Advanced Boron-Mediated Synthesis of Natural Curcumin Compounds for Commercial Scale-Up
Advanced Boron-Mediated Synthesis of Natural Curcumin Compounds for Commercial Scale-Up
The global demand for high-purity curcuminoids, driven by their potent pharmacological properties including anti-inflammatory and antioxidant activities, has necessitated a shift from traditional extraction methods to robust synthetic pathways. Patent CN102911022A introduces a groundbreaking methodology for the artificial synthesis of natural curcumin compounds that addresses critical bottlenecks in yield and purity. This technical insight report analyzes the proprietary boron-mediated condensation strategy, which utilizes acetylacetone and boron trioxide to form a stable intermediate complex, subsequently reacting with benzaldehyde derivatives under mild conditions. For R&D directors and procurement specialists in the fine chemical sector, this patent represents a significant opportunity to secure a reliable pharmaceutical intermediates supplier capable of delivering consistent quality without the volatility associated with agricultural sourcing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industry has relied heavily on extracting curcumin from the dried rhizomes of Curcuma longa, a process fraught with inherent inefficiencies and supply chain vulnerabilities. The curcumin content in raw turmeric medicinal material is notoriously low, typically hovering around 1%, which necessitates the processing of massive volumes of biomass to achieve meaningful yields. Furthermore, the extraction process involves the use of large quantities of organic solvents, creating significant environmental burdens and safety hazards in manufacturing facilities. From a purification standpoint, separating individual curcuminoids with close structural similarities into monomers is exceptionally difficult, often requiring repetitive chromatography that drives up costs and extends production cycles. These factors combine to create a high-cost, low-yield operational model that is unfavorable for large-scale commercial production and fails to meet the stringent consistency requirements of modern pharmaceutical applications.
The Novel Approach
In stark contrast, the novel synthetic route detailed in the patent data offers a streamlined, chemically precise alternative that bypasses the limitations of biological variability. By employing a boron-complex mediated condensation, the process enables the direct assembly of the curcuminoid skeleton from simple, commodity-grade starting materials such as acetylacetone and vanillin or p-hydroxybenzaldehyde. This approach allows for the targeted synthesis of specific analogs, including curcumin, demethoxycurcumin, and bis-demethoxycurcumin, simply by adjusting the aldehyde feedstock. The reaction proceeds at room temperature, eliminating the need for energy-intensive heating or cooling systems, and utilizes ethyl acetate, a solvent with a favorable safety profile. This shift from extraction to synthesis not only stabilizes the supply chain against crop failures but also provides a level of molecular control that ensures batch-to-batch reproducibility essential for regulatory compliance.
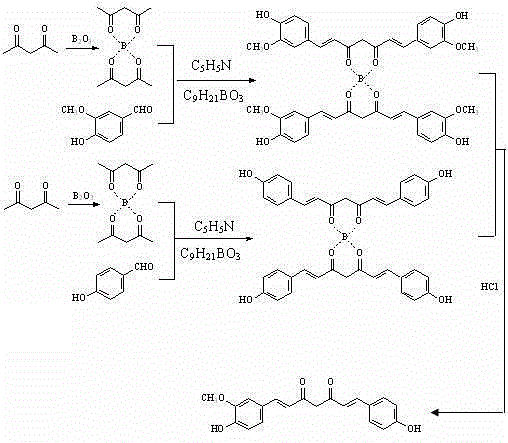
Mechanistic Insights into Boron-Mediated Condensation
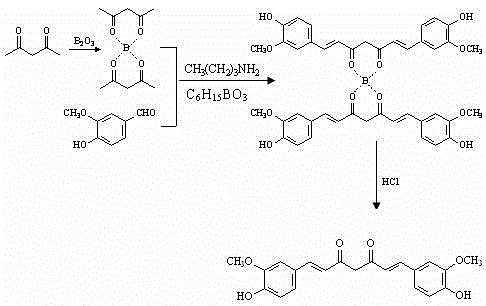
The core innovation of this technology lies in the formation and utilization of the acetylacetone-diboron trioxide complex, which serves as a highly reactive nucleophile in the subsequent aldol-type condensation. The initial step involves the stirring reaction of acetylacetone with diboron trioxide in ethyl acetate at room temperature, generating a stable boron-chelated species. This complexation activates the methylene group of the diketone, significantly enhancing its acidity and nucleophilicity towards the electrophilic carbonyl carbon of the benzaldehyde derivatives. The presence of a dehydrating agent, such as triethyl borate or tributyl borate, further drives the equilibrium towards the formation of the conjugated system by sequestering water produced during the condensation. This mechanistic pathway avoids the harsh basic conditions often used in traditional Claisen-Schmidt condensations, thereby minimizing side reactions such as polymerization or degradation of the sensitive phenolic groups.
Impurity control is meticulously managed through the final hydrolysis and recrystallization steps, which are critical for achieving the reported purity of greater than 98%. Upon completion of the room temperature stirring reaction, the addition of aqueous hydrochloric acid hydrolyzes the boron ester linkages, releasing the free curcumin compound and converting the boron species into boric acid, which can be easily removed. The crude product is then subjected to recrystallization using acetonitrile or an acetonitrile-water mixture with a volume percentage concentration of 80% to 99%. This specific solvent system is selected for its ability to differentially solubilize the target curcuminoid while precipitating structurally similar impurities and residual starting materials. The result is a crystalline product with a sharp melting point and high spectral purity, suitable for direct use in high-value pharmaceutical formulations without further extensive purification.
How to Synthesize Curcumin Efficiently
The synthesis protocol outlined in the patent provides a robust framework for laboratory and pilot-scale production, emphasizing simplicity and safety. The process begins with the preparation of the boron complex followed by the controlled addition of the aldehyde component and catalyst. Operators must ensure that all reagents, particularly the ethyl acetate and boron trioxide, are anhydrous to prevent premature hydrolysis of the active intermediate. The reaction mixture is stirred at ambient temperature for a duration ranging from 0.5 to 10 hours, depending on the specific derivative being synthesized, before quenching with acid. For a comprehensive understanding of the exact molar ratios, specific catalyst loading, and detailed workup procedures required to replicate these results, please refer to the standardized synthesis guide below.
- Prepare the acetylacetone-diboron trioxide solution by reacting acetylacetone with boron trioxide in ethyl acetate at room temperature.
- Mix the benzaldehyde derivative solution containing a dehydrating agent with the boron complex solution and add an organic amine catalyst.
- Hydrolyze the reaction mixture with hydrochloric acid, separate the organic layer, and recrystallize the product from acetonitrile to obtain high-purity curcumin.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this synthetic methodology offers profound strategic advantages beyond mere technical feasibility. The reliance on petrochemical-derived starting materials rather than agricultural crops decouples production from seasonal fluctuations, weather patterns, and geopolitical instability affecting spice-growing regions. This stability translates directly into more predictable lead times and the ability to enter into long-term supply contracts with fixed pricing structures, a critical factor for cost management in the pharmaceutical industry. Furthermore, the simplified downstream processing reduces the burden on waste treatment facilities, as the process avoids the generation of massive amounts of plant biomass waste and minimizes the use of hazardous extraction solvents.
- Cost Reduction in Manufacturing: The elimination of expensive and inefficient extraction steps leads to substantial cost savings in the overall manufacturing budget. By utilizing commodity chemicals like acetylacetone and vanillin, which are produced at massive scales globally, the raw material costs are significantly lower and more stable compared to purified turmeric oleoresin. Additionally, the room temperature reaction conditions drastically reduce energy consumption associated with heating and cooling, while the high selectivity of the boron-mediated reaction minimizes the loss of valuable materials to byproduct formation. These factors combine to create a highly cost-effective production model that enhances margin potential for downstream API manufacturers.
- Enhanced Supply Chain Reliability: Sourcing raw materials for this synthetic route is inherently more reliable than relying on agricultural harvests, as the supply chains for acetylacetone and benzaldehyde derivatives are well-established and diversified. This reduces the risk of supply disruptions caused by crop diseases or export restrictions, ensuring continuous operation of manufacturing facilities. The ability to produce specific curcuminoids on demand also allows for a more responsive supply chain that can adapt quickly to changes in market demand for specific analogs, such as the higher potency bis-demethoxycurcumin, without the need to stockpile varying grades of plant extracts.
- Scalability and Environmental Compliance: The process is designed for easy scale-up, utilizing standard reactor equipment and common solvents that pose minimal challenges in terms of heat transfer or mixing at large volumes. The use of ethyl acetate, a solvent with relatively low toxicity and favorable environmental regulations, simplifies compliance with increasingly stringent environmental protection standards. Moreover, the solid byproduct, boric acid, can be separated and potentially recycled or disposed of with greater ease than the complex organic sludge generated by traditional extraction methods, further reducing the environmental footprint and associated disposal costs of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this boron-mediated synthesis technology. These answers are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on purity standards, reaction conditions, and scalability potential. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing production portfolios.
Q: What are the purity levels achievable with this synthetic route?
A: The patented process consistently achieves purity levels higher than 98% through optimized recrystallization steps using acetonitrile solutions, eliminating the need for complex chromatographic separations often required in extraction methods.
Q: How does this method compare to traditional turmeric extraction?
A: Unlike traditional extraction which suffers from low curcumin content (approx. 1%) and difficult separation of structural analogs, this synthetic approach allows for the targeted production of specific curcuminoids like curcumin, demethoxycurcumin, and bis-demethoxycurcumin with controlled stoichiometry.
Q: Is the process scalable for industrial manufacturing?
A: Yes, the reaction operates at room temperature using common solvents like ethyl acetate and readily available raw materials, making it highly suitable for commercial scale-up without the need for extreme pressure or temperature equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Curcumin Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this boron-mediated synthetic route for the global curcumin market. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of synthetic curcumin meets the highest international standards for pharmaceutical and nutraceutical applications. We are committed to leveraging this advanced technology to provide our partners with a consistent, high-quality supply of curcumin intermediates.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis method can optimize your supply chain and reduce your overall cost of goods. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic benefits of switching from extraction-based sourcing to our synthetic solutions. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your unique project requirements, ensuring a partnership built on transparency, quality, and mutual growth.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
