Advanced Synthesis of Bridged Near-Infrared Fluorescent Dyes for Commercial Scale-Up
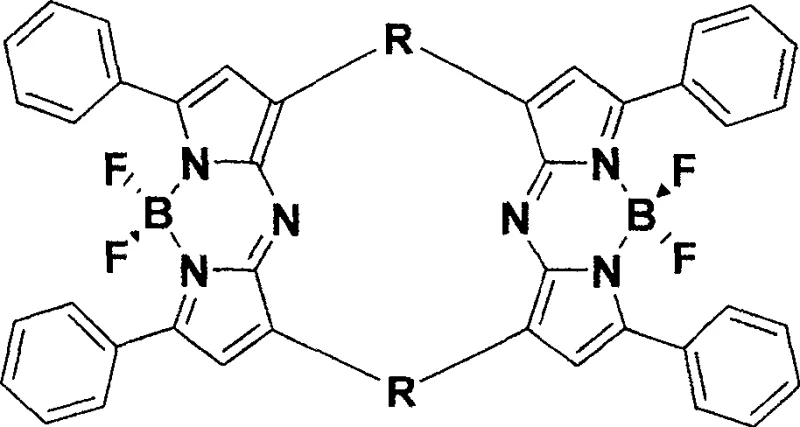
The landscape of optoelectronic materials and biological imaging is undergoing a significant transformation driven by the demand for superior near-infrared (NIR) fluorophores. Patent CN100487063C introduces a groundbreaking methodology for synthesizing a novel class of near-infrared fluorescent dyes characterized by a unique bridged structure connecting two pyrromethene nuclei. This architectural innovation directly addresses the critical limitations of existing dyes, such as low quantum yields and susceptibility to photobleaching, which have long hindered their application in high-precision fields like in vivo imaging and laser technology. By establishing a robust synthetic pathway that leverages accessible starting materials like acetophenone and terephthalaldehyde, this technology offers a viable route for the reliable near-infrared fluorescent dye supplier market to deliver products with enhanced stability and performance. The strategic importance of this patent lies not only in the molecular design but also in the practical feasibility of its four-step synthesis, which avoids extreme reaction conditions while ensuring high purity standards essential for electronic chemical manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the development of near-infrared fluorescent dyes has been plagued by inherent structural instabilities and suboptimal optical properties that restrict their utility in commercial applications. Traditional cyanine-based dyes, while widely used, frequently exhibit fluorescence quantum yields that are unacceptably low, often falling below 0.36, and in some standard cases like cyanine green, as low as 0.13. Furthermore, these conventional molecules are prone to aggregation and rapid photodegradation upon exposure to excitation sources, leading to signal loss and unreliable data in long-term imaging experiments. The synthesis of these older generations of dyes often involves complex multi-step sequences with poor atom economy, generating significant waste and requiring expensive purification protocols to remove toxic metal impurities. Additionally, the narrow Stokes shifts associated with many legacy dyes result in self-absorption issues, which drastically reduce detection sensitivity and complicate the optical setup for end-users in both medical and industrial sectors.
The Novel Approach
In stark contrast, the methodology outlined in the patent data presents a sophisticated yet streamlined approach that constructs a rigid, bridged macrocyclic framework to lock the fluorophore into a highly emissive state. This novel strategy utilizes a condensation-addition-cyclization-complexation sequence that effectively minimizes non-radiative decay pathways, thereby maximizing the fluorescence quantum yield and extending the fluorescence lifetime. By bridging two pyrromethene units through aromatic linkers such as phenylene or pyridine rings, the resulting molecule achieves exceptional rigidity, which significantly reduces vibrational energy loss and enhances photostability against bleaching. This structural reinforcement allows the dye to maintain consistent performance even under prolonged irradiation, a critical requirement for laser dyes and continuous monitoring sensors. Moreover, the versatility of the synthetic route permits the easy substitution of the bridging aldehyde component, enabling manufacturers to tailor the emission wavelengths across the near-infrared spectrum without redesigning the entire synthetic pathway, thus offering unparalleled flexibility for cost reduction in electronic chemical manufacturing.
Mechanistic Insights into Bridged Pyrromethene Cyclization
The core of this technological breakthrough lies in the precise orchestration of organic transformations that build the conjugated system step-by-step. The process initiates with a base-catalyzed carbonyl-aldehyde condensation between acetophenone and a dialdehyde, forming an α,β-unsaturated ketone intermediate under mild ambient conditions. This is followed by a Michael-type addition where nitromethane adds across the double bond, facilitated by a secondary amine catalyst like diethylamine, introducing the necessary nitrogen functionality for the subsequent ring closure. The critical cyclization step involves heating the nitro-ketone precursor with a large excess of ammonium acetate, which acts as both a nitrogen source and a buffer, promoting the formation of the pyrrole rings through a Paal-Knorr-like mechanism. Finally, the stabilization of the dipyrromethene ligand is achieved through coordination with boron trifluoride etherate in the presence of a base, locking the planar structure and completing the BODIPY-like core.
Beyond the primary reaction pathway, the mechanism inherently supports rigorous impurity control, which is paramount for R&D directors focused on product consistency. The use of recrystallization and column chromatography at intermediate stages ensures that side products, such as incomplete condensation oligomers or unreacted starting materials, are systematically removed before the final complexation step. The choice of solvents, transitioning from polar protic media like ethanol and methanol in the early stages to non-polar dichloromethane in the final step, facilitates the selective precipitation of the desired intermediates, effectively purifying the stream without the need for excessive chromatographic burden. This staged purification strategy minimizes the carryover of colored impurities that could otherwise quench fluorescence or interfere with downstream bioconjugation processes. Furthermore, the inert atmosphere protection during the boron complexation prevents oxidative degradation of the sensitive pyrromethene intermediate, ensuring that the final product retains its intended optical properties and chemical integrity.
How to Synthesize Bridged Near-Infrared Fluorescent Dye Efficiently
Implementing this synthesis requires careful attention to stoichiometric ratios and reaction times to maximize yield and purity. The process is designed to be scalable, moving from gram-scale laboratory optimization to kilogram-level production with minimal modification to the core protocol. Operators must ensure strict temperature control during the exothermic addition phases and maintain anhydrous conditions during the final boron complexation to prevent hydrolysis of the BF2 group. The following guide outlines the standardized operational parameters derived from the patent examples to ensure reproducible results.
- Perform carbonyl-aldehyde condensation between acetophenone and terephthalaldehyde under alkaline conditions at room temperature.
- Execute a catalytic addition reaction using nitromethane in methanol with diethylamine, followed by reflux.
- Conduct cyclization using ammonium acetate in ethanol or melt form to obtain the blue solid intermediate.
- Complete the synthesis by complexing with boron trifluoride etherate in dichloromethane under inert gas protection.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route represents a strategic opportunity to optimize sourcing strategies and reduce dependency on volatile specialty chemical markets. The reliance on bulk commodity chemicals such as acetophenone, benzaldehydes, and ammonium acetate means that raw material costs are inherently stable and predictable, shielding the production budget from the price fluctuations often seen with exotic organometallic catalysts. The elimination of precious metal catalysts, which are common in alternative cross-coupling approaches for similar structures, removes the need for costly metal scavenging steps and simplifies the waste treatment process, leading to substantial cost savings in overall manufacturing operations. Additionally, the robustness of the reaction conditions, which do not require high-pressure reactors or cryogenic temperatures, allows for production in standard glass-lined steel reactors, enhancing the agility of the supply chain to respond to sudden increases in demand.
The scalability of this process is further reinforced by the simplicity of the workup procedures, which primarily involve filtration and solvent evaporation rather than complex distillation or extraction trains. This operational simplicity translates directly into reduced lead time for high-purity near-infrared fluorescent dyes, as the turnaround time between batches can be significantly shortened compared to more labor-intensive synthetic routes. From an environmental compliance perspective, the use of ethanol and water in the early stages aligns well with green chemistry initiatives, reducing the volume of hazardous halogenated waste generated per kilogram of product. The ability to substitute solvents, such as using n-butanol instead of ethanol or ammonium formate instead of ammonium acetate, provides further flexibility to adapt to local regulatory constraints or raw material availability without compromising the quality of the final commercial scale-up of complex optoelectronic materials.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and application of this patented technology. These insights are derived directly from the experimental data and background analysis provided in the patent documentation to assist decision-makers in evaluating the feasibility of integration.
Q: What are the primary advantages of this bridged dye structure over traditional cyanine dyes?
A: Unlike traditional cyanine dyes which often suffer from low quantum yields (below 0.36) and poor photostability, this bridged pyrromethene structure offers significantly higher fluorescence quantum yields, narrower half-peak widths, and superior chemical and optical stability, making it ideal for demanding bio-imaging and laser applications.
Q: Can the emission wavelength be tuned for specific electronic applications?
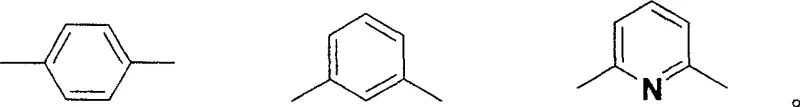
A: Yes, the synthesis allows for structural modulation by replacing the bridging aldehyde component. Using isophthalaldehyde, o-phthalaldehyde, or 2,6-diformylpyridine instead of terephthalaldehyde enables precise tuning of the spectral properties to match specific optoelectronic requirements.
Q: Is this synthesis route scalable for industrial production?
A: The process utilizes readily available commodity chemicals such as acetophenone and common aldehydes, and operates under relatively mild conditions (room temperature to reflux), indicating strong potential for commercial scale-up without requiring exotic high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Near-Infrared Fluorescent Dye Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance optical materials play in advancing next-generation diagnostic and electronic technologies. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We are committed to maintaining stringent purity specifications through our rigorous QC labs, utilizing advanced HPLC and spectroscopic analysis to guarantee that every batch of near-infrared fluorescent dye meets the exacting standards required for sensitive biological and electronic applications. Our infrastructure is specifically designed to handle the nuanced requirements of fluorophore synthesis, including inert atmosphere handling and precise temperature control, delivering a product that consistently outperforms market alternatives.
We invite global partners to collaborate with us to unlock the full potential of this innovative dye class for their specific product lines. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements, along with specific COA data and route feasibility assessments to validate the fit for your manufacturing ecosystem. Whether you are developing new laser systems or advanced bio-imaging probes, our expertise ensures a secure and high-quality supply chain foundation.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
