Advanced Three-Step Synthesis of Antiviral Drug Intermediates for Commercial Scale-Up
The pharmaceutical industry is constantly seeking robust and scalable pathways for the production of critical antiviral agents, particularly for Hepatitis C treatments where demand remains steady despite the emergence of newer therapies. A significant breakthrough in this domain is detailed in patent CN115322167B, which discloses a novel preparation method for a key antiviral drug intermediate, specifically related to the synthesis of Boceprevir. This patent outlines a streamlined three-step synthetic route that fundamentally shifts away from the hazardous and costly methodologies of the past. By leveraging a sulfur ylide strategy, the inventors have achieved a process that not only improves overall yield but also aligns with modern green chemistry principles. For R&D directors and procurement specialists, this represents a pivotal opportunity to optimize the supply chain for high-purity pharmaceutical intermediates. The transition from oxidative cleavage methods to this constructive cyclization approach minimizes waste generation and relies on widely available commodity starting materials, thereby enhancing the economic viability of large-scale manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex bicyclic lactones required for protease inhibitors like Boceprevir has been plagued by significant technical and environmental hurdles. Prior art, such as the methods disclosed in Chinese patents CN201110442108.2 and CN201410423672.3, relied heavily on aggressive oxidative conditions. One prominent legacy route utilized ethyl chrysanthemate as a starting material, which is not only expensive but also subject to supply chain volatility due to its natural sourcing or complex synthesis. Furthermore, alternative routes employed potassium permanganate as a stoichiometric oxidant. While chemically effective, the use of permanganate generates substantial quantities of manganese dioxide sludge and acidic waste streams, creating a severe burden on wastewater treatment facilities and increasing the environmental compliance costs for manufacturers. Another approach involving ozonation, while cleaner in terms of byproducts, suffers from extremely high energy consumption and requires specialized, capital-intensive equipment to handle ozone safely on an industrial scale. These factors collectively result in a high cost of goods sold (COGS) and a fragile supply chain that is vulnerable to regulatory crackdowns on pollution.
The Novel Approach
In stark contrast, the methodology presented in patent CN115322167B introduces a paradigm shift by utilizing a sulfur-mediated cyclization strategy that circumvents the need for harsh oxidants. This novel approach initiates with the formation of a sulfonium salt from 2-bromopropane and dimethyl sulfide, two inexpensive and readily available bulk chemicals. The subsequent generation of a sulfur ylide under mild basic conditions allows for a highly selective reaction with maleic anhydride. This pathway eliminates the generation of heavy metal waste entirely, addressing the primary environmental pain point of the legacy permanganate routes. Moreover, the reaction conditions are remarkably mild, proceeding effectively at temperatures ranging from 0°C to 80°C, which reduces the energy load on the manufacturing plant compared to high-temperature pyrolysis or cryogenic ozonation. By simplifying the synthetic sequence to just three high-yielding steps, this method offers a reliable pharmaceutical intermediate supplier with a distinct competitive advantage in terms of both operational efficiency and sustainability metrics.
Mechanistic Insights into Sulfur Ylide-Mediated Cyclization
The core chemical innovation of this process lies in the generation and reactivity of the sulfur ylide intermediate, a powerful tool in organic synthesis for constructing strained ring systems. The mechanism begins with a nucleophilic substitution where dimethyl sulfide attacks 2-bromopropane to form a stable sulfonium salt (Intermediate I). This step is crucial as it activates the alpha-carbon for deprotonation. In the second stage, the addition of bases such as sodium hydroxide and potassium carbonate in a chloroform solvent facilitates the abstraction of a proton from the carbon adjacent to the sulfur atom. This generates the reactive sulfur ylide (Intermediate II), a species characterized by a negative charge on the carbon and a positive charge on the sulfur. This dipolar character makes the carbon highly nucleophilic, poised to attack electrophilic centers. The final step involves the reaction of this ylide with maleic anhydride. The nucleophilic carbon of the ylide attacks one of the carbonyl carbons of the anhydride, followed by an intramolecular displacement of the dimethyl sulfide group. This results in the formation of the desired bicyclic lactone structure with high stereochemical control. Understanding this mechanism is vital for process chemists aiming to replicate the high purity levels described in the patent, as controlling the concentration of the ylide is key to minimizing side reactions.
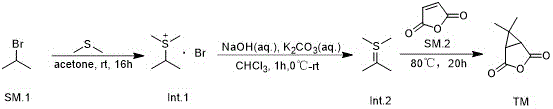
Furthermore, the choice of reagents plays a critical role in impurity control and overall process robustness. The use of a dual-base system (NaOH and K2CO3) in the ylide generation step ensures complete deprotonation while buffering the reaction medium to prevent the hydrolysis of the sensitive anhydride in the subsequent step. The patent data indicates that maintaining the reaction temperature between 0°C and room temperature during ylide formation is essential to prevent decomposition of the intermediate before it reacts with the maleic anhydride. In the final cyclization step, heating to 80°C provides the necessary activation energy to drive the ring closure to completion without degrading the product. This precise control over reaction parameters allows for the production of high-purity OLED material precursors or pharmaceutical intermediates with minimal chromatographic purification, often requiring only simple solvent rinses. For quality assurance teams, this mechanistic clarity translates to a well-defined Critical Process Parameter (CPP) space, ensuring batch-to-batch consistency and reducing the risk of unexpected impurities that could delay regulatory filings.
How to Synthesize Boceprevir Intermediate Efficiently
Implementing this synthesis route requires careful attention to solvent selection and stoichiometric ratios to maximize the yield at each stage. The patent provides specific guidance on using acetone for the initial salt formation and chloroform or dioxane for the subsequent steps, highlighting the importance of solvent polarity in stabilizing the ionic intermediates. Operators must ensure that the molar ratio of dimethyl sulfide to 2-bromopropane is maintained at approximately 2.5:1 to drive the first equilibrium forward. Following the isolation of the sulfonium salt, the base-mediated elimination must be performed under anhydrous conditions to preserve the reactivity of the ylide. The final coupling with maleic anhydride is the longest step, requiring up to 20 hours of reflux, which suggests that patience and thermal stability of the equipment are key operational requirements. For a detailed breakdown of the specific operational parameters and safety considerations, please refer to the standardized synthesis guide below.
- React 2-bromopropane with dimethyl sulfide in acetone at room temperature for 16 hours to form the sulfonium salt intermediate.
- Treat the sulfonium salt with sodium hydroxide and potassium carbonate in chloroform at 0°C to generate the sulfur ylide.
- React the generated ylide with maleic anhydride at 80°C for 20 hours to obtain the final bicyclic lactone product.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, the adoption of this synthetic route offers profound benefits that extend beyond simple yield improvements. The primary advantage lies in the radical simplification of the raw material portfolio. By replacing specialty starting materials like ethyl chrysanthemate with commodity chemicals such as 2-bromopropane and dimethyl sulfide, manufacturers can leverage existing global supply chains that are less prone to disruption. These bulk chemicals are produced in massive volumes for various industries, ensuring a stable supply and predictable pricing dynamics. This shift significantly mitigates the risk of raw material shortages that often plague the production of complex pharmaceutical intermediates. Additionally, the elimination of potassium permanganate removes the logistical and financial burden associated with the disposal of hazardous heavy metal waste. This not only reduces direct waste treatment costs but also streamlines the environmental permitting process, allowing for faster scale-up and continuous production without regulatory bottlenecks.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the removal of expensive reagents and the simplification of downstream processing. Traditional routes often require extensive workup procedures to remove metal residues, involving filtration, chelation, and multiple washes, all of which consume time, solvent, and labor. By utilizing a metal-free organocatalytic-style approach, this new method eliminates the need for expensive重金属 scavengers and the associated validation testing for residual metals. Furthermore, the high yield of the ylide formation step (reported up to 92%) ensures that the majority of the input material is converted into valuable intermediate, minimizing raw material waste. The cumulative effect of these efficiencies results in a substantially lower cost of goods, providing a competitive margin advantage for generic drug manufacturers.
- Enhanced Supply Chain Reliability: Supply chain resilience is paramount for life-saving antiviral medications. This synthesis route enhances reliability by decoupling production from the volatile markets of natural product derivatives. Since 2-bromopropane and maleic anhydride are petrochemical derivatives with established, multi-vendor supply bases, the risk of a single-source failure is drastically reduced. The robustness of the reaction conditions, which tolerate a range of temperatures and solvents (acetone, dioxane, THF), further adds to this reliability by allowing flexibility in manufacturing locations. Facilities can source solvents locally based on availability without compromising the reaction outcome. This flexibility ensures that production schedules can be maintained even during regional supply disruptions, guaranteeing a continuous flow of high-purity pharmaceutical intermediates to downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden challenges, but this three-step method is inherently designed for industrial expansion. The reactions do not require extreme pressures or cryogenic temperatures, meaning they can be executed in standard glass-lined or stainless steel reactors found in most multipurpose chemical plants. The absence of ozone generation equipment removes a significant capital expenditure barrier for scale-up. From an environmental standpoint, the process aligns with increasingly stringent global regulations regarding industrial emissions. By avoiding the generation of acidic waste streams containing manganese, the facility's effluent load is significantly lighter, reducing the complexity and cost of wastewater treatment. This 'green' profile not only ensures compliance with current EPA and REACH standards but also future-proofs the manufacturing site against tighter environmental legislation, securing long-term operational continuity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. These insights are derived directly from the experimental data and comparative analysis provided in patent CN115322167B. They are intended to assist technical decision-makers in evaluating the feasibility of adopting this method for their own production lines. Understanding the nuances of solvent recovery, yield optimization, and impurity profiles is essential for a successful technology transfer.
Q: What are the environmental advantages of this synthesis method compared to traditional routes?
A: Unlike traditional methods that utilize potassium permanganate which generates heavy waste acid pollution, or ozonation which consumes high electricity, this patented route utilizes common organic solvents and avoids heavy metal oxidants, resulting in a significantly greener production profile.
Q: Why is the sulfur ylide method considered more cost-effective for Boceprevir intermediates?
A: The process replaces expensive starting materials like ethyl chrysanthemate with commodity chemicals such as 2-bromopropane and dimethyl sulfide. Additionally, the elimination of complex purification steps associated with heavy metal removal drastically simplifies the downstream processing costs.
Q: Is this three-step process suitable for large-scale industrial production?
A: Yes, the patent explicitly states the method is suitable for industrial production due to its mild reaction conditions (room temperature to 80°C), high overall yield, and the use of standard solvents like acetone and chloroform which are easily managed in large-scale reactors.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Boceprevir Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and sustainable synthesis routes in the modern pharmaceutical landscape. As a leading CDMO partner, we possess the technical expertise to translate the innovations found in patent CN115322167B into commercial reality. Our facilities are equipped to handle the specific solvent systems and reaction conditions required for this sulfur ylide chemistry, ensuring that the transition from lab scale to pilot and commercial production is seamless. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, adhering to the highest international quality standards. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize advanced analytical techniques to verify the identity and purity of every batch, ensuring that the final intermediate meets the exacting requirements for antiviral drug synthesis.
We invite potential partners to engage with our technical team to explore how this optimized route can benefit your specific supply chain needs. Whether you are looking to reduce costs in API manufacturing or secure a more reliable source of critical intermediates, we are ready to assist. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our capabilities align with your strategic goals for high-quality pharmaceutical production.
