continues to rise, partnering with an experienced CDMO is crucial for navigating the complexities of process development and commercialization. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our commitment to quality is evidenced by our stringent purity specifications and rigorous QC labs, which guarantee that every batch of 3-mercaptoindole derivatives meets the exacting standards required for pharmaceutical applications. We leverage advanced analytical techniques to monitor impurity profiles and ensure batch-to-batch consistency, providing you with a reliable foundation for your drug development programs.
We invite you to engage with our technical procurement team to discuss how this innovative catalytic oxidation technology can be tailored to your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits this route offers for your specific portfolio. We encourage potential partners to contact us to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions based on concrete technical evidence. Let us collaborate to accelerate your timeline to market with efficient, sustainable, and cost-effective chemical solutions.
