Scalable Green Synthesis of (E) 1-Acyl-2-(2-(9-alkyl)carbazole-3-)vinyl-benzimidazole Intermediates
Introduction to Advanced Carbazole-Benzimidazole Hybrids
The pharmaceutical and fine chemical industries are constantly seeking novel heterocyclic scaffolds that combine the unique electronic properties of carbazole with the biological activity of benzimidazole. Patent CN111233839B discloses a significant breakthrough in this domain by introducing a robust preparation method for (E) 1-acyl-2-(2-(9-alkyl)carbazole-3-)vinyl-benzimidazole derivatives. These compounds represent a class of large conjugated system molecules with rigid condensed ring structures, offering relatively strong intramolecular charge transfer capacity and distinct photoelectric properties. As shown in the general structural formula below, the versatility of the R and R' groups allows for extensive chemical modification to tailor specific substituent groups for diverse applications ranging from organic optical materials to potent antibacterial agents.
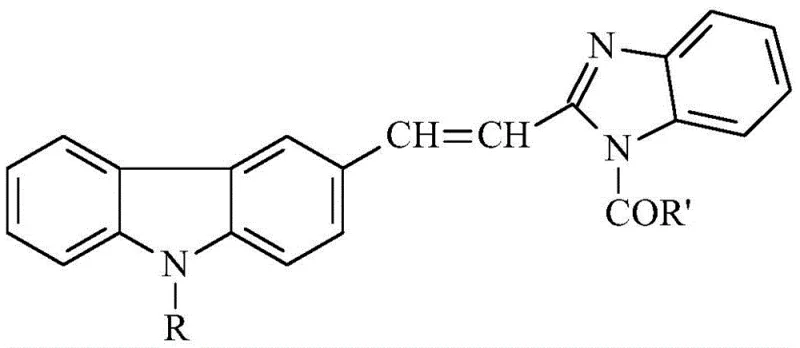
The strategic integration of the benzimidazole ring into the carbazolyl framework generates a novel compound with special photoelectric activity, which is of immense research significance for developing next-generation functional materials and active pharmaceutical ingredients (APIs). The disclosed technology not only expands the library of available carbazole derivatives but also provides a scalable pathway for producing high-purity intermediates essential for the synthesis of insect repellents, bactericides, and metal anticorrosive agents. For R&D directors and procurement specialists, understanding the nuances of this synthesis is critical for securing a reliable supply chain of these high-value specialty chemicals.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the synthesis of complex N-acylated benzimidazole derivatives has been plagued by several inefficiencies that hinder commercial viability. Conventional acylation protocols often rely on expensive coupling reagents such as DCC (dicyclohexylcarbodiimide) or EDC (1-ethyl-3-(3-dimethylaminopropyl)carbodiimide), which generate stoichiometric amounts of urea byproducts that are notoriously difficult to remove from the final product matrix. Furthermore, many existing routes require harsh reaction conditions, including the use of strong bases or sensitive organometallic catalysts that demand strict anhydrous environments and cryogenic temperatures, thereby escalating energy consumption and operational complexity. The purification of intermediates in these traditional pathways frequently involves multiple chromatographic steps, leading to significant yield erosion and increased solvent waste, which contradicts modern green chemistry principles and inflates the cost of goods sold (COGS) for the final API intermediate.
The Novel Approach
In stark contrast, the methodology outlined in patent CN111233839B offers a streamlined and economically superior alternative that bypasses these historical bottlenecks. The novel approach utilizes a direct reflux reaction between the (E)2-(2-(9-alkyl)carbazole-3-)vinyl-benzimidazole precursor and simple carboxylic acids or acid anhydrides in acetonitrile, eliminating the need for exotic coupling agents entirely. This simplification of the reaction manifold allows for a straightforward workup procedure where the majority of the solvent is evaporated, and the product precipitates as a solid that can be isolated via simple suction filtration and drying. The result is a process that consistently delivers yields exceeding 90%, with the final products exhibiting high purity and thermal stability (melting points >260°C), making it an ideal candidate for cost reduction in fine chemical manufacturing and large-scale industrial adoption.
Mechanistic Insights into the Multi-Step Synthesis Strategy
The synthetic route described in the patent is a masterpiece of stepwise construction, beginning with the formation of the acrylic acid linker via a green Knoevenagel condensation. As illustrated in the reaction scheme below, 3-formyl-9-alkyl carbazole reacts with malonic acid under the catalysis of a choline chloride-urea deep eutectic solvent (DES) at a mild temperature of approximately 50°C. This choice of catalyst is particularly ingenious; the DES acts as both a solvent and a catalyst, facilitating the condensation through hydrogen bonding networks that activate the carbonyl group of the aldehyde and stabilize the transition state, thereby avoiding the use of volatile organic solvents and toxic piperidine catalysts typically associated with this transformation. The resulting (E)1-(9-alkyl-carbazole-3-)-acrylic acid serves as a crucial building block, establishing the conjugated vinyl bridge that connects the electron-rich carbazole donor to the future benzimidazole acceptor.
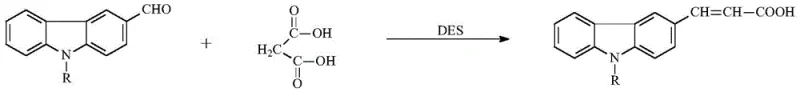
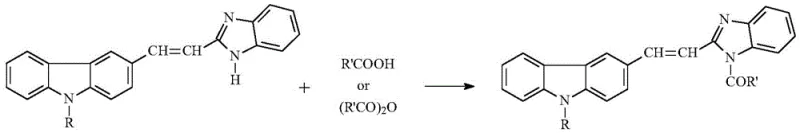
Following the formation of the acrylic acid intermediate, the synthesis proceeds through a cyclization step where the acid reacts with o-phenylenediamine in the presence of phosphorus oxychloride (POCl3) or concentrated hydrochloric acid at temperatures between 80-105°C. This step constructs the benzimidazole ring fused to the vinyl linkage, creating the core (E)2-(2-(9-alkyl)carbazole-3-)vinyl-benzimidazole scaffold. The final mechanistic transformation involves the nucleophilic attack of the benzimidazole nitrogen on the carbonyl carbon of the acylating agent (carboxylic acid or anhydride). As depicted in the final reaction equation, this acylation occurs efficiently under reflux in acetonitrile, driven by the thermal energy that overcomes the activation barrier for the nucleophilic substitution. The simplicity of this mechanism ensures minimal side reactions, such as polymerization or hydrolysis, resulting in a clean impurity profile that is highly desirable for pharmaceutical applications.
How to Synthesize (E) 1-Acyl-2-(2-(9-alkyl)carbazole-3-)vinyl-benzimidazole Efficiently
Executing this synthesis requires precise control over reaction parameters to maximize the yield and purity of the final dark green solid. The process begins with the preparation of the vinyl-benzimidazole precursor, which must be thoroughly dried before entering the final acylation step to prevent hydrolysis of the acylating agent. Operators should sequentially add the precursor, the carboxylic acid (or anhydride), and acetonitrile into a dry three-neck flask equipped with a reflux condenser and a magnetic stirrer. The mixture is then heated to reflux, and the reaction progress is meticulously monitored using Thin Layer Chromatography (TLC) with a petroleum ether and ethyl acetate (3:1 v/v) developing system until the starting material is completely consumed. Upon completion, the bulk of the acetonitrile solvent is removed under reduced pressure, inducing the precipitation of the product, which is then collected by filtration and dried to constant weight. For a detailed, step-by-step Standard Operating Procedure (SOP) including specific molar ratios and safety precautions, please refer to the technical guide below.
- Synthesize (E)1-(9-alkyl-carbazole-3-)-acrylic acid via Knoevenagel condensation using choline chloride-urea eutectic solvent at 50°C.
- Perform cyclization with o-phenylenediamine in phosphorus oxychloride at 80-105°C to form the vinyl-benzimidazole core.
- Execute final acylation by refluxing the vinyl-benzimidazole precursor with carboxylic acid or anhydride in acetonitrile.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented synthesis route offers transformative benefits that directly impact the bottom line and operational resilience. The elimination of expensive coupling reagents and the use of commodity chemicals like acetonitrile and carboxylic acids drastically simplify the raw material sourcing strategy, reducing dependency on specialized chemical suppliers and mitigating the risk of supply disruptions. Furthermore, the high yield (>90%) and simple isolation method (filtration) mean that less raw material is required to produce the same amount of product, effectively lowering the unit cost of production without compromising on quality. The robustness of the process also translates to reduced batch failure rates, ensuring a consistent and reliable flow of high-purity pharmaceutical intermediates to downstream manufacturing units.
- Cost Reduction in Manufacturing: The economic advantage of this process is primarily derived from the substitution of costly activation reagents with inexpensive carboxylic acids and the implementation of a solvent system that allows for easy recovery and recycling. By removing the need for chromatographic purification and complex aqueous workups, the facility can significantly reduce solvent consumption and waste disposal fees, leading to substantial cost savings in the overall manufacturing budget. Additionally, the high thermal stability of the product minimizes degradation losses during drying and packaging, further enhancing the effective yield and profitability of the production campaign.
- Enhanced Supply Chain Reliability: The reliance on readily available starting materials such as substituted carbazoles, malonic acid, and o-phenylenediamine ensures that the supply chain is not vulnerable to the bottlenecks often associated with exotic catalysts or custom-synthesized reagents. This accessibility allows for rapid scaling of production capacity in response to market demand fluctuations, providing a strategic buffer against global supply chain volatility. Moreover, the simplicity of the reaction conditions reduces the technical barrier for contract manufacturing organizations (CMOs), enabling a broader base of qualified suppliers to produce these intermediates, thereby diversifying the supply source and enhancing security of supply.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, the use of Deep Eutectic Solvents (DES) in the early stages and acetonitrile in the final step aligns well with green chemistry initiatives, simplifying the permitting process for new production lines. The absence of heavy metal catalysts eliminates the need for expensive and time-consuming metal scavenging steps, which are often a bottleneck in API manufacturing. The process generates minimal hazardous waste, and the solid product isolation method reduces the volume of liquid effluent, making it easier to meet stringent environmental discharge standards and ensuring long-term operational sustainability.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these carbazole-benzimidazole derivatives. The answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a factual basis for decision-making. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing production portfolios or for specifying the correct grade of material for research and development purposes.
Q: What are the primary advantages of the DES-catalyzed route for the acrylic acid intermediate?
A: The use of choline chloride-urea deep eutectic solvents eliminates the need for volatile organic solvents and toxic catalysts in the initial condensation step, significantly reducing environmental impact and waste treatment costs while maintaining high reaction efficiency at mild temperatures (50°C).
Q: How does the novel acylation method improve yield compared to traditional coupling methods?
A: By utilizing direct reflux with carboxylic acids or anhydrides in acetonitrile rather than expensive peptide coupling reagents, the process achieves yields exceeding 90% with a simplified workup procedure involving solvent evaporation and filtration, thereby minimizing material loss.
Q: Is this synthesis route suitable for large-scale commercial production?
A: Yes, the process relies on standard unit operations such as reflux, evaporation, and filtration without requiring cryogenic conditions or sensitive organometallic catalysts, making it highly robust and easily transferable from laboratory to multi-ton industrial scale.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (E) 1-Acyl-2-(2-(9-alkyl)carbazole-3-)vinyl-benzimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of novel therapeutics and advanced materials. Our team of expert chemists has extensively analyzed the synthetic route disclosed in CN111233839B and possesses the technical capability to optimize and scale this process to meet your specific requirements. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can support your needs from early-stage clinical trials through to full-scale commercial launch. Our state-of-the-art facilities are equipped with rigorous QC labs and advanced analytical instrumentation to guarantee stringent purity specifications, delivering materials that consistently meet the highest industry standards for pharmaceutical and fine chemical applications.
We invite you to collaborate with us to leverage this innovative synthesis technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to reach out today to discuss your project needs, obtain specific COA data for our available batches, and receive comprehensive route feasibility assessments that will accelerate your time-to-market and enhance your competitive advantage in the global marketplace.
