Advanced Three-Step Synthesis of Caronic Anhydride for Commercial Scale-up
Advanced Three-Step Synthesis of Caronic Anhydride for Commercial Scale-up
The pharmaceutical industry constantly seeks robust, scalable, and environmentally sustainable pathways for critical intermediates, particularly those serving high-value antiviral therapies. Patent CN102070575B introduces a transformative methodology for the synthesis of Caronic Anhydride, a pivotal building block in the production of Boceprevir, a potent protease inhibitor used in the treatment of Hepatitis C. This innovation addresses long-standing inefficiencies in traditional manufacturing by replacing hazardous oxidants and scarce starting materials with a streamlined, three-step sequence originating from methyl isobutenyl ketone. For R&D directors and procurement strategists, this patent represents a significant opportunity to optimize supply chains while adhering to stricter environmental regulations. The structural relationship between the target anhydride and the final drug substance underscores the importance of purity and stereochemical control in this synthesis.
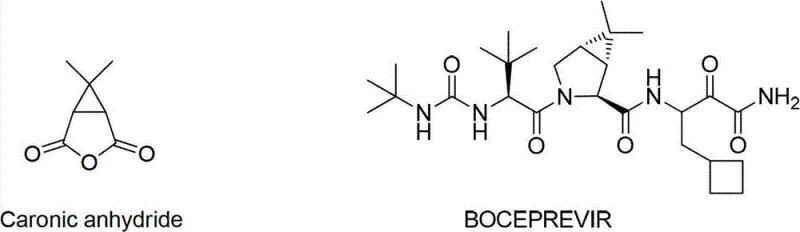
As a reliable pharmaceutical intermediate supplier, understanding the nuances of such process improvements is essential for maintaining competitive advantage. The shift from complex natural product derivatives to simple petrochemical feedstocks not only stabilizes pricing but also ensures consistent quality across batches. This report delves into the technical specifics of this novel route, analyzing its mechanistic advantages and commercial implications for global API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of Caronic Anhydride has relied heavily on the oxidation of ethyl chrysanthemate, a derivative of pyrethrum acids. As illustrated in the traditional pathway, this method necessitates the use of potassium permanganate (KMnO4) in acetone as the primary oxidizing system. While chemically effective, this approach presents severe operational drawbacks that hinder modern large-scale production. The reliance on KMnO4 generates substantial quantities of manganese dioxide sludge, a heavy metal waste product that requires complex and costly disposal procedures to meet environmental compliance standards. Furthermore, the use of acetone as a solvent in large-scale oxidation reactions introduces significant safety risks due to its high flammability and volatility, especially when heated during the reaction or recovery phases.
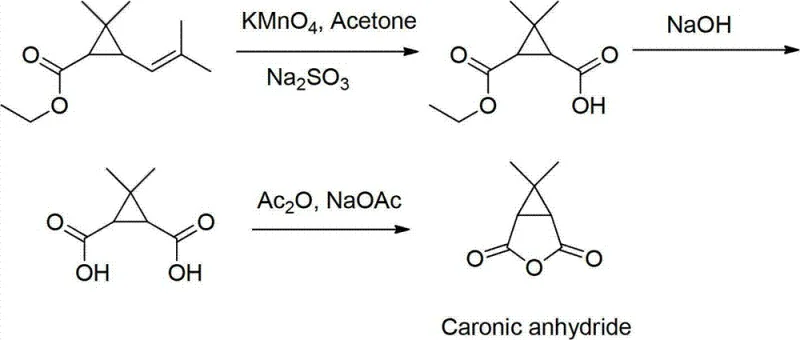
Beyond safety and environmental concerns, the economic viability of the conventional route is compromised by the raw material itself. Ethyl chrysanthemate is derived from natural sources or complex semi-synthetic pathways, resulting in limited availability and fluctuating market prices that can destabilize production budgets. The inability to recycle acetone effectively after the oxidation step further exacerbates cost inefficiencies, as fresh solvent must be continuously sourced. For supply chain heads, these factors create a fragile procurement landscape where lead times are unpredictable and regulatory scrutiny regarding heavy metal residues in the final API is intense. Consequently, there is a pressing industry demand for a synthetic alternative that circumvents these legacy issues.
The Novel Approach
The methodology disclosed in patent CN102070575B offers a compelling solution by fundamentally reimagining the carbon skeleton construction of the molecule. Instead of degrading a larger terpene structure, this novel approach builds the cyclopropane ring directly from methyl isobutenyl ketone, a low-cost and abundantly available industrial chemical. The process utilizes a sulfur ylide-mediated cyclopropanation followed by a mild oxidative hydrolysis using sodium hypochlorite (bleach), completely eliminating the need for heavy metal oxidants. This strategic shift not only simplifies the reaction workflow but also drastically reduces the environmental footprint of the manufacturing process. By avoiding manganese contamination, the downstream purification burden is significantly lightened, allowing for higher overall yields and superior product purity.
Moreover, the reaction conditions employed in this new route are notably milder, typically proceeding at temperatures between 20°C and 80°C, which enhances operational safety and reduces energy consumption compared to the vigorous conditions often required for permanganate oxidations. The use of aqueous sodium hypochlorite as an oxidant is particularly advantageous from a cost-reduction in API manufacturing perspective, as it is an inexpensive, commercially ubiquitous reagent that produces benign salt byproducts. This transition from a degradation-based strategy to a constructive synthesis enables a more predictable and scalable production model, aligning perfectly with the needs of high-volume pharmaceutical supply chains.
Mechanistic Insights into Sulfur Ylide-Mediated Cyclopropanation and Oxidation
The core of this innovative synthesis lies in the efficient construction of the gem-dimethyl cyclopropane ring, achieved through a reaction between methyl isobutenyl ketone and (ethoxy carbonyl methyl) dimethyl sulphide ylide. This transformation, akin to a Corey-Chaykovsky reaction, facilitates the formation of the strained three-membered ring with high regioselectivity. The mechanism involves the nucleophilic attack of the sulfur ylide on the electron-deficient double bond of the ketone, followed by intramolecular displacement of the dimethyl sulfide group to close the ring. This step is critical as it establishes the foundational carbon framework of the Caronic Anhydride precursor. The reaction is typically conducted at elevated temperatures, around 80°C, to ensure complete conversion, yielding the ester-functionalized cyclopropane intermediate in high efficiency.
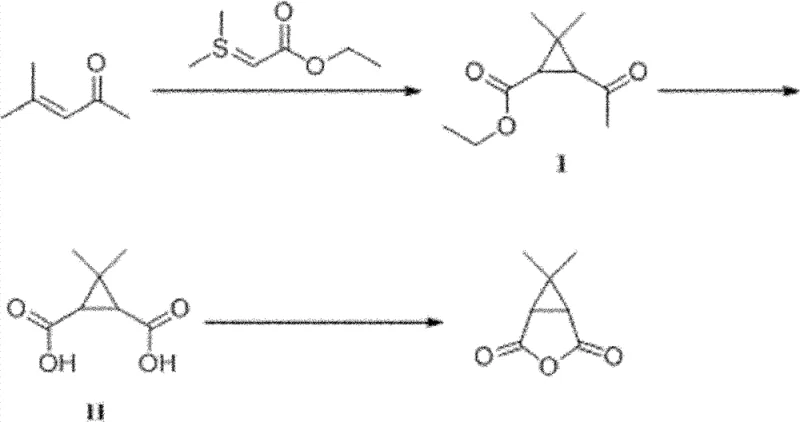
Following the ring formation, the subsequent oxidative hydrolysis step employs sodium hypochlorite to simultaneously cleave the acetyl group and hydrolyze the ester functionality, generating the dicarboxylic acid intermediate. This dual-function transformation is mechanistically elegant, utilizing the oxidizing power of hypochlorite to effect bond cleavage without over-oxidizing the sensitive cyclopropane ring. The final cyclization to the anhydride is driven by acetic anhydride under reflux conditions, which acts as both a dehydrating agent and a solvent. From an impurity control standpoint, this route is superior because it avoids the introduction of transition metals that are notoriously difficult to remove to ppm levels required for pharmaceutical grades. The absence of metal catalysts simplifies the workup procedure, often requiring only acidification and extraction, thereby minimizing the risk of introducing new contaminants during purification.
How to Synthesize Caronic Anhydride Efficiently
Implementing this synthesis in a pilot or commercial plant requires precise control over reaction parameters to maximize yield and safety. The process begins with the careful addition of the sulfur ylide to the ketone substrate, ensuring thermal management during the exothermic cyclopropanation phase. Subsequent oxidation must be monitored to prevent excessive chlorination, while the final dehydration step relies on efficient removal of acetic acid byproducts to drive the equilibrium toward the anhydride. The detailed standardized operating procedures for each stage, including specific stoichiometric ratios and isolation techniques, are critical for reproducibility.
- React methyl isobutenyl ketone with (ethoxy carbonyl methyl) dimethyl sulphide ylide at 20-80°C to form the cyclopropane intermediate.
- Perform oxidative hydrolysis on the intermediate using 5-15wt% aqueous sodium hypochlorite solution at room temperature to generate the diacid.
- Execute ring-closure by refluxing the diacid with acetic anhydride to yield the final Caronic Anhydride product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers tangible strategic benefits that extend beyond simple chemistry. The primary advantage lies in the decoupling of production costs from the volatile agricultural markets that influence the price of chrysanthemic acid derivatives. By switching to methyl isobutenyl ketone, a commodity chemical produced on a massive scale for other industries, manufacturers can secure a stable and predictable raw material supply. This stability is crucial for long-term contract negotiations and inventory planning, reducing the risk of production stoppages due to raw material shortages. Furthermore, the simplified waste profile of the new process translates directly into lower operational expenditures related to environmental compliance and waste disposal.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents like potassium permanganate and the reduction in solvent usage significantly lower the variable costs per kilogram of product. Since the process avoids the generation of heavy metal sludge, the costs associated with hazardous waste treatment and disposal are drastically minimized. Additionally, the high atom economy of the cyclopropanation step ensures that a greater proportion of raw materials end up in the final product, reducing material waste. These factors combine to create a leaner manufacturing process that improves overall profit margins without compromising on quality standards.
- Enhanced Supply Chain Reliability: Sourcing methyl isobutenyl ketone is far less complex than procuring specialized terpene esters, as it is available from multiple global suppliers with established logistics networks. This diversification of the supply base mitigates the risk of single-source dependency, ensuring continuous production even if one vendor faces disruptions. The robustness of the chemical process itself, characterized by mild conditions and tolerant reagents, further enhances reliability by reducing the likelihood of batch failures due to minor process deviations. Consequently, lead times for high-purity pharmaceutical intermediates can be shortened, allowing for more responsive fulfillment of customer demands.
- Scalability and Environmental Compliance: The absence of toxic heavy metals and the use of aqueous oxidation systems make this process inherently greener and easier to scale from laboratory to multi-ton production. Regulatory bodies increasingly favor manufacturing processes that minimize environmental impact, and this route aligns well with green chemistry principles by reducing solvent intensity and eliminating persistent pollutants. The ease of scale-up means that capacity can be expanded rapidly to meet surging market demand for Hepatitis C treatments or other applications of Caronic Anhydride. This scalability ensures that the supply chain remains agile and capable of adapting to dynamic market conditions.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the operational feasibility and strategic value of the new method for industry stakeholders. Understanding these details is vital for making informed decisions about technology transfer and process adoption.
Q: What are the primary safety advantages of the new Caronic Anhydride synthesis route?
A: The new method eliminates the use of potassium permanganate and large volumes of acetone, removing fire hazards and the generation of toxic manganese sludge associated with conventional oxidation processes.
Q: How does the raw material cost compare to traditional ethyl chrysanthemate routes?
A: By utilizing methyl isobutenyl ketone, a widely available and inexpensive commodity chemical, the process avoids the high cost and limited supply chain of ethyl chrysanthemate, leading to substantial raw material savings.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the mild reaction conditions (room temperature to 80°C), absence of heavy metal catalysts, and simplified purification steps make this route highly scalable and environmentally compliant for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Caronic Anhydride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of modern pharmaceutical development. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale innovation to industrial reality is seamless. We are committed to delivering Caronic Anhydride with stringent purity specifications, utilizing rigorous QC labs to verify that every batch meets the exacting standards required for API synthesis. Our infrastructure is designed to support the complex logistical needs of global clients, providing a secure and consistent supply of this essential building block.
We invite you to collaborate with us to leverage these advanced manufacturing capabilities for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain efficiency and reduce total landed costs.
