Advanced Catalytic Oxidation Strategy for Scalable 3-Mercaptoindole Pharmaceutical Intermediates Production
Advanced Catalytic Oxidation Strategy for Scalable 3-Mercaptoindole Pharmaceutical Intermediates Production
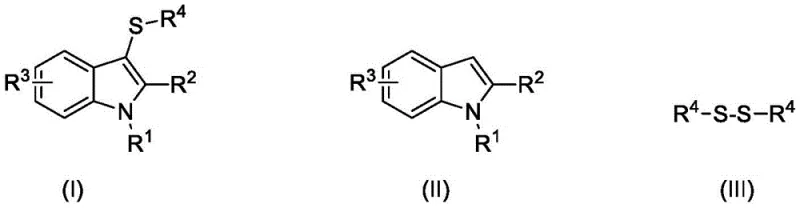
The pharmaceutical industry continuously seeks robust and environmentally benign synthetic routes for bioactive heterocycles, particularly 3-mercaptoindole derivatives which serve as critical scaffolds in drug discovery. Patent CN108440375B discloses a groundbreaking catalytic oxidation synthesis method that utilizes disulfides as sulfur sources to construct these valuable motifs efficiently. This technology represents a significant leap forward in green chemistry, replacing hazardous reagents with molecular oxygen and inexpensive inorganic catalysts. For R&D directors and procurement specialists, this method offers a compelling alternative to traditional pathways by ensuring high atom economy and simplified downstream processing. The ability to operate under mild conditions while maintaining high selectivity makes this process highly attractive for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 3-mercaptoindole compounds has relied heavily on nucleophilic substitution reactions involving thiols or sulfinic acids, which present severe logistical and safety challenges. Thiol reagents are notoriously malodorous and toxic, requiring specialized containment infrastructure that drastically increases capital expenditure for manufacturing facilities. Furthermore, many conventional protocols depend on stoichiometric amounts of harsh oxidants or expensive transition metal catalysts that leave behind difficult-to-remove heavy metal residues. Previous attempts to use dimethyl sulfoxide (DMSO) as an oxidant have been plagued by the formation of dimethyl sulfide, a volatile and unpleasant byproduct that complicates waste management and violates strict environmental regulations. These factors collectively hinder the cost reduction in API manufacturing and limit the scalability of existing processes.
The Novel Approach
The innovative methodology described in the patent overcomes these barriers by employing a dual-catalyst system of potassium iodide and sodium nitrite with molecular oxygen as the terminal oxidant. This approach utilizes stable disulfide solids rather than volatile thiols, significantly enhancing operator safety and supply chain reliability. The reaction proceeds smoothly in common organic solvents like acetonitrile or ethanol at moderate temperatures ranging from 40 to 80 degrees Celsius under atmospheric pressure. By shifting to a catalytic cycle driven by clean oxygen, the process generates water as the sole byproduct, aligning perfectly with modern sustainability goals. This transformation not only simplifies the reaction setup but also streamlines the purification workflow, making it an ideal candidate for reliable pharmaceutical intermediate supplier operations seeking to optimize their production lines.
Mechanistic Insights into KI and NaNO2 Catalyzed Oxidative Coupling
The core of this synthetic breakthrough lies in the synergistic interaction between the iodide/nitrite catalyst pair and the oxygen atmosphere to generate active electrophilic sulfur species in situ. Mechanistically, the potassium iodide and sodium nitrite in the presence of acetic acid likely generate reactive iodine species that activate the disulfide bond, facilitating the cleavage of the S-S bond to form a transient sulfenyl iodide or similar electrophile. This activated sulfur species then undergoes regioselective electrophilic substitution at the C3 position of the indole ring, which is the most nucleophilic site on the heterocycle. The use of molecular oxygen serves to regenerate the active catalytic species, closing the catalytic loop without consuming additional chemical oxidants. This elegant mechanism ensures that the reaction remains highly efficient and minimizes the formation of over-oxidized byproducts or polymeric tars that often plague indole functionalization reactions.
From an impurity control perspective, the mild acidic conditions provided by acetic acid and the moderate temperature range are crucial for maintaining the integrity of sensitive functional groups on the indole scaffold. Unlike harsh Lewis acid conditions that might promote decomposition or rearrangement, this buffered system allows for the tolerance of diverse substituents such as halogens, cyano groups, and alkoxy chains. The selectivity for the C3 position is inherently high due to the electronic nature of the indole ring, reducing the need for protecting group strategies that add steps and cost. For quality assurance teams, this means a cleaner crude reaction profile, which translates to higher isolated yields and reduced solvent consumption during chromatographic purification. The robustness of this mechanism against varying electronic demands of the substrates ensures consistent quality across different batches of high-purity OLED material or pharmaceutical precursors.
How to Synthesize 3-Mercaptoindole Efficiently
The practical implementation of this synthesis involves a straightforward one-pot procedure that is amenable to both laboratory optimization and industrial replication. The process begins by charging the reactor with the indole substrate, the chosen disulfide, and the catalytic amounts of potassium iodide and sodium nitrite in a suitable solvent system. Acetic acid is introduced as a promoter to facilitate the generation of the active catalytic species, and the headspace is purged with oxygen to ensure an oxidative environment. The reaction mixture is then heated to the optimal temperature window, typically around 60 degrees Celsius, and stirred for a duration sufficient to reach full conversion, usually between 1 to 8 hours depending on the substrate reactivity. Detailed standardized synthesis steps see the guide below.
- Combine indole substrate, disulfide source, KI, and NaNO2 in acetonitrile or ethanol solvent.
- Add acetic acid auxiliary and replace atmosphere with oxygen gas at normal pressure.
- Heat mixture to 40-80°C for 1-8 hours, then purify via extraction and column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this catalytic oxidation technology offers substantial strategic advantages regarding cost structure and operational continuity. The shift away from precious metal catalysts and hazardous thiol reagents fundamentally alters the cost drivers of the manufacturing process, removing the volatility associated with rare earth metals and specialized odor-control infrastructure. The use of commodity chemicals like potassium iodide, sodium nitrite, and acetic acid ensures a stable and predictable supply base, mitigating the risk of raw material shortages that can disrupt production schedules. Furthermore, the elimination of toxic byproducts reduces the burden on waste treatment facilities, leading to significant operational expenditure savings over the lifecycle of the product. This process stability is essential for reducing lead time for high-purity pharmaceutical intermediates and ensuring consistent delivery to downstream clients.
- Cost Reduction in Manufacturing: The replacement of expensive transition metal catalysts with abundant inorganic salts like KI and NaNO2 results in a drastic reduction in raw material costs. Additionally, the use of molecular oxygen from air as the oxidant eliminates the need for purchasing and storing hazardous chemical oxidants, further lowering the cost of goods sold. The simplified workup procedure, which avoids complex metal scavenging steps, reduces solvent usage and labor hours, contributing to overall margin improvement. These factors combine to create a highly competitive cost structure for the commercial scale-up of complex pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: Utilizing disulfides as sulfur sources instead of thiols addresses a major logistical pain point, as disulfides are generally stable solids with long shelf lives and no offensive odor. This stability simplifies storage and transportation requirements, allowing for bulk purchasing and inventory buffering without degradation concerns. The reliance on widely available industrial solvents like acetonitrile and ethanol ensures that the supply chain is not dependent on niche reagents that might face allocation issues. This robustness guarantees a continuous supply of critical building blocks for global pharmaceutical partners.
- Scalability and Environmental Compliance: The reaction operates at atmospheric pressure and moderate temperatures, removing the need for high-pressure autoclaves or cryogenic cooling systems, which simplifies equipment requirements for scale-up. The generation of water as the primary byproduct aligns with increasingly stringent environmental regulations, minimizing the risk of regulatory fines or shutdowns due to emissions. The absence of heavy metals in the catalyst system simplifies the validation process for GMP manufacturing, accelerating the time to market for new drug candidates. This green profile enhances the corporate sustainability metrics of any organization adopting this technology.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this catalytic oxidation method. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide clarity for potential licensees and manufacturing partners. Understanding these nuances is critical for assessing the feasibility of integrating this route into existing production workflows. The responses cover aspects ranging from substrate compatibility to safety protocols, ensuring a comprehensive understanding of the technology's capabilities.
Q: Why are disulfides preferred over thiols as sulfur sources in this synthesis?
A: Disulfides are significantly more stable and less malodorous than thiol compounds, which greatly improves operational safety and environmental compliance during large-scale manufacturing.
Q: What are the environmental benefits of using molecular oxygen as the oxidant?
A: Using molecular oxygen eliminates the generation of toxic byproducts like dimethyl sulfide associated with DMSO oxidants, resulting in water as the only byproduct and reducing waste treatment costs.
Q: Is the removal of the KI and NaNO2 catalyst system difficult?
A: No, the catalyst system consists of inexpensive inorganic salts that are easily removed during the aqueous workup and extraction phases, simplifying the purification process.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Mercaptoindole Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this catalytic oxidation technology for the production of high-value sulfur-containing heterocycles. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from lab bench to pilot plant is seamless. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications required for clinical and commercial grade materials. We are committed to leveraging this green synthesis route to deliver cost-effective and environmentally responsible solutions for our global clientele.
We invite you to engage with our technical procurement team to discuss how this novel synthesis method can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain detailed insights into the economic benefits of switching to this catalytic system for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that demonstrate our capability to support your long-term manufacturing goals with reliability and excellence.
