Scalable Synthesis of (E) 1-Acyl-2-(2-(9-alkyl)carbazole-3-)vinyl-benzimidazole for Advanced Applications
Scalable Synthesis of (E) 1-Acyl-2-(2-(9-alkyl)carbazole-3-)vinyl-benzimidazole for Advanced Applications
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes for complex heterocyclic compounds that offer both high purity and economic viability. Patent CN111233839A introduces a significant advancement in the preparation of (E) 1-acyl-2-(2-(9-alkyl)carbazole-3-)vinyl-benzimidazole derivatives. These molecules represent a sophisticated class of conjugated systems, merging the rigid, electron-rich architecture of carbazole with the bioactive potential of the benzimidazole scaffold. Such structures are increasingly valued not only as critical pharmaceutical intermediates for developing novel antimicrobial and anticancer agents but also as key components in organic optoelectronic materials due to their exceptional charge transfer capabilities. The disclosed methodology addresses long-standing challenges in synthesizing these large conjugated systems by providing a pathway that is operationally simple, environmentally considerate, and capable of delivering yields exceeding 90%. For R&D teams and procurement specialists alike, understanding the nuances of this technology is essential for securing a reliable pharmaceutical intermediate supplier partnership that can deliver high-quality materials consistently.
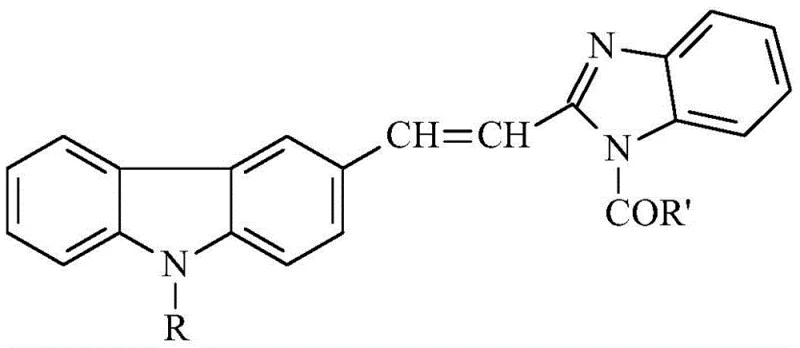
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of benzimidazole rings, particularly at the N-1 position when linked to sensitive vinyl-carbazole systems, has presented significant synthetic hurdles. Traditional acylation protocols often necessitate the use of aggressive activating agents, such as thionyl chloride or oxalyl chloride, to generate reactive acid chlorides in situ. These reagents are not only hazardous and difficult to handle on a large scale but also generate stoichiometric amounts of corrosive waste streams that complicate downstream processing. Furthermore, conventional methods frequently suffer from poor regioselectivity or side reactions involving the vinyl double bond, leading to complex impurity profiles that require extensive chromatographic purification. This reliance on multi-step activation and purification drives up the cost of goods sold (COGS) and extends lead times, creating bottlenecks for cost reduction in API manufacturing. Additionally, the sensitivity of the (E)-vinyl linkage to harsh acidic or basic conditions often results in isomerization or decomposition, severely impacting the overall yield and optical purity of the final product.
The Novel Approach
In stark contrast, the methodology outlined in CN111233839A offers a streamlined, direct acylation strategy that circumvents the need for pre-activation of the carboxylic acid. By utilizing the parent carboxylic acid or its corresponding anhydride directly in a refluxing acetonitrile medium, the process achieves efficient N-acylation of the benzimidazole nitrogen. This approach eliminates the generation of hazardous gaseous byproducts and significantly reduces the E-factor of the synthesis. The reaction conditions are remarkably mild yet effective, preserving the integrity of the sensitive (E)-vinyl bridge and the extended pi-conjugated system. As illustrated in the reaction scheme below, the transformation proceeds cleanly to afford the target (E) 1-acyl-2-(2-(9-alkyl)carbazole-3-)vinyl-benzimidazole with minimal byproduct formation. The simplicity of the workup procedure, which involves merely evaporating the solvent and filtering the precipitated solid, represents a paradigm shift towards greener chemistry. This operational simplicity translates directly into substantial cost reduction in pharmaceutical intermediate manufacturing, making it an attractive route for commercial adoption.
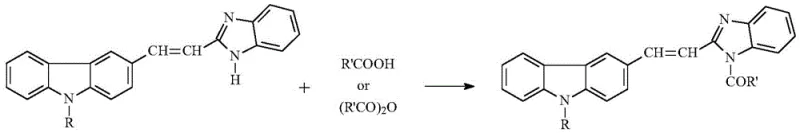
Mechanistic Insights into Direct N-Acylation
The success of this synthetic route relies on the inherent nucleophilicity of the benzimidazole nitrogen atom within the specific electronic environment created by the 2-vinyl-carbazole substituent. Under reflux conditions in a polar aprotic solvent like acetonitrile, the thermal energy provided is sufficient to overcome the activation barrier for the nucleophilic attack of the benzimidazole N-H on the carbonyl carbon of the carboxylic acid or anhydride. In the case of anhydrides, the reaction is driven by the formation of a stable carboxylic acid byproduct, while direct acid acylation likely proceeds through a thermodynamic equilibrium that favors the amide bond formation due to the precipitation of the product from the reaction mixture. This precipitation acts as a driving force, pulling the equilibrium towards completion and preventing reverse hydrolysis. The rigidity of the carbazole-benzimidazole conjugated system likely stabilizes the transition state, facilitating the reaction without the need for external catalysts. For process chemists, understanding this mechanism is crucial for optimizing reaction parameters such as temperature and concentration to maximize throughput.
From an impurity control perspective, the mechanism inherently limits the formation of common side products. Since no strong activating agents are used, there is no risk of chlorination of the aromatic rings or the vinyl group, which are common pitfalls in traditional methods. The primary impurities would theoretically be unreacted starting materials or symmetric anhydrides if formed in situ, both of which are easily removed during the filtration and washing steps. The high crystallinity of the resulting products, evidenced by sharp melting points reported in the patent examples (often >260°C), suggests a highly ordered lattice structure that naturally excludes impurities during crystal growth. This self-purification effect is a significant advantage for ensuring high-purity carbazole derivatives suitable for sensitive biological assays or electronic applications, reducing the burden on analytical QC laboratories.
How to Synthesize (E) 1-Acyl-2-(2-(9-alkyl)carbazole-3-)vinyl-benzimidazole Efficiently
Implementing this synthesis in a pilot or production plant requires strict adherence to the optimized parameters defined in the patent to ensure reproducibility and safety. The process begins with the precise charging of stoichiometric amounts of the benzimidazole precursor and the acylating agent into a dry reactor to prevent hydrolysis. The choice of acetonitrile as the solvent is critical, as it provides the necessary solubility at reflux temperatures while allowing the product to crash out upon cooling or concentration. Operators must monitor the reaction progress closely using Thin Layer Chromatography (TLC) with a petroleum ether and ethyl acetate system to determine the exact endpoint, preventing over-reaction or degradation. Once the reaction is deemed complete, the efficient removal of the solvent is key to maximizing recovery. The following guide outlines the standardized operational steps derived from the patent data to assist your technical team in replicating this high-yield process.
- Charge a dry three-neck flask with (E)2-(2-(9-alkyl)carbazole-3-)vinyl-benzimidazole, carboxylic acid (or anhydride), and acetonitrile.
- Heat the mixture to reflux and monitor reaction progress via TLC until completion.
- Evaporate the majority of the solvent, filter the precipitated solid, and dry to obtain the pure product with yields exceeding 90%.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers compelling strategic advantages that extend beyond simple technical feasibility. The elimination of hazardous reagents and complex purification steps fundamentally alters the cost structure of producing these valuable intermediates. By simplifying the manufacturing workflow, companies can achieve faster batch turnover times and reduce the dependency on specialized equipment required for handling corrosive gases. This operational efficiency translates into a more resilient supply chain capable of responding rapidly to market demands. Furthermore, the versatility of the method allows for the production of a diverse library of derivatives by simply varying the carboxylic acid component, providing flexibility in product offerings without requiring significant retooling of the production line.
- Cost Reduction in Manufacturing: The most immediate financial benefit arises from the drastic simplification of the reaction protocol. By avoiding the use of expensive coupling reagents like DCC or EDC, and eliminating the need for acid chloride formation, the raw material costs are significantly lowered. Additionally, the workup procedure requires no aqueous extraction or column chromatography; the product is isolated via simple filtration. This reduction in unit operations leads to lower labor costs, reduced solvent consumption, and minimized waste disposal fees. The high yields reported (consistently above 90%) mean that less starting material is wasted, further enhancing the overall atom economy and profitability of the process. These factors combined result in a highly competitive pricing structure for the final pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The robustness of this synthetic method ensures consistent supply continuity. The starting materials, including various 9-alkyl-carbazole derivatives and common carboxylic acids, are readily available from global chemical suppliers, mitigating the risk of raw material shortages. The process tolerance is high, meaning that minor fluctuations in reaction conditions are unlikely to result in batch failures, thereby guaranteeing on-time delivery to customers. For supply chain heads, this reliability is paramount in maintaining uninterrupted production schedules for downstream API manufacturing. The ability to scale this reaction from gram to kilogram quantities without losing efficiency demonstrates the commercial scale-up of complex heterocycles is feasible and low-risk.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental sustainability intensify, this green chemistry approach positions manufacturers favorably. The absence of heavy metal catalysts and halogenated waste streams simplifies compliance with stringent environmental regulations. The use of acetonitrile, a solvent that can be efficiently recovered and recycled, further reduces the environmental footprint. Scalability is inherently supported by the use of standard reflux equipment found in most multipurpose chemical plants. This ease of scale-up allows for the rapid expansion of production capacity to meet surging demand for high-purity carbazole derivatives in the OLED and pharmaceutical sectors, ensuring long-term supply security.
Frequently Asked Questions (FAQ)
To further clarify the technical and commercial implications of this technology, we have compiled answers to common inquiries from our industry partners. These questions address the specific capabilities of the synthesis method, the scope of applicable substituents, and the quality standards achievable. Understanding these details is vital for R&D directors evaluating this route for their pipeline projects and for procurement teams assessing vendor qualifications. The following insights are derived directly from the experimental data and claims within the patent documentation.
Q: What are the primary advantages of the acylation method described in CN111233839A?
A: The method utilizes a simple reflux process in acetonitrile without complex catalysts, achieving yields over 90% and simplifying purification to mere filtration.
Q: Can the alkyl chain length on the carbazole nitrogen be varied?
A: Yes, the patent specifies a wide range of R groups from methyl to hexadecyl, allowing for tunable lipophilicity and electronic properties.
Q: Is this intermediate suitable for large-scale pharmaceutical production?
A: Absolutely. The use of common solvents like acetonitrile and standard reflux conditions makes the process highly amenable to commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (E) 1-Acyl-2-(2-(9-alkyl)carbazole-3-)vinyl-benzimidazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation therapeutics and advanced materials. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising laboratory results of CN111233839A can be seamlessly translated into industrial reality. We are committed to maintaining stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify the structural integrity and purity of every batch of (E) 1-acyl-2-(2-(9-alkyl)carbazole-3-)vinyl-benzimidazole we produce. Our facility is equipped to handle the specific solvent recovery and filtration requirements of this process, guaranteeing a consistent supply of material that meets the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to leverage this innovative synthesis technology for your specific applications. Whether you require custom derivatives with specific alkyl chain lengths or large volumes of the standard compounds, our team is ready to provide a Customized Cost-Saving Analysis tailored to your project needs. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us be your partner in accelerating your R&D timeline and optimizing your supply chain for these high-value carbazole-benzimidazole derivatives.
